Region:Middle East
Author(s):Geetanshi
Product Code:KRAD4111
Pages:82
Published On:December 2025
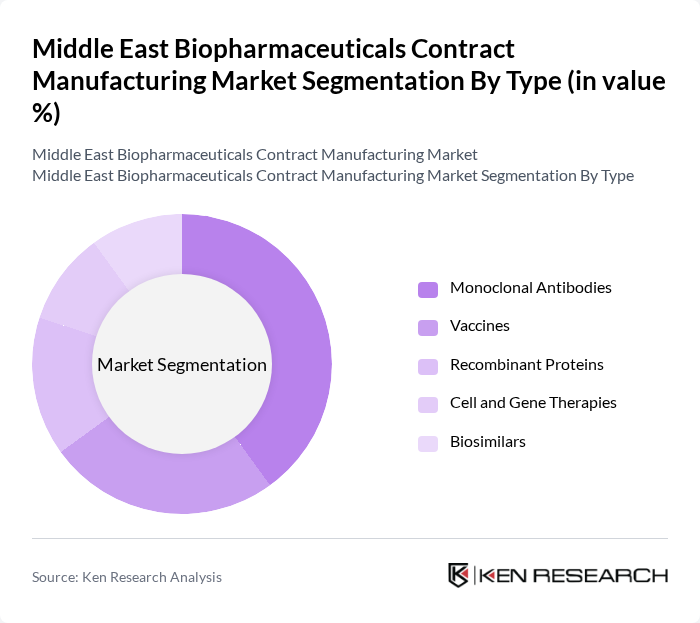
By Type:The biopharmaceuticals contract manufacturing market is segmented into various types, including Monoclonal Antibodies, Vaccines, Recombinant Proteins, Cell and Gene Therapies, and Biosimilars. Among these, Monoclonal Antibodies have emerged as the leading sub-segment due to their extensive application in treating various diseases, including cancer and autoimmune disorders. The increasing demand for targeted therapies and the growing number of clinical trials are driving the growth of this segment. Vaccines also hold significant market share, particularly in light of the global focus on immunization and pandemic preparedness.
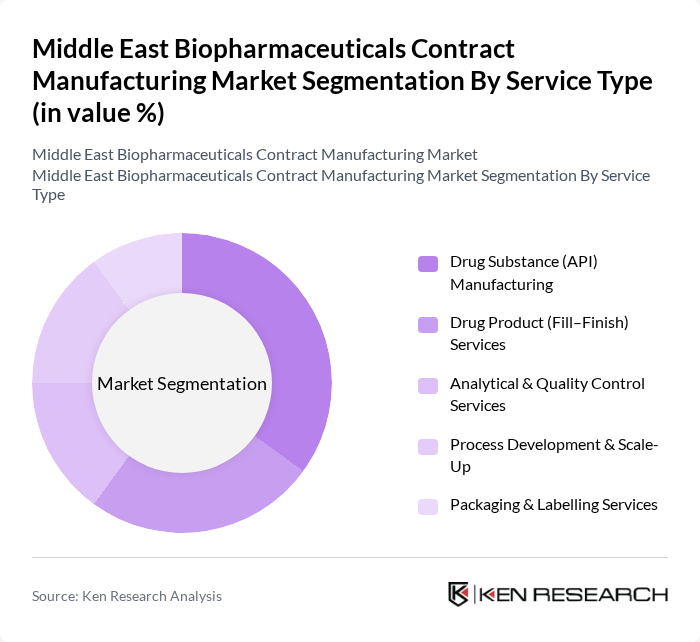
By Service Type:The market is also segmented by service type, which includes Drug Substance (API) Manufacturing, Drug Product (Fill–Finish) Services, Analytical & Quality Control Services, Process Development & Scale-Up, and Packaging & Labelling Services. Drug Substance (API) Manufacturing is the dominant service type, driven by the increasing need for high-quality active pharmaceutical ingredients. The rise in outsourcing by pharmaceutical companies to reduce costs and enhance efficiency further supports the growth of this segment. Additionally, the demand for comprehensive service offerings, including analytical and quality control services, is on the rise.
The Middle East Biopharmaceuticals Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saudi Bioindustries Company (SBC), Lifera (Saudi Arabia), Julphar Gulf Pharmaceutical Industries (UAE), Neopharma (UAE), Biocon Biologics – Regional Operations, Lonza – Middle East Partnerships, Samsung Biologics – Middle East Client Base, WuXi Biologics – MENA Projects, Global Pharma (UAE), Tabuk Pharmaceuticals – Biologics & Biosimilars Manufacturing, Hikma Pharmaceuticals – Injectables & Biologics, SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corp.), Egypt International Pharmaceutical Industries Company (EIPICO) – Biopharma Initiatives, Pharco Corporation (Egypt) – Biologics and Biosimilar Lines, Other Emerging Regional CDMOs and Fill–Finish Players contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East biopharmaceuticals contract manufacturing market appears promising, driven by technological advancements and a focus on personalized medicine. As the region continues to enhance its healthcare infrastructure, the demand for innovative biopharmaceuticals is expected to rise. Additionally, the increasing trend of outsourcing manufacturing processes will likely create new opportunities for contract manufacturers, enabling them to leverage advanced technologies and improve operational efficiencies in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Vaccines Recombinant Proteins Cell and Gene Therapies Biosimilars |
| By Service Type | Drug Substance (API) Manufacturing Drug Product (Fill–Finish) Services Analytical & Quality Control Services Process Development & Scale?Up Packaging & Labelling Services |
| By Expression System | Mammalian Cell Culture Microbial (Bacterial/Fungal) Cell?free and Other Systems |
| By Scale of Operation | Preclinical and Clinical Scale Commercial Scale |
| By End-User | Multinational Biopharma Companies Regional and Local Pharmaceutical Manufacturers Emerging Biotechnology Firms Academic and Research Institutes |
| By Country | Saudi Arabia United Arab Emirates Qatar Kuwait, Bahrain, and Oman Egypt Rest of Middle East |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical CMOs | 120 | Operations Managers, Business Development Executives |
| Regulatory Affairs in Biopharmaceuticals | 90 | Regulatory Affairs Specialists, Compliance Managers |
| Quality Control in Biopharmaceutical Manufacturing | 80 | Quality Assurance Managers, Lab Directors |
| Market Access and Pricing Strategies | 60 | Market Access Managers, Pricing Analysts |
| Research & Development in Biopharmaceuticals | 100 | R&D Directors, Clinical Development Managers |
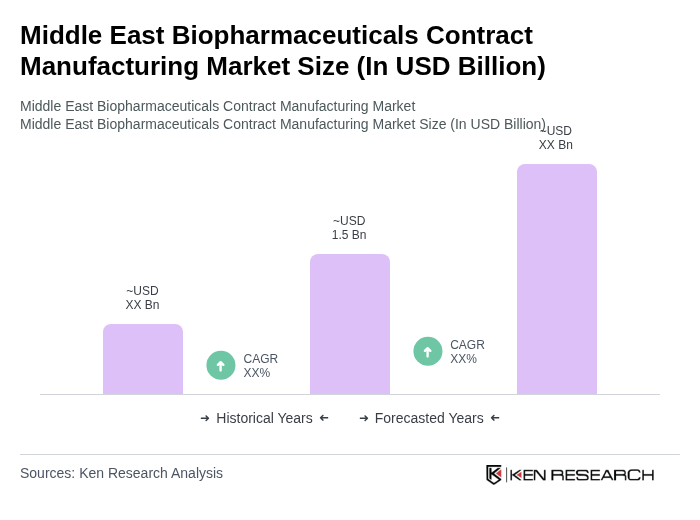
The Middle East Biopharmaceuticals Contract Manufacturing Market is valued at approximately USD 1.5 billion, reflecting a significant growth driven by increasing demand for biopharmaceuticals, advancements in biotechnology, and rising chronic disease prevalence.