Region:Asia
Author(s):Dev
Product Code:KRAA3777
Pages:97
Published On:January 2026
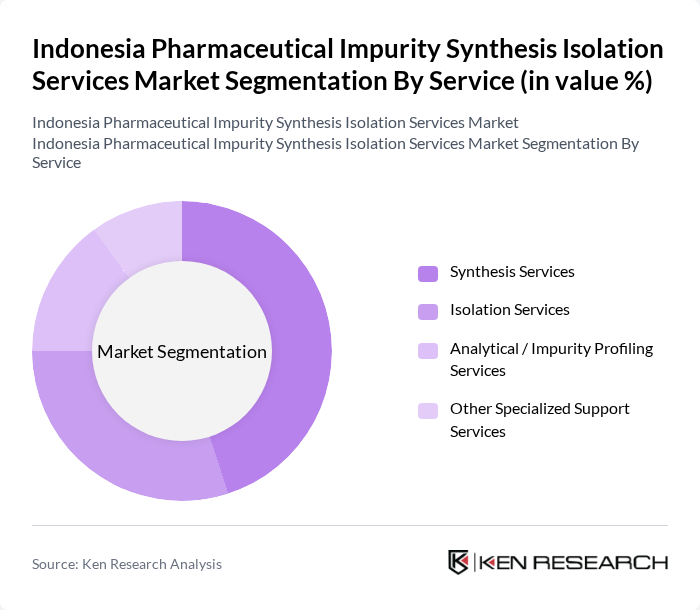
By Service:The services offered in this market include Synthesis Services, Isolation Services, Analytical / Impurity Profiling Services, and Other Specialized Support Services. Among these, Synthesis Services are currently leading due to the increasing need for customized synthesis of pharmaceutical compounds and reference standards, driven by the growing demand for novel drug formulations and complex APIs in Indonesia’s expanding API and finished dose markets. Analytical / Impurity Profiling Services are gaining traction in line with global trends where analytical services represent one of the fastest?growing segments, supported by investments in advanced LC?MS, GC?MS, NMR, and hyphenated techniques for comprehensive impurity characterization. Isolation Services also hold significant importance as they ensure the purity and safety of pharmaceutical products, which is critical for regulatory compliance and for supporting local manufacturers seeking to meet international pharmacopeial and export standards.
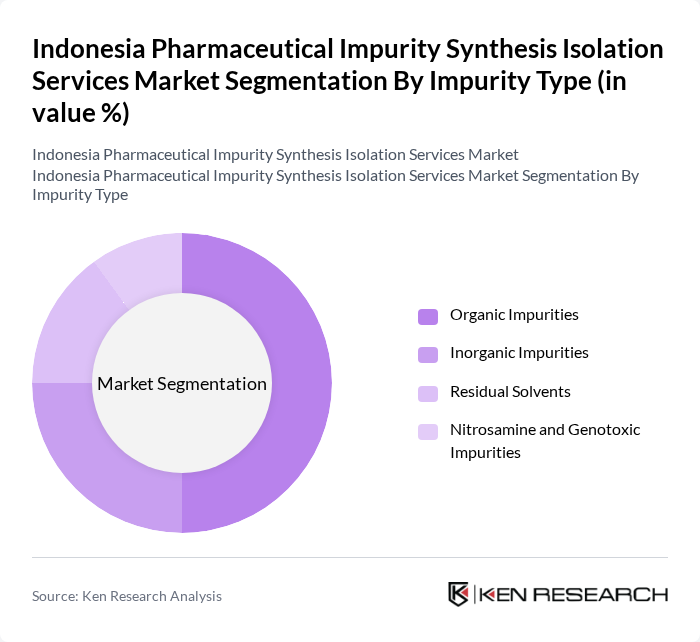
By Impurity Type:The market is segmented based on impurity types, including Organic Impurities, Inorganic Impurities, Residual Solvents, and Nitrosamine and Genotoxic Impurities. Organic Impurities dominate the market due to their prevalence in pharmaceutical formulations and the increasing regulatory scrutiny surrounding them, which aligns with global impurity services trends where organic impurities account for a substantial share of service demand. The focus on reducing these impurities is driving demand for specialized services that can effectively identify and mitigate them, particularly as Indonesian manufacturers expand synthetic and biotech API production and must comply with ICH?aligned impurity guidelines.
The Indonesia Pharmaceutical Impurity Synthesis Isolation Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kimia Farma Tbk, PT Indofarma Tbk, PT Bio Farma (Persero), PT Kalbe Farma Tbk, PT Ferron Par Pharmaceuticals, PT Phapros Tbk, PT Dexa Medica, PT Sanbe Farma, PT Pfizer Indonesia, PT Novartis Indonesia, PT Bayer Indonesia, PT Sanofi Indonesia, PT Takeda Indonesia, PT GlaxoSmithKline Indonesia, Selected Regional CROs/CDMOs Serving Indonesia contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Indonesian pharmaceutical impurity synthesis isolation services market appears promising, driven by increasing investments in research and development, projected to reach IDR 20 trillion (approximately USD 1.4 billion) in future. Additionally, the trend towards outsourcing synthesis services is expected to grow, as companies seek to reduce operational costs while ensuring compliance with stringent regulations. This shift will likely create a more competitive landscape, fostering innovation and collaboration within the industry.
| Segment | Sub-Segments |
|---|---|
| By Service | Synthesis Services Isolation Services Analytical / Impurity Profiling Services Other Specialized Support Services |
| By Impurity Type | Organic Impurities Inorganic Impurities Residual Solvents Nitrosamine and Genotoxic Impurities |
| By Technique / Technology | Chromatography (HPLC, UPLC, GC, Flash, Prep) Spectroscopy (UV-Vis, IR, NMR) Mass Spectrometry & Hyphenated Techniques (LC?MS, GC?MS, LC?MS/MS) Crystallization and Other Isolation Techniques |
| By Application | Drug Development & Clinical Studies Commercial Manufacturing Support Quality Control & Batch Release Testing Regulatory Filing & Compliance Support |
| By End User | Biotechnology and Pharmaceutical Companies Contract Research Organizations (CROs) Contract Development and Manufacturing Organizations (CDMOs/CMOs) Academic & Research Institutes / Others |
| By Molecule Type | Small Molecule Drugs Biologics and Biosimilars Advanced Therapies (e.g., Gene and Cell Therapies) Others |
| By Region | Java Sumatra Kalimantan, Sulawesi, Papua & Other Islands Greater Jakarta and Key Industrial Clusters |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 110 | R&D Managers, Quality Assurance Heads |
| Contract Research Organizations (CROs) | 85 | Project Managers, Laboratory Directors |
| Regulatory Bodies | 45 | Regulatory Affairs Specialists, Compliance Officers |
| Academic Institutions | 55 | Research Professors, Graduate Students in Pharmaceutical Sciences |
| Quality Control Laboratories | 65 | Laboratory Technicians, Quality Control Managers |
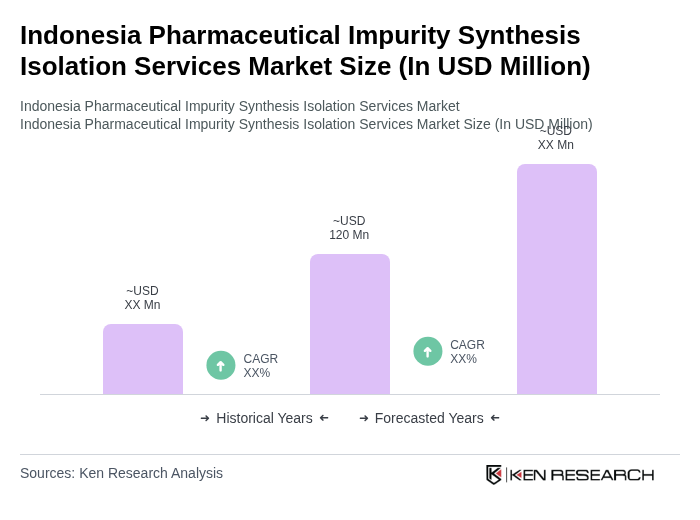
The Indonesia Pharmaceutical Impurity Synthesis Isolation Services Market is valued at approximately USD 120 million, reflecting a significant growth trajectory influenced by increasing demand for high-quality pharmaceuticals and stringent regulatory requirements.