Region:Global
Author(s):Geetanshi
Product Code:KRAD4805
Pages:94
Published On:December 2025
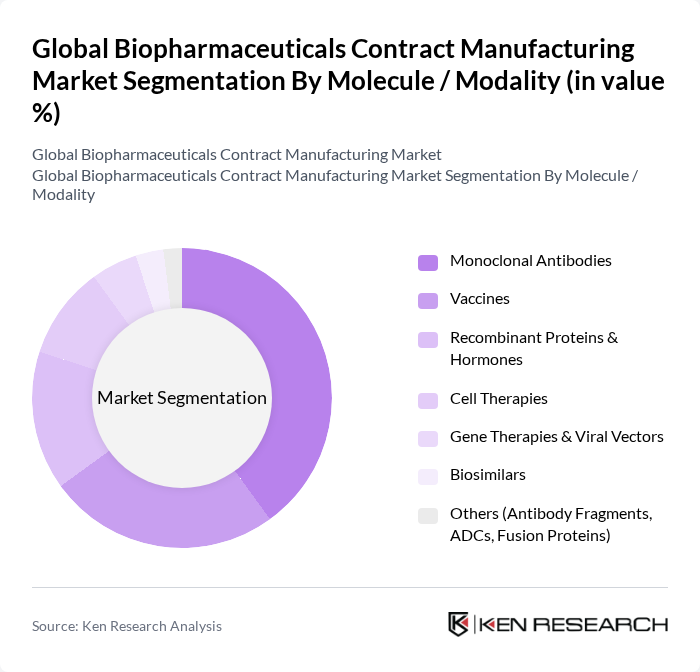
By Molecule / Modality:This segmentation includes various types of biopharmaceuticals that are manufactured through contract services. The dominant sub-segment is Monoclonal Antibodies, which represent the largest share of commercial biologics and late?stage pipelines and are widely used in therapeutic applications due to their specificity and efficacy in treating diseases such as cancer, autoimmune disorders, and inflammatory conditions. Vaccines also hold a significant share, driven by the global focus on routine immunization, pandemic preparedness, and the expansion of recombinant, viral?vector, and mRNA vaccine platforms. The demand for Recombinant Proteins & Hormones is increasing, particularly in the treatment of diabetes, anemia, and hormonal and metabolic disorders, where originator biologics and biosimilars require high?quality large?scale production. Cell Therapies and Gene Therapies & Viral Vectors are emerging segments, reflecting advancements in personalized and regenerative medicine and driving specialized CDMO capabilities in cell processing and vector manufacturing. Biosimilars are gaining traction as cost-effective alternatives to branded biologics, accelerating demand for process development, scale?up, and commercial manufacturing support from CDMOs. The "Others" category includes innovative products like Antibody Fragments, ADCs, and Fusion Proteins, which are increasingly being explored for their therapeutic potential and require complex conjugation and analytics capabilities often sourced from specialized contract manufacturers.
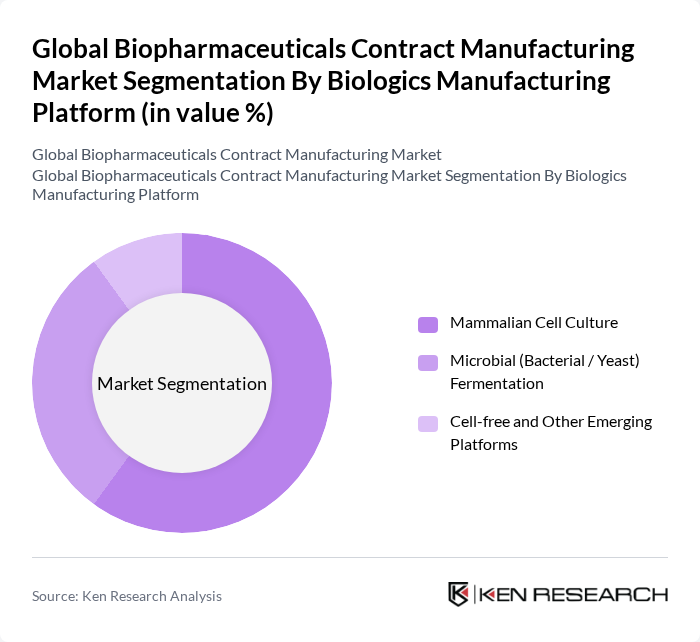
By Biologics Manufacturing Platform:This segmentation focuses on the various platforms used for the production of biopharmaceuticals. The Mammalian Cell Culture platform is the most widely used due to its ability to produce complex glycosylated proteins that are similar to human proteins and its dominance in monoclonal antibody and many recombinant protein manufacturing. Microbial (Bacterial / Yeast) Fermentation is also significant, particularly for the production of simpler recombinant proteins, enzymes, and certain vaccines where high volumetric productivity and cost efficiency are critical. Emerging platforms, such as Cell-free systems and novel expression technologies, are gaining attention for their potential to streamline development, enable rapid synthesis of complex biologics (including difficult?to?express proteins), and reduce cycle times and some scale?up risks. The choice of platform often depends on the specific requirements of the biopharmaceutical being produced, including molecular complexity, yield, scalability, cost of goods, and regulatory and quality considerations related to product comparability and process robustness.
The Global Biopharmaceuticals Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group Ltd., Samsung Biologics Co., Ltd., Catalent, Inc., WuXi Biologics (Cayman) Inc., Boehringer Ingelheim BioXcellence, FUJIFILM Diosynth Biotechnologies, AbbVie Contract Manufacturing, Thermo Fisher Scientific (Patheon Pharmaceuticals), AGC Biologics, Samsung Biopis Manufacturing Partners, Rentschler Biopharma SE, KBI Biopharma, Inc., WuXi AppTec Co., Ltd. (Biologics & ATMP Segments), Sartorius Stedim BioOutsource Ltd., Eurofins CDMO & Biologics Solutions contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biopharmaceuticals contract manufacturing market appears promising, driven by ongoing technological advancements and increasing demand for biologics. As companies continue to invest in automation and AI integration, production efficiency is expected to improve significantly. Additionally, the growing trend towards personalized medicine will likely create new opportunities for contract manufacturers to develop tailored solutions, further enhancing their role in the biopharmaceutical supply chain and fostering innovation in drug development.
| Segment | Sub-Segments |
|---|---|
| By Molecule / Modality | Monoclonal Antibodies Vaccines Recombinant Proteins & Hormones Cell Therapies Gene Therapies & Viral Vectors Biosimilars Others (Antibody Fragments, ADCs, Fusion Proteins) |
| By Biologics Manufacturing Platform | Mammalian Cell Culture Microbial (Bacterial / Yeast) Fermentation Cell-free and Other Emerging Platforms |
| By Service Type | Process Development & Optimization cGMP Clinical & Commercial Manufacturing Fill-Finish & Lyophilization Analytical & Bioanalytical Testing Quality Control & Regulatory Support Others (Tech Transfer, Scale-up, Validation) |
| By Stage of Manufacturing | Preclinical & Early Clinical (Phase I–II) Late-Stage Clinical (Phase III) Commercial-Scale Manufacturing |
| By Scale of Production | Small Scale (Up to 2,000 L) Medium Scale (2,000–10,000 L) Large Scale (>10,000 L) |
| By End-User | Large Pharmaceutical Companies Small & Mid-size Biotech Firms Virtual / Emerging Biopharma Academia & Research Institutes Others (Non-profit, Government Agencies) |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical CMOs | 120 | Operations Managers, Business Development Executives |
| Pharmaceutical Companies | 100 | R&D Directors, Procurement Managers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Industry Consultants | 60 | Market Analysts, Strategy Consultants |
| Healthcare Providers | 80 | Clinical Research Coordinators, Pharmacists |
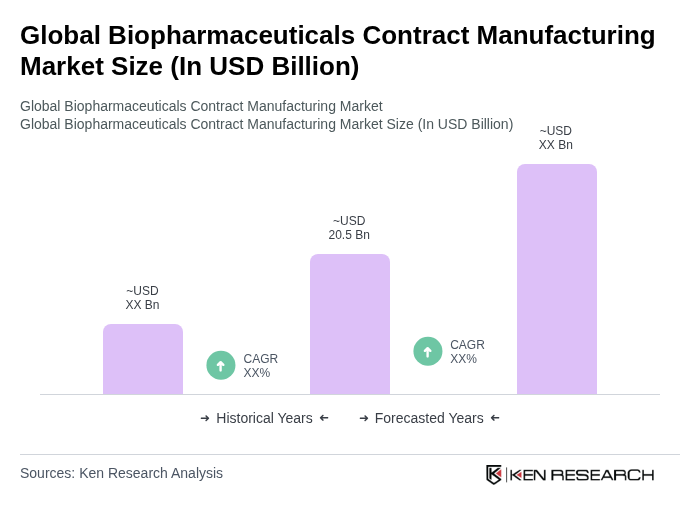
The Global Biopharmaceuticals Contract Manufacturing Market is valued at approximately USD 20.5 billion, driven by increasing demand for biopharmaceuticals, advancements in biotechnology, and the rising prevalence of chronic and infectious diseases.