Region:Middle East
Author(s):Rebecca
Product Code:KRAD7460
Pages:99
Published On:December 2025
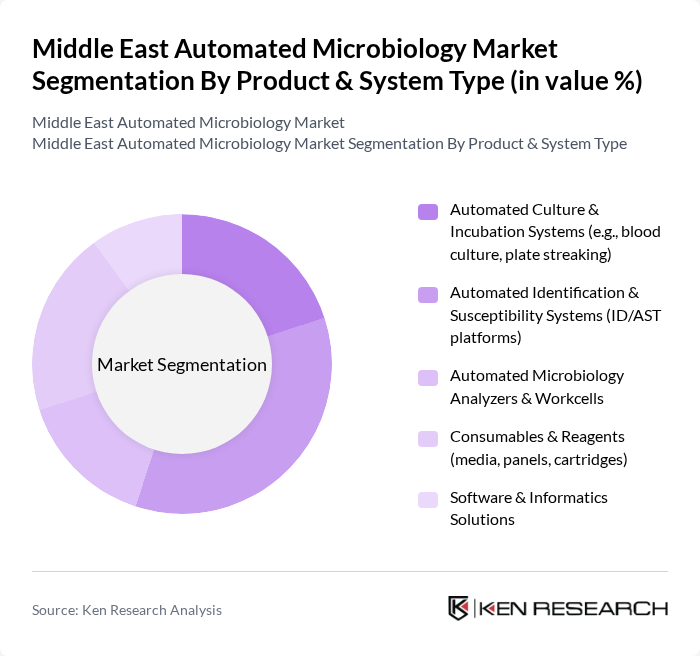
By Product & System Type:The product and system type segmentation includes various subsegments that cater to different aspects of automated microbiology. The key subsegments are Automated Culture & Incubation Systems, Automated Identification & Susceptibility Systems, Automated Microbiology Analyzers & Workcells, Consumables & Reagents, and Software & Informatics Solutions. Among these, Automated Identification & Susceptibility Systems are leading the market due to their critical role in diagnosing infections and determining appropriate treatments, particularly in the context of rising antimicrobial resistance, where rapid and accurate ID/AST supports effective stewardship programs in clinical settings.
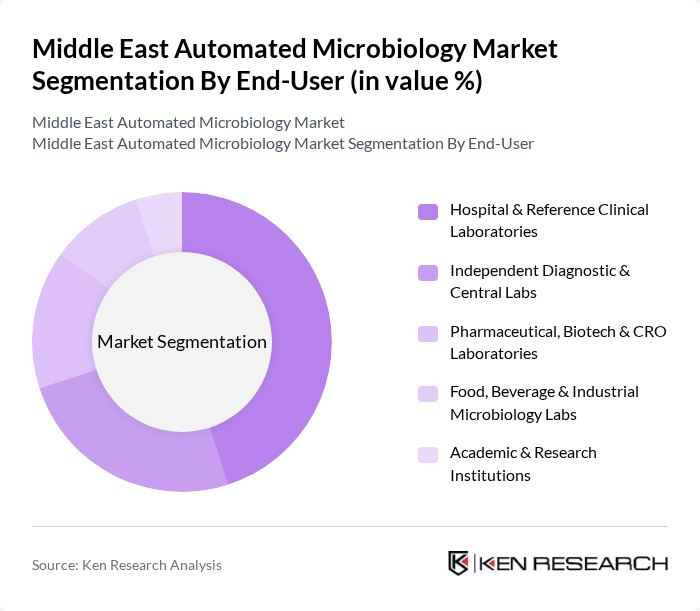
By End-User:The end-user segmentation encompasses various sectors utilizing automated microbiology solutions, including Hospital & Reference Clinical Laboratories, Independent Diagnostic & Central Labs, Pharmaceutical, Biotech & CRO Laboratories, Food, Beverage & Industrial Microbiology Labs, and Academic & Research Institutions. Hospital & Reference Clinical Laboratories dominate this segment due to their high volume of diagnostic testing, continuous pressure to reduce turnaround times, and the need for standardized, traceable workflows that support infection control and antimicrobial stewardship programs in patient care.
The Middle East Automated Microbiology Market is characterized by a dynamic mix of regional and international players. Leading participants such as bioMérieux SA, Becton, Dickinson and Company (BD), Thermo Fisher Scientific Inc., F. Hoffmann-La Roche Ltd (Roche Diagnostics), Abbott Laboratories (Abbott Diagnostics), Siemens Healthineers AG, Beckman Coulter, Inc. (a Danaher company), QIAGEN N.V., Bio-Rad Laboratories, Inc., Cepheid (a Danaher company), Hologic, Inc., Bruker Corporation, Copan Diagnostics Inc., Accelerate Diagnostics, Inc., Saudi Diagnostic Holding Company and Key Regional Distributors contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East automated microbiology market appears promising, driven by technological advancements and increasing healthcare investments. As healthcare facilities expand and modernize, the integration of artificial intelligence and machine learning into diagnostic processes will enhance accuracy and efficiency. Additionally, the growing emphasis on personalized medicine will further propel the demand for tailored diagnostic solutions, ensuring that healthcare providers can meet the evolving needs of patients in the region effectively.
| Segment | Sub-Segments |
|---|---|
| By Product & System Type | Automated Culture & Incubation Systems (e.g., blood culture, plate streaking) Automated Identification & Susceptibility Systems (ID/AST platforms) Automated Microbiology Analyzers & Workcells Consumables & Reagents (media, panels, cartridges) Software & Informatics Solutions |
| By End-User | Hospital & Reference Clinical Laboratories Independent Diagnostic & Central Labs Pharmaceutical, Biotech & CRO Laboratories Food, Beverage & Industrial Microbiology Labs Academic & Research Institutions |
| By Application | Clinical Infectious Disease Diagnostics Antimicrobial Susceptibility Testing (AST) & Stewardship Programs Pharmaceutical & Bioprocess Sterility / Environmental Monitoring Food & Water Safety Testing Hospital Infection Control & Surveillance |
| By Technology | Automated Culture-Based & Colony Counting Technologies Molecular Diagnostics (PCR, real-time PCR, syndromic panels) Mass Spectrometry (e.g., MALDI-TOF MS) Immunoassays & Rapid Tests AI-Enabled Image Analysis & Digital Microbiology |
| By Country / Sub-Region | Saudi Arabia United Arab Emirates Qatar Kuwait, Oman and Bahrain Rest of Gulf Cooperation Council (GCC) Levant (e.g., Jordan, Lebanon) Egypt Rest of Middle East & North Africa |
| By Distribution / Sales Channel | Direct Sales by OEMs Regional Distributors & Dealers Group Purchasing Organizations (GPOs) & Tender-Based Procurement Online & E-Procurement Platforms |
| By Commercial Model | Capital Equipment Purchase (Capex) Reagent Rental / Placement Contracts Managed Laboratory Services & Long-Term Service Agreements Pay-Per-Test / Pay-Per-Use Models |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 100 | Laboratory Managers, Chief Microbiologists |
| Diagnostic Centers | 80 | Procurement Managers, Operations Directors |
| Research Institutions | 60 | Research Scientists, Lab Technicians |
| Healthcare Technology Providers | 70 | Product Managers, Sales Executives |
| Regulatory Bodies | 50 | Policy Makers, Compliance Officers |
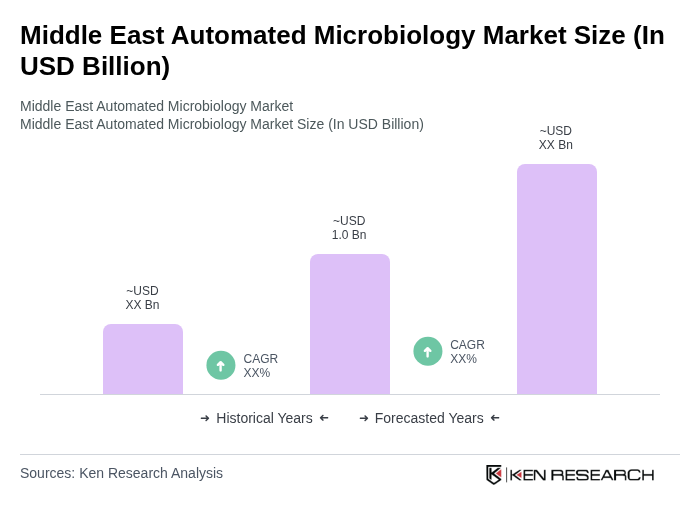
The Middle East Automated Microbiology Market is valued at approximately USD 1.0 billion, representing a significant share of the global automated microbiology market, which is estimated at around USD 8.8 billion.