Region:Asia
Author(s):Rebecca
Product Code:KRAC2630
Pages:83
Published On:October 2025
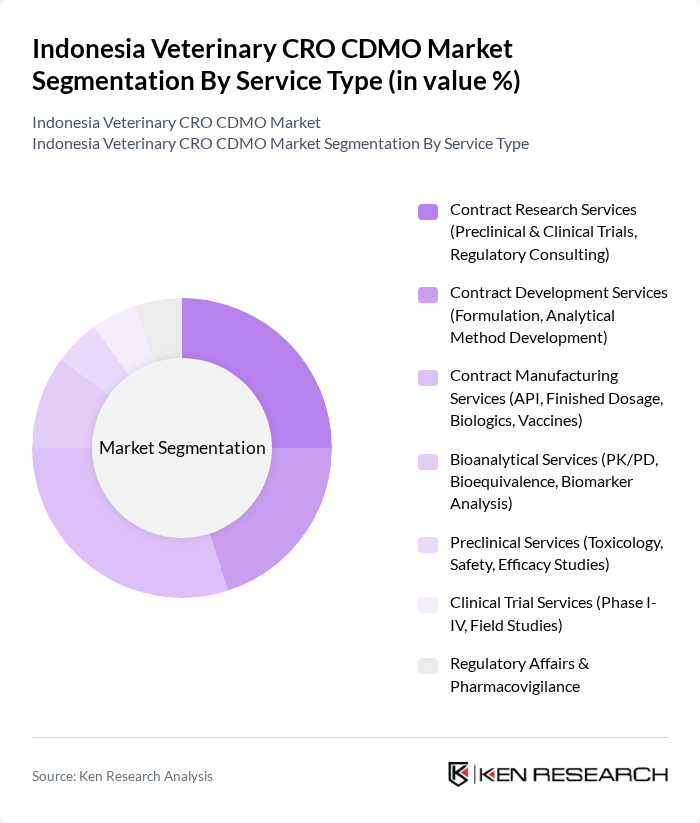
By Service Type:The service type segmentation includes categories such as Contract Research Services, Contract Development Services, Contract Manufacturing Services, Bioanalytical Services, Preclinical Services, Clinical Trial Services, and Regulatory Affairs & Pharmacovigilance. These subsegments are critical to the market, supporting veterinary pharmaceutical companies and research institutions in drug development, safety testing, regulatory compliance, and manufacturing. The market is witnessing a shift toward integrated service offerings, with growing demand for bioanalytical and regulatory consulting services as regulatory requirements become more stringent.
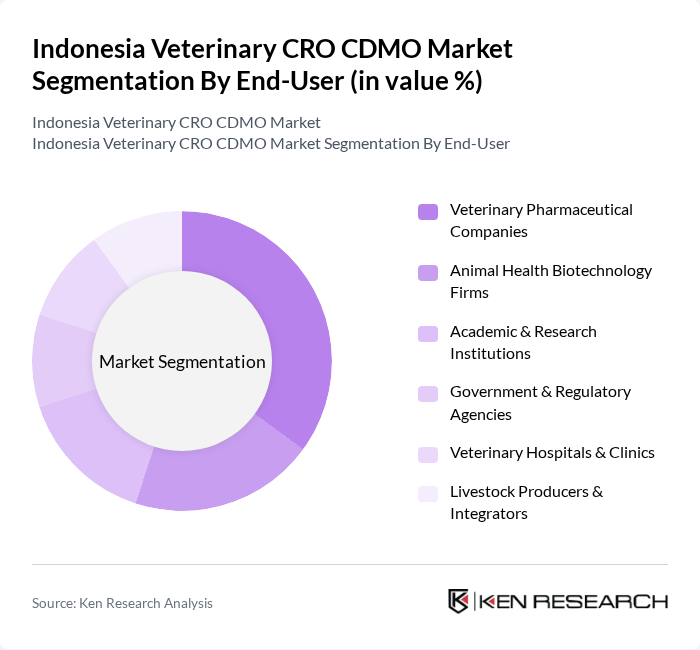
By End-User:The end-user segmentation encompasses Veterinary Pharmaceutical Companies, Animal Health Biotechnology Firms, Academic & Research Institutions, Government & Regulatory Agencies, Veterinary Hospitals & Clinics, and Livestock Producers & Integrators. Each segment has distinct requirements, with pharmaceutical companies and biotechnology firms driving innovation, academic institutions focusing on research, and government agencies ensuring regulatory oversight. The market is increasingly shaped by collaborative R&D initiatives and public-private partnerships to address emerging animal health challenges.
The Indonesia Veterinary CRO CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Bio Farma, PT. Vaksindo Satwa Nusantara, PT. Medion Farma Jaya, PT. Sanbe Farma, PT. Kimia Farma Tbk, PT. Kalbe Farma Tbk, PT. Phapros Tbk, PT. Sido Muncul Tbk, PT. Darya-Varia Laboratoria Tbk, PT. Novell Pharmaceutical Laboratories, PT. Anugerah Pharmindo Lestari, PT. Soho Global Health, PT. Caprifarmindo Laboratories, Argenta, Vaxxinova Indonesia contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Indonesian veterinary CRO CDMO market appears promising, driven by increasing investments in animal health and a growing focus on preventive care. As the pet ownership trend continues to rise, the demand for innovative veterinary products will likely expand. Furthermore, the integration of technology in veterinary practices, such as telemedicine and digital health records, is expected to enhance service delivery, improving overall animal health outcomes and market growth.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Contract Research Services (Preclinical & Clinical Trials, Regulatory Consulting) Contract Development Services (Formulation, Analytical Method Development) Contract Manufacturing Services (API, Finished Dosage, Biologics, Vaccines) Bioanalytical Services (PK/PD, Bioequivalence, Biomarker Analysis) Preclinical Services (Toxicology, Safety, Efficacy Studies) Clinical Trial Services (Phase I-IV, Field Studies) Regulatory Affairs & Pharmacovigilance |
| By End-User | Veterinary Pharmaceutical Companies Animal Health Biotechnology Firms Academic & Research Institutions Government & Regulatory Agencies Veterinary Hospitals & Clinics Livestock Producers & Integrators |
| By Animal Type | Companion Animals (Dogs, Cats, Others) Livestock (Cattle, Poultry, Swine, Others) Aquaculture Others (Equine, Exotic, Wildlife) |
| By Application | Pharmaceuticals Vaccines Feed Additives Diagnostics Others |
| By Region | Java Sumatra Kalimantan Sulawesi Bali Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Pricing Model | Fixed Pricing Variable Pricing Subscription-Based Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Veterinary CRO Services | 60 | Research Directors, Project Managers |
| CDMO for Animal Health Products | 50 | Operations Managers, Quality Assurance Heads |
| Veterinary Pharmaceutical Development | 40 | Regulatory Affairs Specialists, R&D Managers |
| Animal Clinical Trials | 45 | Clinical Research Coordinators, Veterinarians |
| Market Access and Regulatory Compliance | 55 | Market Access Managers, Compliance Officers |
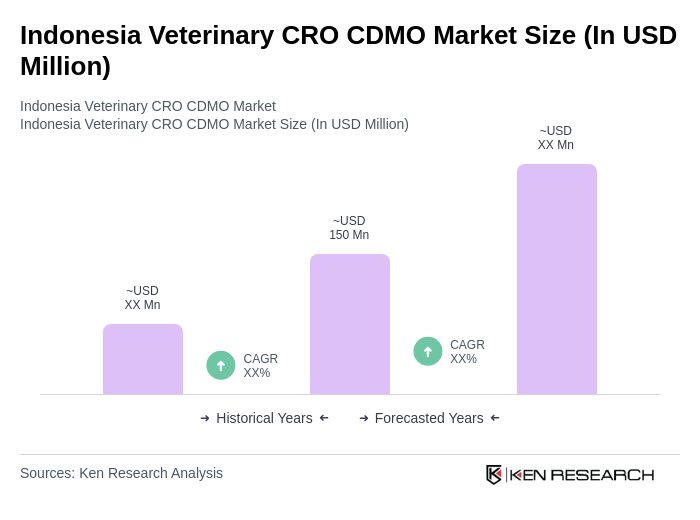
The Indonesia Veterinary CRO CDMO Market is valued at approximately USD 150 million, reflecting its share of the global veterinary CRO and CDMO market, which exceeds USD 7 billion. This market is experiencing growth due to rising demand for animal health products and services.