About the Report
Base Year 2024Japan Neuroendocrine Carcinoma Market Overview
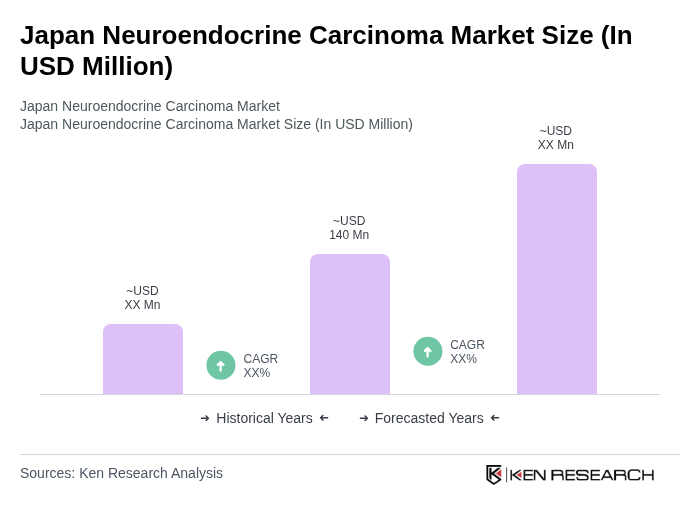
- The Japan Neuroendocrine Carcinoma Market is valued at USD 140 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of neuroendocrine tumors, advancements in diagnostic technologies such as PET and MRI imaging, and the rising prevalence of these cancers in the aging population. The market is also supported by the development of targeted therapies including everolimus and sunitinib, along with improved treatment protocols like peptide receptor radionuclide therapy, which enhance patient outcomes and drive demand for innovative treatment options.
- Key regions dominating the market include Tokyo, Osaka, and Nagoya. These cities are home to advanced healthcare infrastructure, leading research institutions, and a high concentration of specialized medical professionals. The presence of major pharmaceutical companies and clinical trial activities in these urban centers further contributes to their dominance in the neuroendocrine carcinoma market.
- The Act on the Safety of Regenerative Medicine, 2014 issued by the Ministry of Health, Labour and Welfare requires healthcare providers to implement standardized diagnostic protocols for rare cancers including neuroendocrine tumors, mandating licensing for facilities using advanced imaging and targeted therapies with compliance thresholds based on annual case volumes exceeding 50 patients.
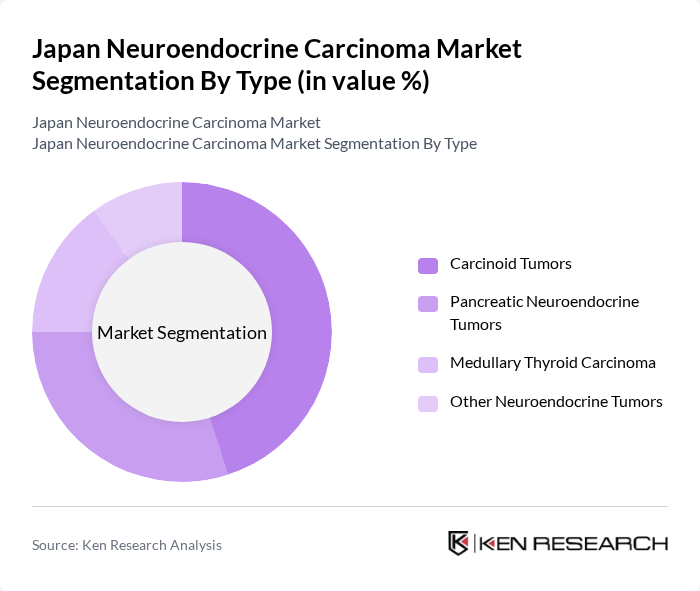
Japan Neuroendocrine Carcinoma Market Segmentation
By Type:The market is segmented into four main types: Carcinoid Tumors, Pancreatic Neuroendocrine Tumors, Medullary Thyroid Carcinoma, and Other Neuroendocrine Tumors. Among these, Carcinoid Tumors are the most prevalent, largely due to their relatively higher incidence rates and better awareness among healthcare professionals. Pancreatic Neuroendocrine Tumors are also gaining attention due to their unique treatment requirements and the increasing number of patients diagnosed with pancreatic cancer.
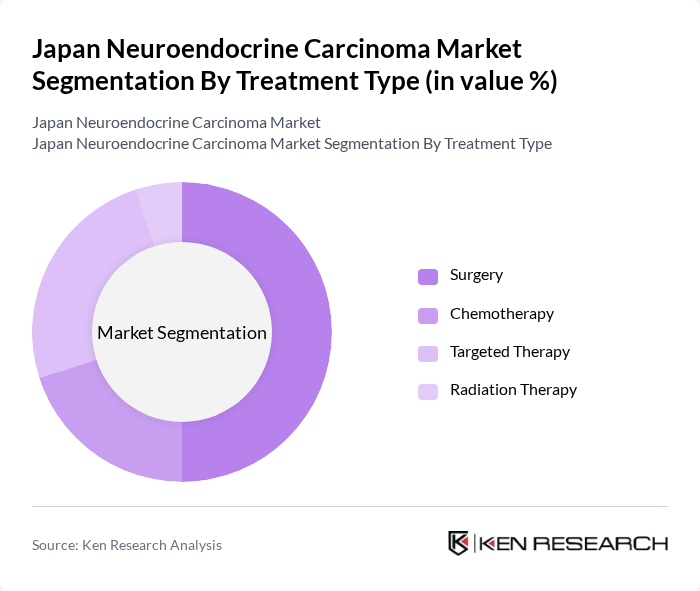
By Treatment Type:The treatment options for neuroendocrine carcinoma include Surgery, Chemotherapy, Targeted Therapy, and Radiation Therapy. Surgery remains the leading treatment modality, particularly for localized tumors, as it offers the best chance for a cure. Targeted Therapy is also gaining traction due to its effectiveness in managing advanced cases, while Chemotherapy and Radiation Therapy are used in specific scenarios based on tumor type and stage.
Japan Neuroendocrine Carcinoma Market Competitive Landscape
The Japan Neuroendocrine Carcinoma Market is characterized by a dynamic mix of regional and international players. Leading participants such as Chugai Pharmaceutical Co., Ltd., Takeda Pharmaceutical Company Limited, Astellas Pharma Inc., Daiichi Sankyo Company, Limited, Otsuka Pharmaceutical Co., Ltd., Eisai Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Kyowa Kirin Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Sanofi K.K., Merck & Co., Inc., Pfizer Japan Inc., Novartis Pharma K.K., Bristol-Myers Squibb K.K., Amgen K.K. contribute to innovation, geographic expansion, and service delivery in this space.
Japan Neuroendocrine Carcinoma Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Neuroendocrine Tumors:The incidence of neuroendocrine tumors (NETs) in Japan has been rising, with approximately 6,000 new cases reported annually. This increase is attributed to better diagnostic capabilities and heightened awareness among healthcare professionals. The Japanese population's aging demographic, with over 29% aged 65 and older, further contributes to this trend, as older individuals are more susceptible to developing NETs, thereby driving demand for specialized treatments and healthcare services.
- Advancements in Diagnostic Technologies:The Japanese healthcare sector has seen significant advancements in diagnostic technologies, including the use of PET scans and advanced imaging techniques. In future, it is estimated that over 1.6 million diagnostic imaging procedures will be performed specifically for NETs. These innovations enhance early detection rates, allowing for timely intervention and improved patient outcomes, which in turn stimulates market growth as more patients seek advanced diagnostic services.
- Enhanced Treatment Options and Therapies:The introduction of novel therapies, such as targeted treatments and immunotherapies, has revolutionized the management of neuroendocrine carcinoma in Japan. In future, the market for NET therapies is projected to exceed ¥55 billion, driven by the availability of new drugs and treatment protocols. This expansion in treatment options not only improves survival rates but also encourages more patients to seek treatment, thereby propelling market growth.
Market Challenges
- High Cost of Treatment:The financial burden associated with neuroendocrine carcinoma treatment remains a significant challenge in Japan. The average cost of treatment can reach up to ¥11 million per patient annually, which poses accessibility issues for many individuals. This high cost can deter patients from seeking necessary care, leading to late-stage diagnoses and poorer outcomes, ultimately hindering market growth and patient access to innovative therapies.
- Limited Availability of Specialized Healthcare Facilities:Japan faces a shortage of specialized healthcare facilities equipped to handle neuroendocrine carcinoma cases. Currently, only about 210 hospitals in Japan are recognized as centers of excellence for treating NETs. This limited access can result in delays in diagnosis and treatment, negatively impacting patient outcomes and creating barriers to market expansion as patients may seek care abroad or avoid treatment altogether.
Japan Neuroendocrine Carcinoma Market Future Outlook
The future of the neuroendocrine carcinoma market in Japan appears promising, driven by ongoing advancements in personalized medicine and the integration of artificial intelligence in diagnostics. As healthcare providers increasingly adopt patient-centric care models, the focus will shift towards tailored treatment plans that enhance patient engagement and outcomes. Additionally, the expansion of telemedicine services is expected to improve access to specialists, facilitating timely interventions and fostering a more robust healthcare ecosystem for neuroendocrine carcinoma patients.
Market Opportunities
- Growth in Personalized Medicine:The rise of personalized medicine presents a significant opportunity for the neuroendocrine carcinoma market. With advancements in genetic profiling, tailored therapies can be developed, improving treatment efficacy. In future, the personalized medicine market in Japan is projected to reach ¥35 billion, indicating a strong potential for innovative treatment approaches that cater to individual patient needs.
- Expansion of Telemedicine Services:The increasing adoption of telemedicine in Japan offers a unique opportunity to enhance patient access to specialized care for neuroendocrine carcinoma. In future, telemedicine consultations are expected to grow by 45%, allowing patients in remote areas to connect with specialists. This expansion can lead to earlier diagnoses and improved management of the disease, ultimately benefiting the overall market landscape.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Carcinoid Tumors Pancreatic Neuroendocrine Tumors Medullary Thyroid Carcinoma Other Neuroendocrine Tumors |
| By Treatment Type | Surgery Chemotherapy Targeted Therapy Radiation Therapy |
| By Stage of Disease | Localized Regional Distant Metastasis Others |
| By Age Group | Pediatric Adult Geriatric |
| By Gender | Male Female |
| By Healthcare Setting | Hospitals Specialty Clinics Home Healthcare |
| By Region | Kanto Kansai Chubu Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Labour and Welfare)
Pharmaceutical Manufacturers
Biotechnology Companies
Medical Device Manufacturers
Healthcare Providers and Hospitals
Clinical Research Organizations
Health Insurance Companies
Players Mentioned in the Report:
Chugai Pharmaceutical Co., Ltd.
Takeda Pharmaceutical Company Limited
Astellas Pharma Inc.
Daiichi Sankyo Company, Limited
Otsuka Pharmaceutical Co., Ltd.
Eisai Co., Ltd.
Sumitomo Dainippon Pharma Co., Ltd.
Kyowa Kirin Co., Ltd.
Mitsubishi Tanabe Pharma Corporation
Sanofi K.K.
Merck & Co., Inc.
Pfizer Japan Inc.
Novartis Pharma K.K.
Bristol-Myers Squibb K.K.
Amgen K.K.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan Neuroendocrine Carcinoma Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan Neuroendocrine Carcinoma Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan Neuroendocrine Carcinoma Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of neuroendocrine tumors
3.1.2 Advancements in diagnostic technologies
3.1.3 Rising awareness and education about neuroendocrine carcinoma
3.1.4 Enhanced treatment options and therapies
3.2 Market Challenges
3.2.1 High cost of treatment
3.2.2 Limited availability of specialized healthcare facilities
3.2.3 Lack of awareness among general practitioners
3.2.4 Regulatory hurdles in drug approval
3.3 Market Opportunities
3.3.1 Growth in personalized medicine
3.3.2 Expansion of telemedicine services
3.3.3 Collaborations with research institutions
3.3.4 Development of novel therapeutics
3.4 Market Trends
3.4.1 Increasing focus on targeted therapies
3.4.2 Rise in clinical trials for neuroendocrine carcinoma
3.4.3 Integration of AI in diagnostics
3.4.4 Shift towards patient-centric care models
3.5 Government Regulation
3.5.1 Approval processes for new treatments
3.5.2 Reimbursement policies for neuroendocrine carcinoma therapies
3.5.3 Guidelines for clinical practice
3.5.4 Regulations on clinical trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan Neuroendocrine Carcinoma Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan Neuroendocrine Carcinoma Market Segmentation
8.1 By Type
8.1.1 Carcinoid Tumors
8.1.2 Pancreatic Neuroendocrine Tumors
8.1.3 Medullary Thyroid Carcinoma
8.1.4 Other Neuroendocrine Tumors
8.2 By Treatment Type
8.2.1 Surgery
8.2.2 Chemotherapy
8.2.3 Targeted Therapy
8.2.4 Radiation Therapy
8.3 By Stage of Disease
8.3.1 Localized
8.3.2 Regional
8.3.3 Distant Metastasis
8.3.4 Others
8.4 By Age Group
8.4.1 Pediatric
8.4.2 Adult
8.4.3 Geriatric
8.5 By Gender
8.5.1 Male
8.5.2 Female
8.6 By Healthcare Setting
8.6.1 Hospitals
8.6.2 Specialty Clinics
8.6.3 Home Healthcare
8.7 By Region
8.7.1 Kanto
8.7.2 Kansai
8.7.3 Chubu
8.7.4 Others
9. Japan Neuroendocrine Carcinoma Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Development Cycle Time
9.2.8 Sales Conversion Rate
9.2.9 Distribution Efficiency
9.2.10 Brand Equity Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Chugai Pharmaceutical Co., Ltd.
9.5.2 Takeda Pharmaceutical Company Limited
9.5.3 Astellas Pharma Inc.
9.5.4 Daiichi Sankyo Company, Limited
9.5.5 Otsuka Pharmaceutical Co., Ltd.
9.5.6 Eisai Co., Ltd.
9.5.7 Sumitomo Dainippon Pharma Co., Ltd.
9.5.8 Kyowa Kirin Co., Ltd.
9.5.9 Mitsubishi Tanabe Pharma Corporation
9.5.10 Sanofi K.K.
9.5.11 Merck & Co., Inc.
9.5.12 Pfizer Japan Inc.
9.5.13 Novartis Pharma K.K.
9.5.14 Bristol-Myers Squibb K.K.
9.5.15 Amgen K.K.
10. Japan Neuroendocrine Carcinoma Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Cancer Treatment
10.1.2 Policy Framework for Neuroendocrine Carcinoma
10.1.3 Collaboration with Healthcare Providers
10.1.4 Procurement Processes and Challenges
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Cancer Research Facilities
10.2.2 Funding for Clinical Trials
10.2.3 Partnerships with Pharmaceutical Companies
10.2.4 Allocation for Patient Support Programs
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Treatment
10.3.2 Affordability of Care
10.3.3 Availability of Specialized Services
10.3.4 Quality of Patient Care
10.4 User Readiness for Adoption
10.4.1 Awareness of Treatment Options
10.4.2 Acceptance of New Therapies
10.4.3 Training for Healthcare Professionals
10.4.4 Patient Education Initiatives
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of Treatment Outcomes
10.5.2 Long-term Patient Monitoring
10.5.3 Expansion of Treatment Protocols
10.5.4 Cost-Benefit Analysis of New Therapies
11. Japan Neuroendocrine Carcinoma Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports and white papers on neuroendocrine carcinoma treatment trends in Japan
- Review of government health statistics and cancer registries for incidence and prevalence data
- Examination of clinical trial databases for ongoing research and emerging therapies in neuroendocrine carcinoma
Primary Research
- Interviews with oncologists specializing in neuroendocrine tumors to gather insights on treatment protocols and patient demographics
- Surveys with healthcare providers and hospital administrators regarding treatment availability and patient access
- Focus groups with patients diagnosed with neuroendocrine carcinoma to understand their treatment experiences and preferences
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including clinical guidelines and expert opinions
- Triangulation of market data with patient registries and treatment outcome studies
- Sanity checks conducted through expert panel reviews comprising oncologists and healthcare economists
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national cancer treatment expenditure and neuroendocrine carcinoma prevalence
- Segmentation of the market by treatment type, including surgery, chemotherapy, and targeted therapies
- Incorporation of demographic factors such as age and gender distribution in cancer incidence
Bottom-up Modeling
- Collection of treatment cost data from hospitals and clinics offering neuroendocrine carcinoma therapies
- Estimation of patient volume based on historical treatment data and projected incidence rates
- Calculation of market size using a volume x price approach for each treatment modality
Forecasting & Scenario Analysis
- Development of forecasting models based on historical growth rates and emerging treatment options
- Scenario analysis considering factors such as advancements in medical technology and changes in healthcare policy
- Projections for market growth through 2030 under various regulatory and economic conditions
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists Specializing in Neuroendocrine Tumors | 100 | Medical Oncologists, Surgical Oncologists |
| Healthcare Administrators in Oncology Departments | 75 | Hospital Administrators, Oncology Program Directors |
| Patients Diagnosed with Neuroendocrine Carcinoma | 60 | Patients, Caregivers |
| Pharmaceutical Representatives for Cancer Therapies | 50 | Sales Representatives, Product Managers |
| Clinical Researchers in Oncology | 40 | Clinical Trial Coordinators, Research Scientists |
Frequently Asked Questions
What is the current value of the Japan Neuroendocrine Carcinoma Market?
The Japan Neuroendocrine Carcinoma Market is valued at approximately USD 140 million, reflecting a five-year historical analysis. This growth is driven by increased awareness, advancements in diagnostic technologies, and the rising prevalence of neuroendocrine tumors among the aging population.