Region:Middle East
Author(s):Rebecca
Product Code:KRAD4919
Pages:100
Published On:December 2025
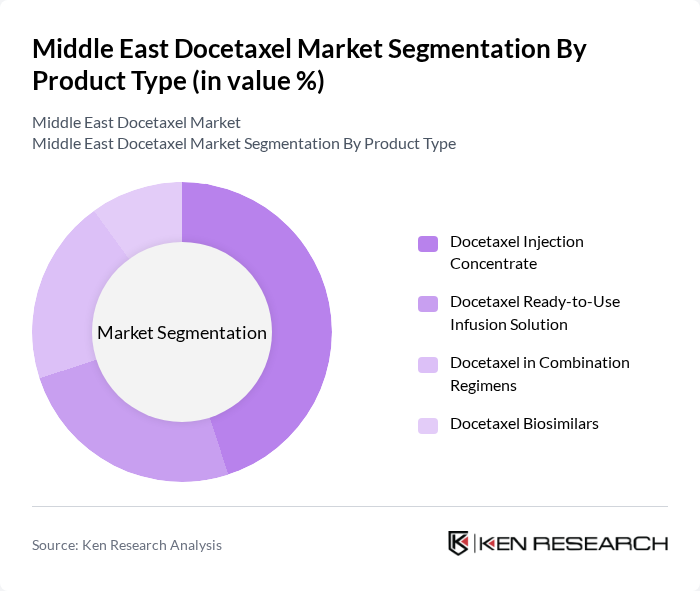
By Product Type:The product type segmentation includes various formulations of docetaxel, each catering to different patient needs and treatment protocols. The subsegments are Docetaxel Injection Concentrate, Docetaxel Ready-to-Use Infusion Solution, Docetaxel in Combination Regimens, and Docetaxel Biosimilars. The Docetaxel Injection Concentrate is currently the leading subsegment due to its widespread use in hospitals and oncology centers, driven by its effectiveness, established clinical protocols, and broad availability in both branded and generic formulations across the region.
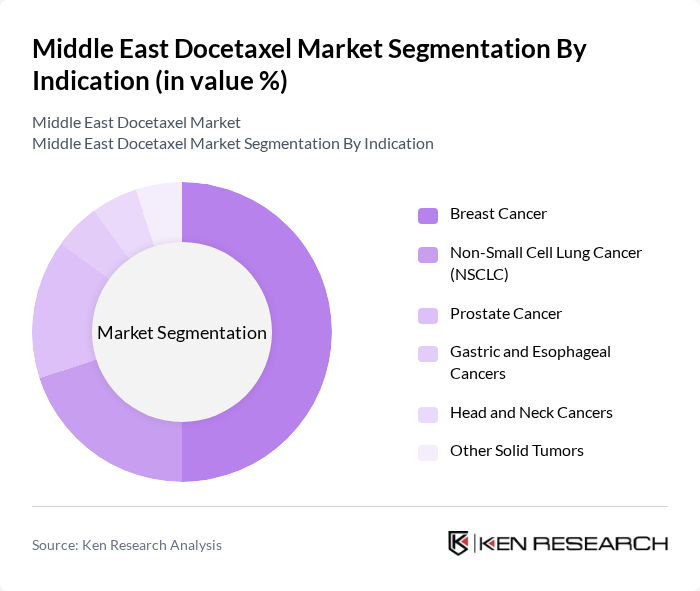
By Indication:The indication segmentation encompasses various cancer types treated with docetaxel, including Breast Cancer, Non-Small Cell Lung Cancer (NSCLC), Prostate Cancer, Gastric and Esophageal Cancers, Head and Neck Cancers, and Other Solid Tumors. Breast Cancer is the dominant indication, driven by the high incidence rates of breast cancer in Middle Eastern women and the established efficacy of docetaxel in adjuvant, neoadjuvant, and metastatic regimens, making it a preferred choice among oncologists.
The Middle East Docetaxel Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi, Teva Pharmaceutical Industries Ltd., Pfizer Inc., Dr. Reddy’s Laboratories Ltd., Cipla Ltd., Hikma Pharmaceuticals plc, Tabuk Pharmaceuticals Manufacturing Co., Julphar (Gulf Pharmaceutical Industries PJSC), SPIMACO ADDWAEIH (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Eczac?ba?? Monrol, Deva Holding A.?., Pharma International Co., Sandoz (a Novartis division), Accord Healthcare Ltd., Mylan N.V. (Viatris Inc.) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East docetaxel market appears promising, driven by increasing investments in healthcare infrastructure and a growing focus on personalized medicine. As governments allocate more resources to oncology, the availability of advanced treatment options is expected to improve. Additionally, the integration of digital health solutions will enhance patient monitoring and treatment adherence, further supporting market growth. The emphasis on patient-centric care will likely lead to better outcomes and increased demand for docetaxel in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Docetaxel Injection Concentrate Docetaxel Ready-to-Use Infusion Solution Docetaxel in Combination Regimens Docetaxel Biosimilars |
| By Indication | Breast Cancer Non-Small Cell Lung Cancer (NSCLC) Prostate Cancer Gastric and Esophageal Cancers Head and Neck Cancers Other Solid Tumors |
| By End-User | Public Hospitals Private Hospitals Specialized Oncology Centers Academic & Research Institutes |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Tender / Institutional Sales |
| By Country | Saudi Arabia United Arab Emirates Turkey Egypt Qatar Rest of Middle East |
| By Line of Therapy | First-Line Therapy Second-Line Therapy Third-Line and Above Palliative / Supportive Use |
| By Payer Type | Government & Public Insurance Schemes Private Health Insurance Out-of-Pocket / Self-Pay Employer-Sponsored Schemes |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Clinics | 100 | Oncologists, Clinic Administrators |
| Hospitals with Oncology Departments | 90 | Pharmacy Directors, Procurement Managers |
| Pharmaceutical Distributors | 60 | Sales Managers, Distribution Coordinators |
| Patient Advocacy Groups | 50 | Patient Representatives, Healthcare Advocates |
| Health Insurance Providers | 70 | Policy Analysts, Claims Managers |
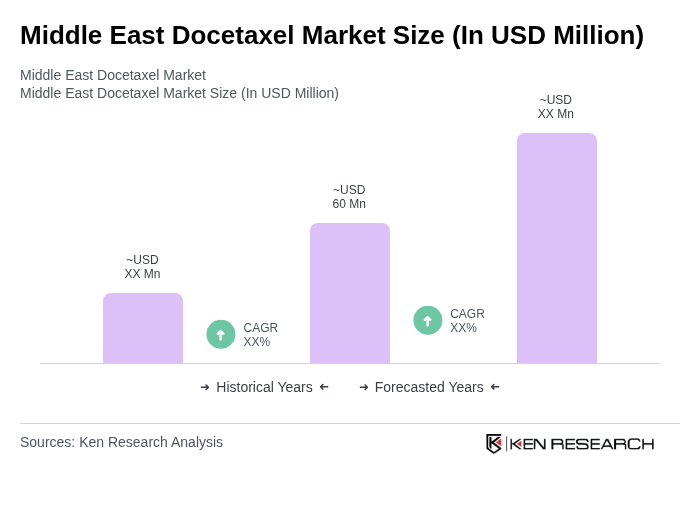
The Middle East Docetaxel Market is valued at approximately USD 60 million, driven by the increasing prevalence of cancer and advancements in healthcare infrastructure and treatment options across the region.