Region:Middle East
Author(s):Dev
Product Code:KRAC8059
Pages:87
Published On:November 2025
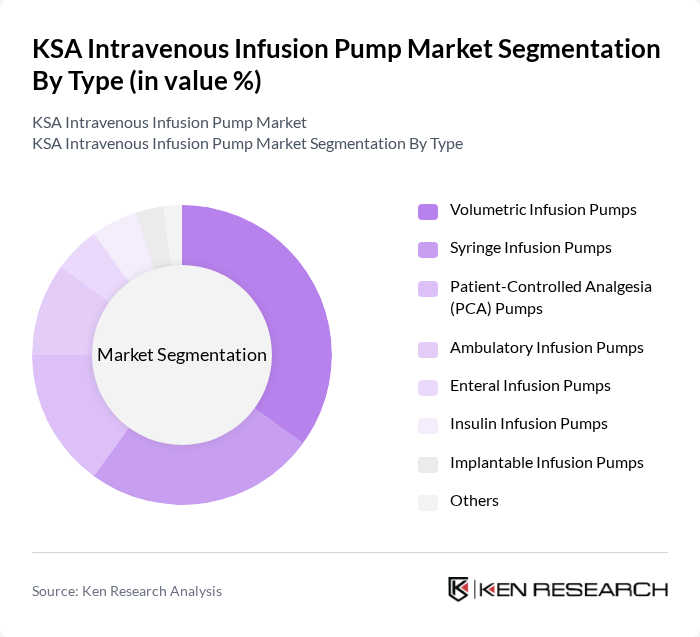
By Type:The market is segmented into volumetric infusion pumps, syringe infusion pumps, patient-controlled analgesia (PCA) pumps, ambulatory infusion pumps, enteral infusion pumps, insulin infusion pumps, implantable infusion pumps, and others. Volumetric infusion pumps are the most widely used due to their versatility, precision in fluid and medication delivery, and essential role in critical care, oncology, and surgical settings. Syringe infusion pumps are also prominent, particularly in pediatric and neonatal care, while ambulatory and PCA pumps are gaining traction for home-based and pain management therapies.
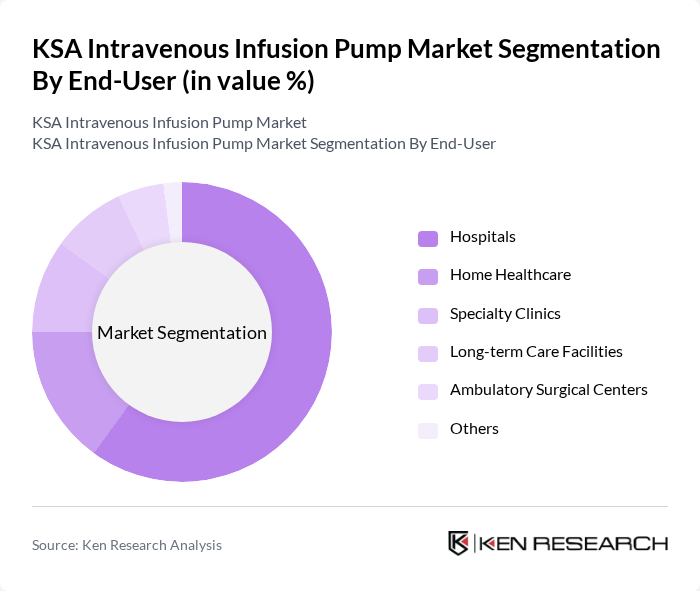
By End-User:The end-user segmentation includes hospitals, home healthcare, specialty clinics, long-term care facilities, ambulatory surgical centers, and others. Hospitals are the leading end-users of intravenous infusion pumps, driven by the high volume of patients requiring intravenous therapy, advanced medical equipment needs, and increasing adoption of smart infusion technologies. Home healthcare is a rapidly growing segment, supported by patient preference for home-based care, government initiatives, and the expansion of outpatient infusion therapy centers. Specialty clinics and long-term care facilities also contribute to market growth, particularly for chronic disease management and palliative care.
The KSA Intravenous Infusion Pump Market is characterized by a dynamic mix of regional and international players. Leading participants such as Baxter International Inc., B. Braun Melsungen AG, Fresenius Kabi AG, Medtronic plc, Smiths Medical (ICU Medical, Inc.), Terumo Corporation, ICU Medical, Inc., Becton, Dickinson and Company (BD), Nipro Corporation, Mindray Medical International Limited, Moog Inc., Shenzhen Mindray Bio-Medical Electronics Co., Ltd., JMS Co., Ltd., Abbott Laboratories, Hospira (Pfizer Inc.) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the KSA intravenous infusion pump market appears promising, driven by ongoing advancements in technology and a growing emphasis on patient-centric care. As healthcare facilities increasingly adopt automated systems, the integration of IoT and mobile health applications will enhance patient monitoring and safety. Furthermore, the expansion of healthcare services in rural areas is expected to facilitate greater access to infusion therapy, ultimately improving health outcomes across the Kingdom.
| Segment | Sub-Segments |
|---|---|
| By Type | Volumetric Infusion Pumps Syringe Infusion Pumps Patient-Controlled Analgesia (PCA) Pumps Ambulatory Infusion Pumps Enteral Infusion Pumps Insulin Infusion Pumps Implantable Infusion Pumps Others |
| By End-User | Hospitals Home Healthcare Specialty Clinics Long-term Care Facilities Ambulatory Surgical Centers Others |
| By Application | Oncology (Chemotherapy) Pain Management Anesthesia Critical Care Parenteral Nutrition Diabetes Management Others |
| By Technology | Smart Infusion Technology Conventional Infusion Technology Wireless Infusion Technology Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Policy Support | Government Subsidies Tax Incentives Regulatory Support Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Hospital Procurement | 60 | Procurement Managers, Hospital Administrators |
| Private Healthcare Facilities | 50 | Clinical Directors, Biomedical Engineers |
| Infusion Pump Usage in ICUs | 40 | Intensive Care Unit Nurses, Medical Directors |
| Home Healthcare Providers | 40 | Home Care Coordinators, Patient Care Managers |
| Medical Device Distributors | 40 | Sales Managers, Product Specialists |
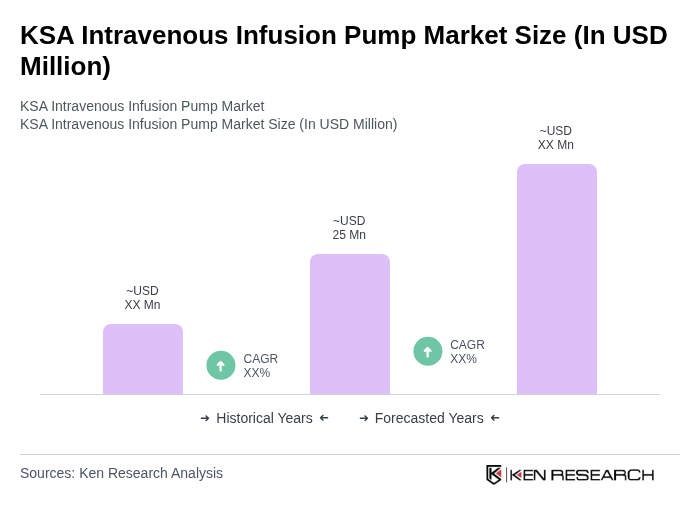
The KSA Intravenous Infusion Pump Market is valued at approximately USD 25 million, reflecting a five-year historical analysis. This growth is driven by the rising prevalence of chronic diseases and advancements in infusion pump technology.