Region:Middle East
Author(s):Rebecca
Product Code:KRAD5020
Pages:88
Published On:December 2025
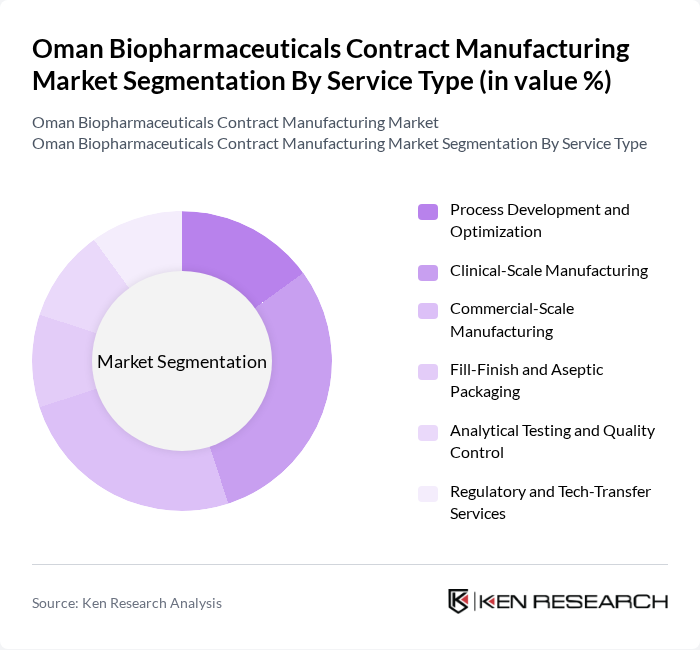
By Service Type:The service type segmentation includes various essential services that cater to the biopharmaceutical manufacturing process. The key subsegments are Process Development and Optimization, Clinical-Scale Manufacturing, Commercial-Scale Manufacturing, Fill-Finish and Aseptic Packaging, Analytical Testing and Quality Control, and Regulatory and Tech-Transfer Services. Among these, Clinical-Scale Manufacturing is currently leading the market due to the increasing number of clinical trials and the demand for efficient production processes. The focus on quality and compliance in clinical trials drives the need for specialized services, making this subsegment crucial for biopharmaceutical companies.
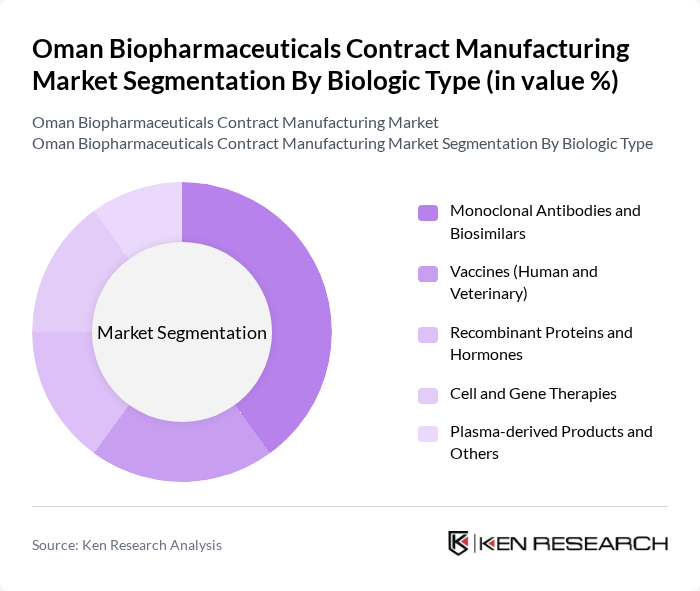
By Biologic Type:This segmentation focuses on the various types of biologics produced in the biopharmaceutical sector. The subsegments include Monoclonal Antibodies and Biosimilars, Vaccines (Human and Veterinary), Recombinant Proteins and Hormones, Cell and Gene Therapies, and Plasma-derived Products and Others. Monoclonal Antibodies and Biosimilars dominate the market due to their widespread application in treating chronic diseases and their increasing acceptance in therapeutic areas. The growing prevalence of diseases requiring monoclonal antibody therapies drives demand, making this subsegment a key player in the biopharmaceutical landscape.
The Oman Biopharmaceuticals Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oman Pharmaceutical Products Co. LLC (Oman), Julphar Gulf Pharmaceutical Industries PJSC (UAE, Active in Oman), Gulf Inject LLC (UAE, Supplying Injectables and Contract Manufacturing to Oman), Biocon Biologics Ltd. (India, Biosimilars Supplier to Oman), Dr. Reddy’s Laboratories Ltd. – Biologics Division (India, Biosimilars in Oman), Celltrion Healthcare Co., Ltd. (South Korea, Biosimilars Partnered in Oman), Samsung Biologics Co., Ltd. (South Korea, Global Biopharmaceutical CDMO with GCC Engagements), Lonza Group Ltd. (Switzerland, Global Biopharmaceutical CDMO with Middle East Reach), Fujifilm Diosynth Biotechnologies (Global CDMO Partnering with MENA Clients), Abu Dhabi Ports – Life Sciences & Pharma Hub / HOPE Consortium (UAE Regional Logistics & Fill-Finish Partner), Aster DM Healthcare – Oman Operations (Regional Healthcare Group Sourcing and Local Partnerships), Badr Al Samaa Group of Hospitals (Oman, Key Buyer and Local Partner for Biopharmaceuticals), Ministry of Health, Sultanate of Oman – Directorate General of Pharmaceutical Affairs and Drug Control (Strategic Buyer and Contracting Authority), Oman Global Logistics Group – ASYAD Group (Logistics and Cold-Chain Partner for Biopharmaceuticals), Selected Emerging Omani Biotech/Pharma Manufacturing Initiatives and Public–Private Partnerships contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman biopharmaceuticals contract manufacturing market appears promising, driven by increasing healthcare demands and government initiatives. As the sector evolves, local manufacturers are likely to enhance their capabilities through technological advancements and strategic partnerships. The focus on sustainability and personalized medicine will further shape the landscape, encouraging innovation and attracting foreign investments. In future, the market is expected to witness significant growth, positioning Oman as a competitive player in the regional biopharmaceutical industry.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Process Development and Optimization Clinical-Scale Manufacturing Commercial-Scale Manufacturing Fill-Finish and Aseptic Packaging Analytical Testing and Quality Control Regulatory and Tech-Transfer Services |
| By Biologic Type | Monoclonal Antibodies and Biosimilars Vaccines (Human and Veterinary) Recombinant Proteins and Hormones Cell and Gene Therapies Plasma-derived Products and Others |
| By Manufacturing Platform | Mammalian Cell Culture Microbial Fermentation Single-use and Modular Systems Hybrid and Continuous Processing |
| By Therapeutic Area | Oncology Infectious Diseases and Vaccines Autoimmune and Inflammatory Disorders Metabolic and Endocrine Disorders Others |
| By Client Type | Global Biopharmaceutical and Vaccine Companies Regional and Local Pharmaceutical Companies Biotechnology Start-ups and Emerging Biotechs Government and Multilateral Agencies Academic and Research Institutions |
| By Contract Type | Project-based Contracts Long-term Strategic Partnerships Dedicated / Captive Capacity Agreements Technology Transfer and Co-development Deals |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah Al Sharqiyah Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical Manufacturing Facilities | 100 | Production Managers, Quality Assurance Leads |
| Contract Manufacturing Organizations (CMOs) | 80 | Business Development Managers, Operations Directors |
| Regulatory Affairs in Biopharmaceuticals | 60 | Regulatory Affairs Specialists, Compliance Officers |
| Research & Development Departments | 70 | R&D Managers, Product Development Scientists |
| Healthcare Providers and Institutions | 90 | Pharmacists, Hospital Administrators |
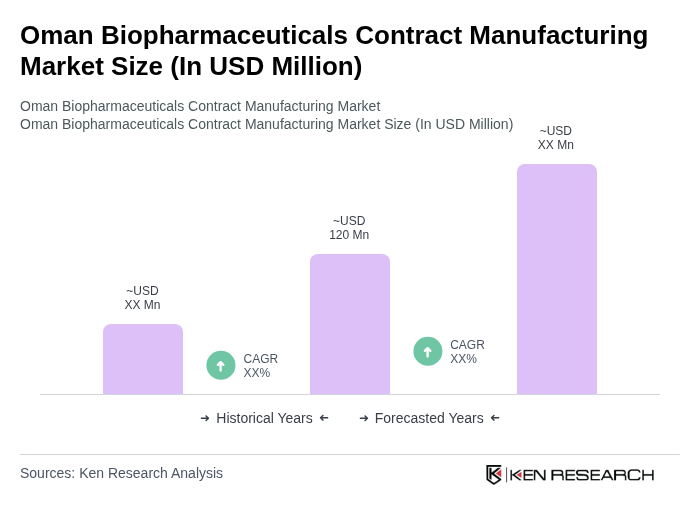
The Oman Biopharmaceuticals Contract Manufacturing Market is valued at approximately USD 120 million, reflecting a significant growth trajectory aligned with the broader Middle East biopharmaceuticals market, which is estimated at around USD 1.5 billion.