About the Report
Base Year 2024Middle East Pyrogen Testing Market Overview
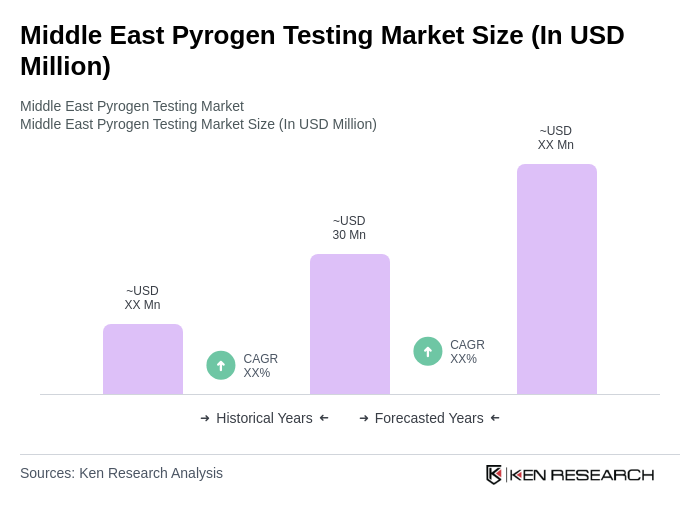
- The Middle East Pyrogen Testing Market is valued at USD 30 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for safety testing in pharmaceuticals and medical devices, alongside stringent regulatory requirements for product safety and efficacy. The rise in biopharmaceuticals, the expansion of pharmaceutical manufacturing, and heightened awareness of the importance of pyrogen testing in ensuring patient safety have further propelled market expansion. Additionally, increased R&D investments, the emergence of personalized medicine, and a growing focus on infection control are key market drivers.
- Countries such as Saudi Arabia, the United Arab Emirates, and Israel dominate the Middle East Pyrogen Testing Market due to their advanced healthcare infrastructure, significant investments in biotechnology, and a growing number of pharmaceutical companies. These nations are also focusing on enhancing their regulatory frameworks to align with international standards, thereby fostering a conducive environment for pyrogen testing services. The region is also witnessing increased adoption of good manufacturing practices (GMP) and international quality standards in pharmaceutical and medical device production.
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Guideline for Testing of Pyrogens in Pharmaceutical Products and Medical Devices, 2023” issued by the Saudi Food and Drug Authority. This regulation mandates that all pharmaceutical and medical device manufacturers conduct pyrogen testing as part of their quality assurance processes. The guideline specifies the required test methods, compliance thresholds, and documentation standards to ensure that products are free from harmful pyrogens, thereby enhancing patient safety and compliance with international standards.
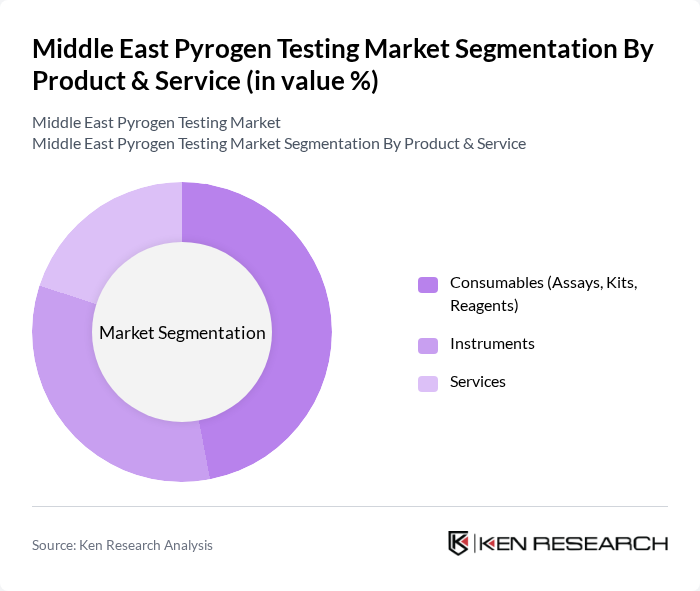
Middle East Pyrogen Testing Market Segmentation
By Product & Service:The product and service segmentation includes consumables, instruments, and services. Consumables, which encompass assays, kits, and reagents, are essential for conducting pyrogen tests. Instruments are critical for executing these tests accurately, while services include testing and consulting provided by specialized laboratories. The consumables segment holds the largest share, reflecting the recurring need for reagents and kits in routine testing.
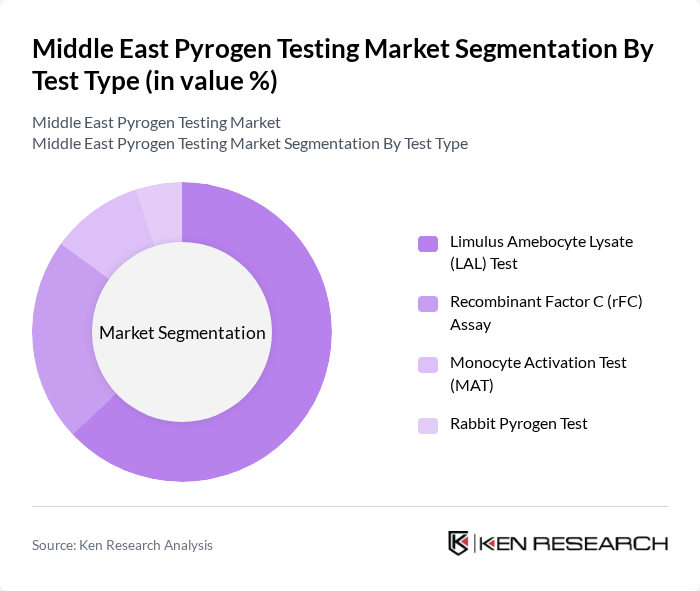
By Test Type:The test type segmentation includes Limulus Amebocyte Lysate (LAL) Test, Recombinant Factor C (rFC) Assay, Monocyte Activation Test (MAT), and Rabbit Pyrogen Test. The LAL test is widely used due to its sensitivity and reliability, while the rFC assay is gaining traction for its ability to detect pyrogens without using animal products. The MAT is increasingly adopted for its ability to detect both endotoxin and non-endotoxin pyrogens, supporting the shift toward animal-free testing methods.
Middle East Pyrogen Testing Market Competitive Landscape
The Middle East Pyrogen Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Charles River Laboratories, Lonza Group AG, WuXi AppTec, Thermo Fisher Scientific Inc., Merck KGaA, bioMérieux SA, FUJIFILM Wako Pure Chemical Corporation, Hyglos GmbH (a bioMérieux company), GenScript Biotech Corporation, Becton, Dickinson and Company (BD), EMD Millipore (Merck Millipore), Sartorius AG, VWR International (Avantor), Seikagaku Corporation, and Toxikon Corporation contribute to innovation, geographic expansion, and service delivery in this space. The competitive landscape is shaped by ongoing investments in R&D, expansion of product portfolios, and strategic collaborations with regional distributors and healthcare institutions.
Middle East Pyrogen Testing Market Industry Analysis
Growth Drivers
- Increasing Demand for Biopharmaceuticals:The Middle East biopharmaceutical market is projected to reach $13 billion in future, driven by a growing population and rising healthcare needs. This surge in biopharmaceutical production necessitates rigorous pyrogen testing to ensure product safety and efficacy. The region's focus on developing advanced therapies, including monoclonal antibodies and vaccines, further amplifies the demand for reliable pyrogen testing solutions, thereby propelling market growth.
- Stringent Regulatory Requirements:Regulatory bodies in the Middle East, such as the Saudi Food and Drug Authority (SFDA) and the UAE Ministry of Health, have implemented strict guidelines for biopharmaceutical testing. In future, compliance with Good Manufacturing Practices (GMP) is expected to be mandatory for over 85% of pharmaceutical companies in the region. This regulatory landscape drives the need for comprehensive pyrogen testing, ensuring that products meet safety standards and gain market approval.
- Technological Advancements in Testing Methods:The introduction of innovative testing technologies, such as the Limulus Amebocyte Lysate (LAL) test and recombinant Factor C assays, is transforming pyrogen testing in the Middle East. In future, it is estimated that 65% of testing laboratories will adopt automated systems, enhancing accuracy and reducing testing times. These advancements not only improve efficiency but also support the growing demand for rapid and reliable testing solutions in the biopharmaceutical sector.
Market Challenges
- High Costs of Testing Procedures:The financial burden associated with pyrogen testing remains a significant challenge for many companies in the Middle East. Testing procedures can cost upwards of $11,000 per batch, which can deter smaller firms from compliance. As the biopharmaceutical industry expands, the need for cost-effective testing solutions becomes critical to ensure that all manufacturers can meet regulatory requirements without compromising product safety.
- Limited Availability of Skilled Professionals:The Middle East faces a shortage of qualified professionals in the field of pyrogen testing, with only about 35% of laboratories employing certified technicians. This gap in expertise can lead to inconsistencies in testing results and compliance issues. As the demand for pyrogen testing grows, addressing this skills shortage through training and education will be essential for maintaining high testing standards in the region.
Middle East Pyrogen Testing Market Future Outlook
The future of the Middle East pyrogen testing market appears promising, driven by ongoing advancements in testing technologies and increasing regulatory scrutiny. As the biopharmaceutical sector continues to expand, the demand for efficient and reliable testing methods will rise. Furthermore, the integration of digital technologies, such as data analytics and automation, is expected to enhance testing accuracy and efficiency, positioning the market for significant growth in the coming years.
Market Opportunities
- Expansion of the Pharmaceutical Industry:The pharmaceutical industry in the Middle East is projected to grow at a rate of 8% annually, creating substantial opportunities for pyrogen testing services. This growth will necessitate increased testing capacity and innovation, allowing testing laboratories to expand their service offerings and cater to a broader client base.
- Growth in Contract Testing Services:The demand for contract testing services is expected to rise, with an estimated market value of $1.6 billion in future. This trend presents opportunities for laboratories to partner with pharmaceutical companies, providing specialized pyrogen testing services that meet regulatory requirements while allowing clients to focus on core business activities.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product & Service | Consumables (Assays, Kits, Reagents) Instruments Services |
| By Test Type | Limulus Amebocyte Lysate (LAL) Test Recombinant Factor C (rFC) Assay Monocyte Activation Test (MAT) Rabbit Pyrogen Test |
| By Application | Pharmaceutical & Biologics Medical Devices Other Applications (e.g., Food, Cosmetics) |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Medical Device Manufacturers Academic & Research Institutes |
| By Country/Region | GCC Countries Levant Region North Africa Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Environmental Protection Agency)
Pharmaceutical and Biotechnology Companies
Medical Device Manufacturers
Laboratory Service Providers
Quality Control and Assurance Departments
Industry Associations (e.g., Middle East Pharmaceutical Association)
Healthcare Providers and Hospitals
Players Mentioned in the Report:
Charles River Laboratories
Lonza Group AG
WuXi AppTec
Thermo Fisher Scientific Inc.
Merck KGaA
bioMerieux SA
FUJIFILM Wako Pure Chemical Corporation
Hyglos GmbH (a bioMerieux company)
GenScript Biotech Corporation
Becton, Dickinson and Company (BD)
EMD Millipore (Merck Millipore)
Sartorius AG
VWR International (Avantor)
Seikagaku Corporation
Toxikon Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Pyrogen Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Pyrogen Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Pyrogen Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for biopharmaceuticals
3.1.2 Stringent regulatory requirements
3.1.3 Rising awareness of product safety
3.1.4 Technological advancements in testing methods
3.2 Market Challenges
3.2.1 High costs of testing procedures
3.2.2 Limited availability of skilled professionals
3.2.3 Variability in regulatory standards across countries
3.2.4 Competition from alternative testing methods
3.3 Market Opportunities
3.3.1 Expansion of the pharmaceutical industry
3.3.2 Growth in contract testing services
3.3.3 Increasing investments in R&D
3.3.4 Collaborations and partnerships among stakeholders
3.4 Market Trends
3.4.1 Shift towards automated testing solutions
3.4.2 Adoption of rapid testing methods
3.4.3 Focus on sustainability in testing processes
3.4.4 Integration of digital technologies in testing
3.5 Government Regulation
3.5.1 Implementation of GMP guidelines
3.5.2 Compliance with ISO standards
3.5.3 Regulatory approvals for new testing methods
3.5.4 Monitoring of testing laboratories
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Pyrogen Testing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Pyrogen Testing Market Segmentation
8.1 By Product & Service
8.1.1 Consumables (Assays, Kits, Reagents)
8.1.2 Instruments
8.1.3 Services
8.2 By Test Type
8.2.1 Limulus Amebocyte Lysate (LAL) Test
8.2.2 Recombinant Factor C (rFC) Assay
8.2.3 Monocyte Activation Test (MAT)
8.2.4 Rabbit Pyrogen Test
8.3 By Application
8.3.1 Pharmaceutical & Biologics
8.3.2 Medical Devices
8.3.3 Other Applications (e.g., Food, Cosmetics)
8.4 By End-User
8.4.1 Pharmaceutical Companies
8.4.2 Biotechnology Firms
8.4.3 Contract Research Organizations (CROs)
8.4.4 Medical Device Manufacturers
8.4.5 Academic & Research Institutes
8.5 By Country/Region
8.5.1 GCC Countries
8.5.2 Levant Region
8.5.3 North Africa
8.5.4 Others
9. Middle East Pyrogen Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Revenue (USD, Middle East)
9.2.2 Market Share (%)
9.2.3 Product Portfolio Breadth (Number of Pyrogen Testing Solutions)
9.2.4 Regional Presence (Number of Middle East Countries Served)
9.2.5 R&D Investment (% of Revenue)
9.2.6 Regulatory Approvals/Certifications (e.g., GMP, ISO)
9.2.7 Customer Base (Number of Key Clients/Institutions)
9.2.8 Distribution Network Strength
9.2.9 Innovation Index (New Product Launches, Patents)
9.2.10 After-Sales Support & Service Quality
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Charles River Laboratories
9.5.2 Lonza Group AG
9.5.3 WuXi AppTec
9.5.4 Thermo Fisher Scientific Inc.
9.5.5 Merck KGaA
9.5.6 bioMérieux SA
9.5.7 FUJIFILM Wako Pure Chemical Corporation
9.5.8 Hyglos GmbH (a bioMérieux company)
9.5.9 GenScript Biotech Corporation
9.5.10 Becton, Dickinson and Company (BD)
9.5.11 EMD Millipore (Merck Millipore)
9.5.12 Sartorius AG
9.5.13 VWR International (Avantor)
9.5.14 Seikagaku Corporation
9.5.15 Toxikon Corporation
10. Middle East Pyrogen Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government procurement policies
10.1.2 Budget allocation for healthcare
10.1.3 Preference for local suppliers
10.1.4 Compliance with international standards
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in laboratory infrastructure
10.2.2 Funding for research initiatives
10.2.3 Expenditure on quality assurance
10.2.4 Allocation for training and development
10.3 Pain Point Analysis by End-User Category
10.3.1 Delays in testing results
10.3.2 High operational costs
10.3.3 Regulatory compliance challenges
10.3.4 Limited access to advanced technologies
10.4 User Readiness for Adoption
10.4.1 Awareness of testing importance
10.4.2 Training needs for staff
10.4.3 Infrastructure readiness
10.4.4 Financial readiness for investment
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of testing efficiency
10.5.2 Expansion into new markets
10.5.3 Long-term cost savings
10.5.4 Increased customer satisfaction
11. Middle East Pyrogen Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategies
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales approaches
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends exploration
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service enhancements
6.3 Customer feedback mechanisms
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling points
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band strategies
9.1.3 Packaging options
9.2 Export Entry Strategy
9.2.1 Target countries analysis
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 JV opportunities
10.2 Greenfield investments
10.3 M&A considerations
10.4 Distributor model evaluation
11. Capital and Timeline Estimation
11.1 Capital requirements analysis
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
14. Potential Partner List
14.1 Distributors identification
14.2 JV opportunities
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity planning
15.2.2 Milestone tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from regional chemical associations and trade bodies
- Review of published market studies and white papers on pyrogen testing technologies
- Examination of regulatory frameworks and compliance guidelines from Middle Eastern health authorities
Primary Research
- Interviews with laboratory managers and technical directors in pyrogen testing facilities
- Surveys with quality assurance professionals in pharmaceutical and biotechnology companies
- Field interviews with regulatory affairs specialists to understand compliance challenges
Validation & Triangulation
- Cross-validation of findings through multiple data sources including industry publications and expert opinions
- Triangulation of market data with insights from key opinion leaders in the pyrogen testing field
- Sanity checks through feedback from industry panels and advisory boards
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on overall pharmaceutical market growth in the Middle East
- Segmentation of the market by application areas such as pharmaceuticals, biotechnology, and medical devices
- Incorporation of regional healthcare spending trends and investment in laboratory infrastructure
Bottom-up Modeling
- Collection of data on the number of pyrogen tests conducted by leading laboratories
- Estimation of average pricing for pyrogen testing services across different sectors
- Volume x price calculations to derive revenue estimates for the pyrogen testing market
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical growth rates and emerging market trends
- Scenario analysis based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Laboratories | 100 | Laboratory Managers, Quality Control Analysts |
| Biotechnology Firms | 60 | Research Scientists, Regulatory Affairs Managers |
| Medical Device Manufacturers | 50 | Product Development Engineers, Compliance Officers |
| Healthcare Regulatory Bodies | 40 | Policy Makers, Health Inspectors |
| Academic Research Institutions | 40 | Research Professors, Laboratory Technicians |
Frequently Asked Questions
What is the current value of the Middle East Pyrogen Testing Market?
The Middle East Pyrogen Testing Market is valued at approximately USD 30 million, driven by the increasing demand for safety testing in pharmaceuticals and medical devices, along with stringent regulatory requirements for product safety and efficacy.