Region:Middle East
Author(s):Geetanshi
Product Code:KRAC4402
Pages:86
Published On:October 2025
By Molecule Type:
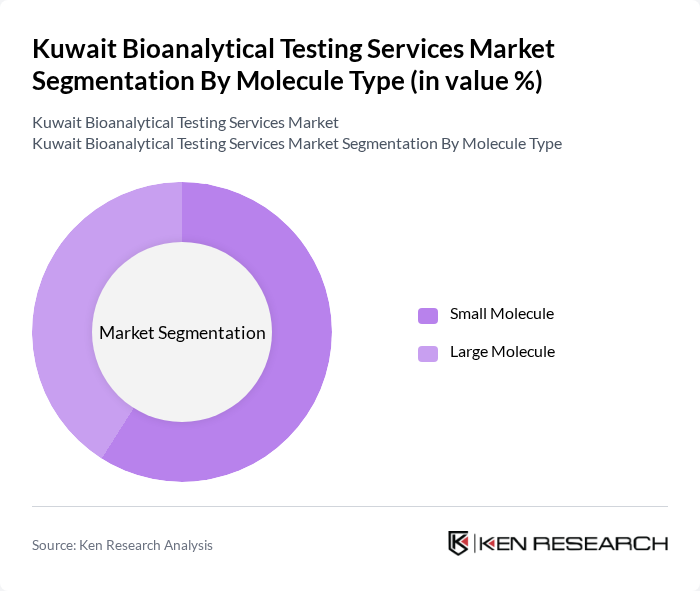
The bioanalytical testing services market in Kuwait is segmented by molecule type into small molecules and large molecules. Small molecules hold the largest market share, primarily due to their established role in drug development and the pharmaceutical industry. These compounds are easier to analyze and have broad applications across therapeutic areas. Large molecules, including biologics and biosimilars, are gaining importance as the focus on personalized medicine and biologics increases. The large molecule segment is registering the fastest growth, driven by rising demand for advanced therapies and ongoing clinical trials.
By Test Type:
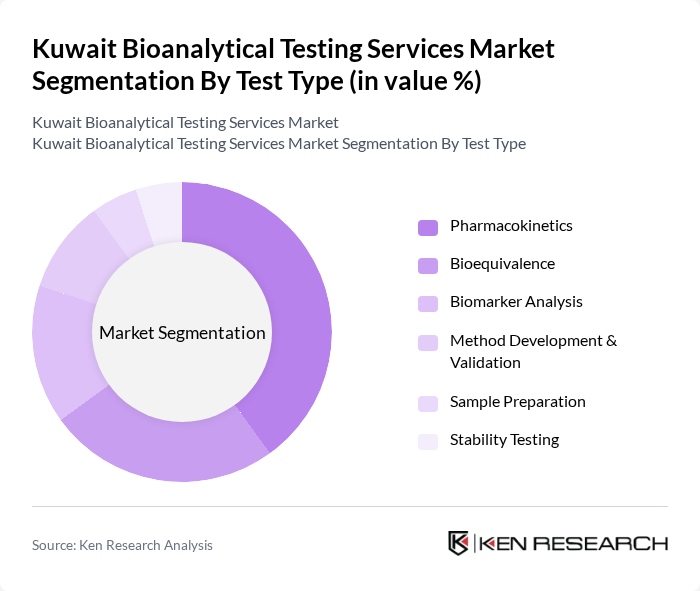
The market is also segmented by test type, which includes pharmacokinetics, bioequivalence, biomarker analysis, method development & validation, sample preparation, and stability testing. Pharmacokinetics is the leading sub-segment, driven by the need to understand drug absorption, distribution, metabolism, and excretion. Bioequivalence testing is significant for generic drug development, ensuring new formulations are comparable to existing products. Biomarker analysis is increasingly important for personalized medicine, while method development and validation are essential for accurate and reliable testing. The expansion of clinical trials and the adoption of advanced analytical techniques are further driving demand across all test types.
The Kuwait Bioanalytical Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Charles River Laboratories International Inc., Labcorp Holdings Inc., ICON plc, IQVIA Holdings Inc., Syneos Health, Eurofins Scientific SE, SGS SA, Intertek Group plc, Pace Analytical Services LLC, Wuxi AppTec, PPD (Part of Thermo Fisher Scientific), Medpace Holdings Inc., Q2 Solutions (IQVIA-Quest Joint Venture), BioAgilytix Labs contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Kuwait bioanalytical testing services market appears promising, driven by increasing healthcare investments and a focus on innovative testing solutions. As the government continues to enhance healthcare infrastructure, the demand for bioanalytical services is expected to rise. Additionally, the integration of artificial intelligence in testing processes is likely to improve efficiency and accuracy, positioning Kuwait as a competitive player in the regional market. Overall, the sector is poised for significant growth, supported by technological advancements and rising healthcare needs.
| Segment | Sub-Segments |
|---|---|
| By Molecule Type | Small Molecule Large Molecule |
| By Test Type | Pharmacokinetics Bioequivalence Biomarker Analysis Method Development & Validation Sample Preparation Stability Testing |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Academic & Research Institutions Government Agencies |
| By Application | Drug Development Clinical Trials Generic Drug Development Biosimilar Development |
| By Sample Type | Blood Samples Urine Samples Tissue Samples Plasma/Serum Samples Others |
| By Service Provider Type | In-house Testing Labs Outsourced CRO Services Hybrid Models |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Services | 65 | Laboratory Managers, Quality Assurance Officers |
| Clinical Research Organizations | 55 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Environmental Testing Services | 45 | Environmental Scientists, Compliance Officers |
| Biotechnology Applications | 50 | Research Scientists, Product Development Managers |
| Academic Research Institutions | 40 | University Professors, Research Coordinators |
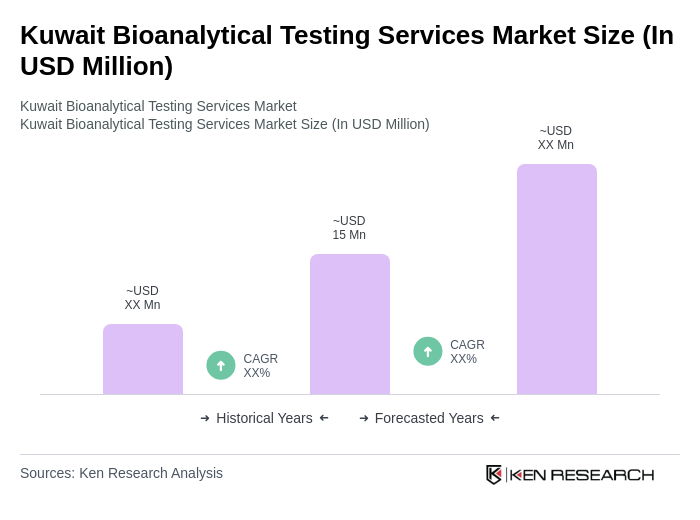
The Kuwait Bioanalytical Testing Services Market is valued at approximately USD 15 million, reflecting a five-year historical analysis. This growth is driven by increased demand for drug development, clinical trials, and the rising prevalence of chronic diseases.