Region:Middle East
Author(s):Shubham
Product Code:KRAC3533
Pages:94
Published On:October 2025
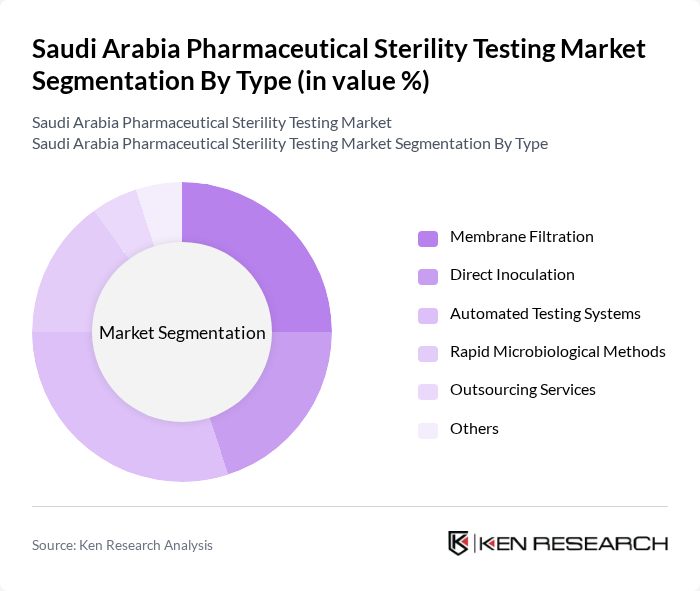
By Type:The market is segmented into various types of sterility testing methods, includingMembrane Filtration, Direct Inoculation, Automated Testing Systems, Rapid Microbiological Methods, Outsourcing Services,andOthers. Each of these methods has unique advantages and applications in ensuring the sterility of pharmaceutical products. Automated and rapid microbiological methods are gaining traction due to their efficiency and compliance with evolving regulatory standards.
By End-User:The end-user segmentation includesPharmaceutical Manufacturers, Biotechnology Companies, Contract Research Organizations (CROs), Contract Manufacturing Organizations (CMOs), Hospitals & Clinical Laboratories,andOthers. Pharmaceutical manufacturers and biotechnology companies are the primary consumers, driven by regulatory compliance and the need to ensure product safety. The trend toward outsourcing sterility testing is also increasing among CROs and CMOs seeking specialized expertise and cost-effective solutions.

The Saudi Arabia Pharmaceutical Sterility Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as SGS S.A., Charles River Laboratories, Merck KGaA, bioMérieux S.A., Thermo Fisher Scientific Inc., WuXi AppTec, Eurofins Scientific, Nelson Labs, Intertek Group plc, BSI Group, Labcorp Drug Development, Pace Analytical Services, LLC, Azzur Group, RSSL (Reading Scientific Services Ltd), Q2 Solutions, Saudi Pharmaceutical Industries & Medical Appliances Corporation (SPIMACO), Tabuk Pharmaceuticals Manufacturing Company, Jamjoom Pharma, Riyadh Pharma, and Sudair Pharma Company contribute to innovation, geographic expansion, and service delivery in this space.
The future of the pharmaceutical sterility testing market in Saudi Arabia appears promising, driven by ongoing advancements in technology and increasing regulatory scrutiny. As the demand for biopharmaceuticals continues to rise, companies are likely to invest in innovative testing solutions that enhance efficiency and accuracy. Furthermore, collaborations with international firms may facilitate knowledge transfer and improve local capabilities, positioning the market for sustainable growth in the coming years while addressing existing challenges effectively.
| Segment | Sub-Segments |
|---|---|
| By Type | Membrane Filtration Direct Inoculation Automated Testing Systems Rapid Microbiological Methods Outsourcing Services Others |
| By End-User | Pharmaceutical Manufacturers Biotechnology Companies Contract Research Organizations (CROs) Contract Manufacturing Organizations (CMOs) Hospitals & Clinical Laboratories Others |
| By Application | Injectable Drugs Biologics & Biosimilars Medical Devices Ophthalmic Solutions Tissue-Based Therapies Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Product Formulation | Liquid Formulations Solid Formulations Semi-Solid Formulations Others |
| By Testing Method | Traditional Methods Rapid Methods Automated Methods Outsourced Testing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing Facilities | 45 | Quality Assurance Managers, Production Supervisors |
| Testing Laboratories | 38 | Laboratory Technicians, Compliance Officers |
| Regulatory Bodies | 22 | Regulatory Affairs Specialists, Policy Makers |
| Healthcare Providers | 32 | Pharmacists, Hospital Administrators |
| Industry Experts and Consultants | 28 | Pharmaceutical Consultants, Industry Analysts |
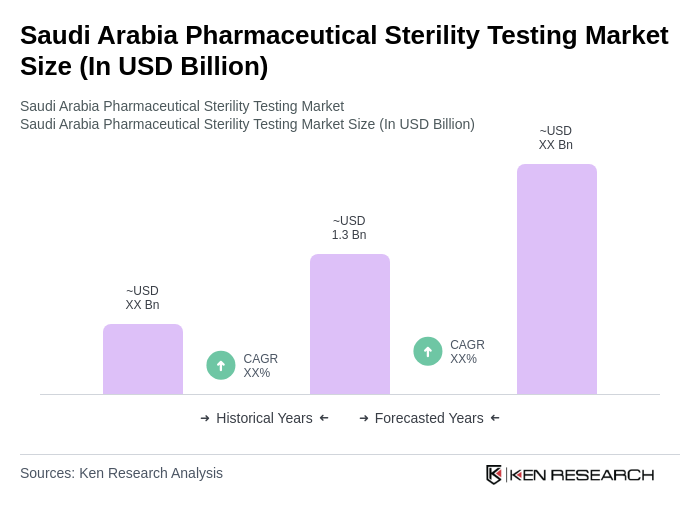
The Saudi Arabia Pharmaceutical Sterility Testing Market is valued at approximately USD 1.3 billion, driven by the increasing demand for high-quality pharmaceuticals and stringent regulatory requirements for sterility testing in the pharmaceutical and biotechnology sectors.