About the Report
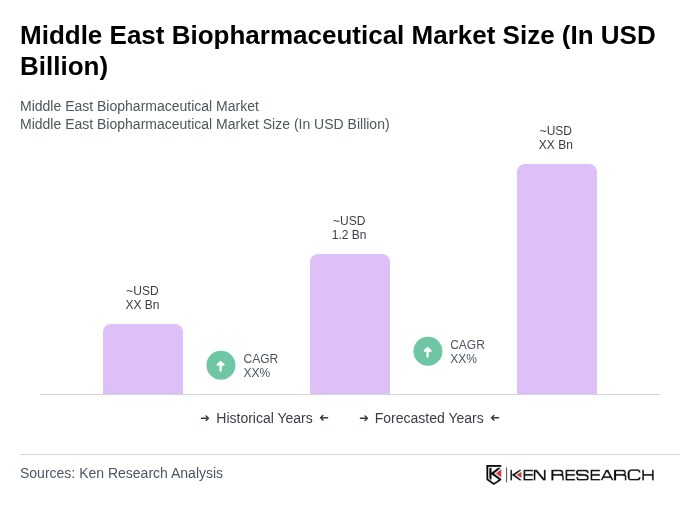
Base Year 2024Middle East Biopharmaceutical Market Overview
- The Middle East Biopharmaceutical Market is valued at USD 1.2 billion, based on a five-year historical analysis of the biologics and advanced biopharmaceuticals segment. This growth is primarily driven by increasing healthcare expenditure, a rising prevalence of chronic diseases such as cancer and diabetes, and robust advancements in biopharmaceutical research and development. The market is further supported by a growing demand for innovative therapies and biologics, especially monoclonal antibodies and vaccines, which are becoming essential for treating complex health conditions. Recent trends include a strong focus on biosimilars and local manufacturing initiatives to reduce dependency on imports and enhance regional supply security .
- Key players in this market include countries such as Saudi Arabia, the United Arab Emirates, and Israel. These regions dominate due to their robust healthcare infrastructure, significant investments in biotechnology, and supportive government policies aimed at fostering innovation and research. The presence of leading pharmaceutical companies and research institutions further enhances their market position. Saudi Arabia, for instance, has allocated substantial government budgets to health and social development, while the UAE is recognized for its rapid adoption of biosimilars and advanced regulatory frameworks .
- The “Ministerial Decree No. 28 of 2023 Concerning the Registration and Marketing Authorization of Biopharmaceutical Products,” issued by the UAE Ministry of Health and Prevention, mandates that all biopharmaceutical products must undergo comprehensive clinical trials and regulatory review prior to market approval. This regulation establishes clear requirements for clinical evidence, pharmacovigilance, and post-marketing surveillance, thereby enhancing public trust in biopharmaceuticals and promoting a more robust healthcare system in the region .
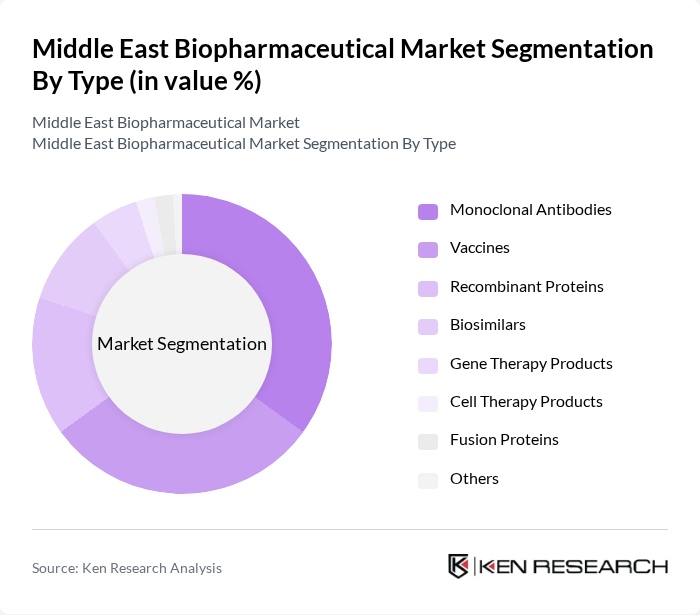
Middle East Biopharmaceutical Market Segmentation
By Type:The biopharmaceutical market can be segmented into various types, including Monoclonal Antibodies, Vaccines, Recombinant Proteins, Biosimilars, Gene Therapy Products, Cell Therapy Products, Fusion Proteins, and Others. Among these, Monoclonal Antibodies and Vaccines are particularly significant due to their widespread application in treating various diseases and their central role in public health initiatives. Biosimilars are also gaining traction as affordability and local production become regional priorities .
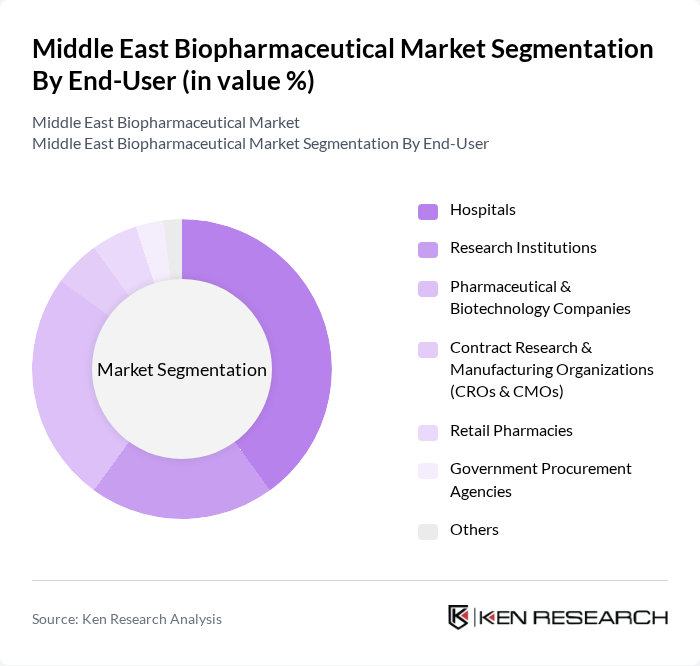
By End-User:The end-user segmentation includes Hospitals, Research Institutions, Pharmaceutical & Biotechnology Companies, Contract Research & Manufacturing Organizations (CROs & CMOs), Retail Pharmacies, Government Procurement Agencies, and Others. Hospitals and Pharmaceutical & Biotechnology Companies are the leading end-users, driven by the increasing demand for advanced treatment options and the need for innovative drug development. Research institutions are also significant contributors, especially in clinical trials and translational research .
Middle East Biopharmaceutical Market Competitive Landscape
The Middle East Biopharmaceutical Market is characterized by a dynamic mix of regional and international players. Leading participants such as Julphar (Gulf Pharmaceutical Industries), SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Tabuk Pharmaceuticals, Hikma Pharmaceuticals, Pharco Pharmaceuticals, Eva Pharma, Amman Pharmaceutical Industries, Bioventure Healthcare (UAE), Pfizer, Roche, Novartis, Sanofi, Merck & Co., Amgen, GSK, AbbVie, AstraZeneca, Takeda Pharmaceutical Company, Biogen, Eli Lilly and Company, Bayer, Sandoz, Regeneron Pharmaceuticals, Celltrion, Biocon contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Biopharmaceutical Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The Middle East is witnessing a significant rise in chronic diseases, with the World Health Organization reporting that non-communicable diseases account for 70% of all deaths in the region. In future, the number of diabetes cases is projected to reach 60 million, while cardiovascular diseases are expected to affect over 25 million individuals. This growing health burden is driving demand for innovative biopharmaceutical solutions, creating a robust market environment for companies focused on chronic disease management.
- Rising Healthcare Expenditure:Healthcare spending in the Middle East is anticipated to reach $220 billion in future, reflecting a compound annual growth rate of 7.5% from previous years. This increase is fueled by government initiatives aimed at improving healthcare infrastructure and access to advanced treatments. As countries like Saudi Arabia and the UAE invest heavily in healthcare, the biopharmaceutical sector stands to benefit significantly from enhanced funding and resources dedicated to research and development.
- Advancements in Biopharmaceutical Technologies:The Middle East is experiencing rapid advancements in biopharmaceutical technologies, with investments in biotechnology research exceeding $1.8 billion in future. Innovations in drug development, including monoclonal antibodies and gene therapies, are becoming more prevalent. This technological evolution is supported by collaborations between local universities and international firms, fostering an environment conducive to breakthroughs that can address unmet medical needs in the region.
Market Challenges
- High Cost of R&D:The biopharmaceutical industry in the Middle East faces significant challenges due to the high costs associated with research and development, which can exceed $3 billion per new drug. This financial burden is compounded by the lengthy timelines required for clinical trials, often taking over a decade. As a result, many local firms struggle to compete with established global players who have more substantial financial resources to invest in R&D.
- Stringent Regulatory Requirements:Regulatory frameworks in the Middle East can be complex and stringent, often leading to delays in drug approvals. For instance, the average time for drug registration can take up to 20 months, significantly impacting market entry for new biopharmaceuticals. These regulatory hurdles can deter investment and innovation, as companies may be reluctant to navigate the intricate approval processes without guaranteed returns on investment.
Middle East Biopharmaceutical Market Future Outlook
The future of the Middle East biopharmaceutical market appears promising, driven by a combination of technological advancements and increasing healthcare investments. As governments prioritize healthcare reforms and local production, the market is likely to see a surge in innovative therapies. Additionally, the integration of digital health solutions and artificial intelligence in drug development will enhance efficiency and effectiveness, paving the way for personalized medicine approaches that cater to the unique needs of the regional population.
Market Opportunities
- Expansion of Biosimilars Market:The biosimilars market in the Middle East is projected to grow significantly, with an estimated value of $1.5 billion in future. This growth is driven by the increasing demand for cost-effective alternatives to expensive biologics, providing a lucrative opportunity for local manufacturers to enter the market and enhance patient access to essential therapies.
- Collaborations with Global Biopharmaceutical Firms:Strategic partnerships between local companies and global biopharmaceutical firms are on the rise, with over 35 collaborations expected in future. These alliances facilitate knowledge transfer, access to advanced technologies, and shared resources, enabling local firms to enhance their product offerings and compete more effectively in the global market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Vaccines Recombinant Proteins Biosimilars Gene Therapy Products Cell Therapy Products Fusion Proteins Others |
| By End-User | Hospitals Research Institutions Pharmaceutical & Biotechnology Companies Contract Research & Manufacturing Organizations (CROs & CMOs) Retail Pharmacies Government Procurement Agencies Others |
| By Therapeutic Area | Oncology Infectious Diseases & Vaccines Autoimmune Disorders Cardiovascular Diseases Diabetes & Metabolic Disorders Rare Diseases Others |
| By Distribution Channel | Direct Sales Distributors/Wholesalers Online Pharmacies Retail Pharmacies Hospital Pharmacies Others |
| By Region | GCC Countries (Saudi Arabia, UAE, Qatar, Kuwait, Bahrain, Oman) Levant Region (Jordan, Lebanon, Syria, Palestine, Iraq) North Africa (Egypt, Morocco, Algeria, Tunisia, Libya) Others |
| By Product Lifecycle Stage | Pre-commercialization Commercialization Post-commercialization Others |
| By Research and Development Phase | Discovery Phase Preclinical Phase Clinical Trials Marketed Products Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Biopharmaceutical Manufacturers and Producers
Pharmaceutical Distributors and Wholesalers
Healthcare Providers and Hospitals
Biotechnology Research Organizations
Industry Associations (e.g., Middle East Pharmaceutical Industry Association)
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Julphar (Gulf Pharmaceutical Industries)
SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
Tabuk Pharmaceuticals
Hikma Pharmaceuticals
Pharco Pharmaceuticals
Eva Pharma
Amman Pharmaceutical Industries
Bioventure Healthcare (UAE)
Pfizer
Roche
Novartis
Sanofi
Merck & Co.
Amgen
GSK
AbbVie
AstraZeneca
Takeda Pharmaceutical Company
Biogen
Eli Lilly and Company
Bayer
Sandoz
Regeneron Pharmaceuticals
Celltrion
Biocon
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Biopharmaceutical Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Biopharmaceutical Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Biopharmaceutical Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising healthcare expenditure
3.1.3 Advancements in biopharmaceutical technologies
3.1.4 Government initiatives to boost local production
3.2 Market Challenges
3.2.1 High cost of R&D
3.2.2 Stringent regulatory requirements
3.2.3 Limited access to advanced technologies
3.2.4 Competition from generic drugs
3.3 Market Opportunities
3.3.1 Expansion of biosimilars market
3.3.2 Collaborations with global biopharmaceutical firms
3.3.3 Growth in personalized medicine
3.3.4 Increasing investment in biotechnology startups
3.4 Market Trends
3.4.1 Shift towards precision medicine
3.4.2 Rise of digital health solutions
3.4.3 Focus on sustainable biopharmaceutical practices
3.4.4 Integration of AI in drug development
3.5 Government Regulation
3.5.1 Regulatory frameworks for drug approval
3.5.2 Intellectual property protection laws
3.5.3 Pricing regulations for biopharmaceuticals
3.5.4 Policies promoting local manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Biopharmaceutical Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Biopharmaceutical Market Segmentation
8.1 By Type
8.1.1 Monoclonal Antibodies
8.1.2 Vaccines
8.1.3 Recombinant Proteins
8.1.4 Biosimilars
8.1.5 Gene Therapy Products
8.1.6 Cell Therapy Products
8.1.7 Fusion Proteins
8.1.8 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Research Institutions
8.2.3 Pharmaceutical & Biotechnology Companies
8.2.4 Contract Research & Manufacturing Organizations (CROs & CMOs)
8.2.5 Retail Pharmacies
8.2.6 Government Procurement Agencies
8.2.7 Others
8.3 By Therapeutic Area
8.3.1 Oncology
8.3.2 Infectious Diseases & Vaccines
8.3.3 Autoimmune Disorders
8.3.4 Cardiovascular Diseases
8.3.5 Diabetes & Metabolic Disorders
8.3.6 Rare Diseases
8.3.7 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors/Wholesalers
8.4.3 Online Pharmacies
8.4.4 Retail Pharmacies
8.4.5 Hospital Pharmacies
8.4.6 Others
8.5 By Region
8.5.1 GCC Countries (Saudi Arabia, UAE, Qatar, Kuwait, Bahrain, Oman)
8.5.2 Levant Region (Jordan, Lebanon, Syria, Palestine, Iraq)
8.5.3 North Africa (Egypt, Morocco, Algeria, Tunisia, Libya)
8.5.4 Others
8.6 By Product Lifecycle Stage
8.6.1 Pre-commercialization
8.6.2 Commercialization
8.6.3 Post-commercialization
8.6.4 Others
8.7 By Research and Development Phase
8.7.1 Discovery Phase
8.7.2 Preclinical Phase
8.7.3 Clinical Trials
8.7.4 Marketed Products
8.7.5 Others
9. Middle East Biopharmaceutical Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Middle East Biopharma Segment)
9.2.4 Market Penetration Rate (by product/region)
9.2.5 Number of Approved Biopharmaceutical Products
9.2.6 R&D Investment as a Percentage of Revenue
9.2.7 Clinical Pipeline Size (Middle East focus)
9.2.8 Regulatory Approval Success Rate (regional)
9.2.9 Local Manufacturing Capacity (units or value)
9.2.10 Strategic Partnerships/Local Collaborations
9.2.11 Product Launch Frequency (Middle East)
9.2.12 Brand Recognition Index (regional)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Julphar (Gulf Pharmaceutical Industries)
9.5.2 SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
9.5.3 Tabuk Pharmaceuticals
9.5.4 Hikma Pharmaceuticals
9.5.5 Pharco Pharmaceuticals
9.5.6 Eva Pharma
9.5.7 Amman Pharmaceutical Industries
9.5.8 Bioventure Healthcare (UAE)
9.5.9 Pfizer
9.5.10 Roche
9.5.11 Novartis
9.5.12 Sanofi
9.5.13 Merck & Co.
9.5.14 Amgen
9.5.15 GSK
9.5.16 AbbVie
9.5.17 AstraZeneca
9.5.18 Takeda Pharmaceutical Company
9.5.19 Biogen
9.5.20 Eli Lilly and Company
9.5.21 Bayer
9.5.22 Sandoz
9.5.23 Regeneron Pharmaceuticals
9.5.24 Celltrion
9.5.25 Biocon
10. Middle East Biopharmaceutical Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Industry
10.1.3 Ministry of Finance
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.2.3 Research Facility Development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Research Institutions
10.3.3 Pharmaceutical Companies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Biopharmaceuticals
10.4.2 Training and Education Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Development
10.5.3 Long-term Sustainability Considerations
10.5.4 Others
11. Middle East Biopharmaceutical Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Market Entry
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from regional biopharmaceutical associations and health ministries
- Review of published articles in scientific journals focusing on biopharmaceutical innovations in the Middle East
- Examination of trade publications and market analysis reports specific to the biopharmaceutical sector
Primary Research
- Interviews with key opinion leaders in biopharmaceutical companies and research institutions
- Surveys conducted with healthcare professionals and pharmacists regarding biopharmaceutical product usage
- Focus group discussions with patients and healthcare providers to understand market needs and preferences
Validation & Triangulation
- Cross-validation of data through multiple sources including industry reports and expert interviews
- Triangulation of findings from primary research with secondary data to ensure consistency
- Sanity checks through expert panel reviews to validate assumptions and findings
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and biopharmaceutical spending trends
- Segmentation of the market by therapeutic areas and product types
- Incorporation of government healthcare initiatives and funding for biopharmaceutical research
Bottom-up Modeling
- Collection of sales data from leading biopharmaceutical firms operating in the region
- Estimation of market share based on product launch timelines and sales performance
- Volume and pricing analysis to derive revenue estimates for various biopharmaceutical segments
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating demographic trends, disease prevalence, and healthcare access
- Scenario modeling based on potential regulatory changes and market entry of new therapies
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Biopharmaceuticals | 100 | Oncologists, Clinical Researchers |
| Cardiovascular Biopharmaceuticals | 80 | Cardiologists, Pharmacists |
| Autoimmune Disease Treatments | 70 | Rheumatologists, Patient Advocacy Groups |
| Biopharmaceutical Supply Chain Management | 60 | Supply Chain Managers, Logistics Coordinators |
| Regulatory Affairs in Biopharmaceuticals | 90 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the Middle East Biopharmaceutical Market?
The Middle East Biopharmaceutical Market is valued at approximately USD 1.2 billion, driven by increasing healthcare expenditure, a rise in chronic diseases, and advancements in biopharmaceutical research and development, particularly in biologics and advanced therapies.