Region:Asia
Author(s):Rebecca
Product Code:KRAC1932
Pages:86
Published On:October 2025
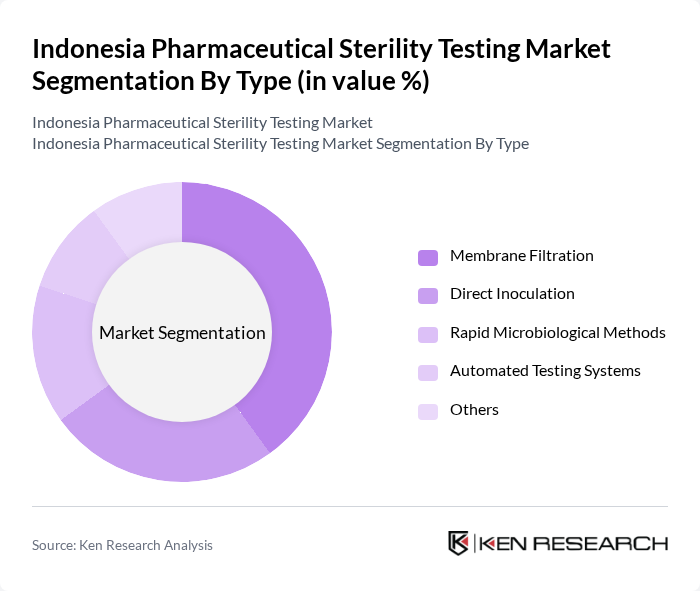
By Type:The market is segmented into various types of sterility testing methods, including Membrane Filtration, Direct Inoculation, Rapid Microbiological Methods, Automated Testing Systems, and Others. Among these, Membrane Filtration remains the most widely adopted method due to its proven effectiveness in detecting microbial contamination in sterile products. The increasing focus on quality assurance, regulatory compliance, and the integration of automated and rapid testing platforms has further accelerated the adoption of Membrane Filtration across pharmaceutical manufacturing facilities .
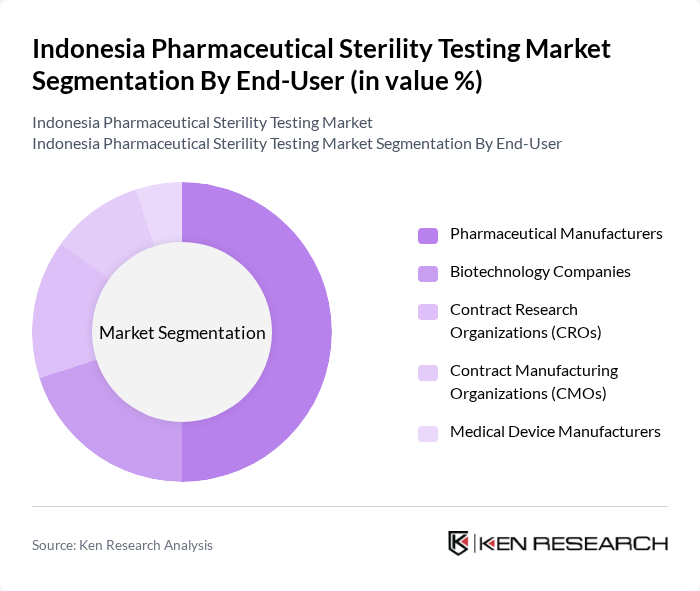
By End-User:The end-user segmentation includes Pharmaceutical Manufacturers, Biotechnology Companies, Contract Research Organizations (CROs), Contract Manufacturing Organizations (CMOs), Medical Device Manufacturers, and Others. Pharmaceutical Manufacturers continue to dominate this segment, driven by the need for stringent quality control measures and adherence to regulatory standards. The increasing production of sterile injectable drugs and the expansion of biopharmaceutical manufacturing have further solidified their position as the leading end-user in the market .
The Indonesia Pharmaceutical Sterility Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Kimia Farma Tbk, PT. Indofarma Tbk, PT. Bio Farma (Persero), PT. Kalbe Farma Tbk, PT. Sanbe Farma, PT. Merck Sharp & Dohme Pharma Tbk, PT. Pfizer Indonesia, PT. Novartis Indonesia, PT. GlaxoSmithKline Indonesia, PT. Johnson & Johnson Indonesia, PT. AstraZeneca Indonesia, PT. Sandoz Indonesia, PT. Roche Indonesia, PT. Takeda Indonesia, PT. Bayer Indonesia, PT. Prodia Widyahusada Tbk, PT. Dexa Medica, PT. Soho Global Health Tbk, PT. Enseval Putera Megatrading Tbk, PT. Tempo Scan Pacific Tbk contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Indonesian pharmaceutical sterility testing market is poised for significant transformation, driven by technological advancements and regulatory evolution. As the demand for biopharmaceuticals continues to rise, companies are increasingly investing in rapid testing methods and automation to enhance efficiency. Furthermore, collaborations between pharmaceutical firms and testing laboratories are expected to foster innovation, ensuring compliance with stringent regulations while improving product safety. This dynamic environment will likely create a robust framework for sustainable growth in the sector.
| Segment | Sub-Segments |
|---|---|
| By Type | Membrane Filtration Direct Inoculation Rapid Microbiological Methods Automated Testing Systems Others |
| By End-User | Pharmaceutical Manufacturers Biotechnology Companies Contract Research Organizations (CROs) Contract Manufacturing Organizations (CMOs) Medical Device Manufacturers Others |
| By Application | Injectable Drugs Biologics & Biosimilars Vaccines Ophthalmic Solutions Medical Devices Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Java Sumatra Bali and Nusa Tenggara Kalimantan Sulawesi Maluku and Papua Others |
| By Testing Method | Traditional Methods Rapid Methods Hybrid Methods Others |
| By Regulatory Compliance | BPOM Compliance ISO Compliance FDA Compliance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Quality Control Managers, Production Supervisors |
| Testing Laboratories | 60 | Laboratory Directors, Microbiologists |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Research Institutions | 50 | Pharmaceutical Researchers, Academic Professors |
| Industry Associations | 40 | Policy Makers, Industry Analysts |
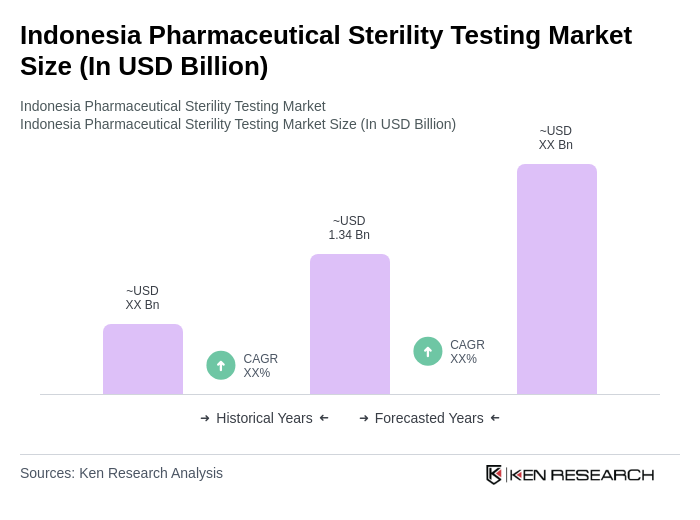
The Indonesia Pharmaceutical Sterility Testing Market is valued at approximately USD 1.34 billion, reflecting significant growth driven by the demand for sterile pharmaceutical products and stringent regulatory requirements.