Region:Middle East
Author(s):Rebecca
Product Code:KRAC3911
Pages:100
Published On:October 2025
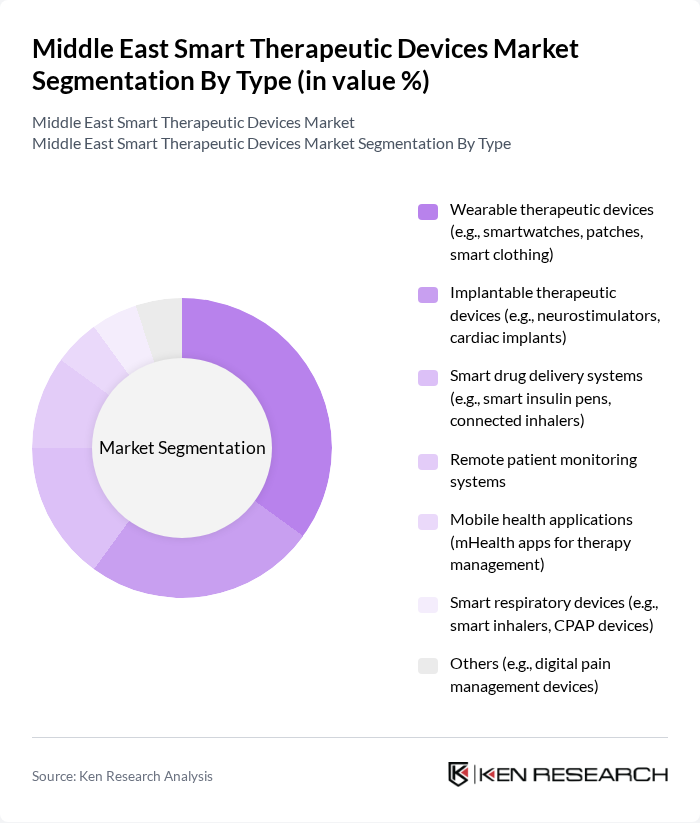
By Type:The market is segmented into various types of smart therapeutic devices, including wearable therapeutic devices, implantable therapeutic devices, smart drug delivery systems, remote patient monitoring systems, mobile health applications, smart respiratory devices, and others. Among these, wearable therapeutic devices are gaining significant traction due to their convenience, real-time health monitoring capabilities, and alignment with preventive healthcare trends. The highest adoption rates are seen in wearables and remote monitoring systems, particularly for chronic disease management and fitness tracking .
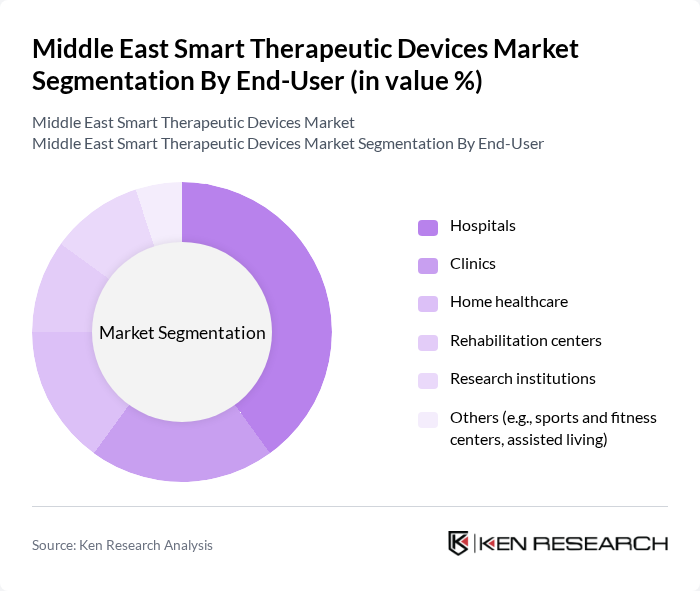
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, rehabilitation centers, research institutions, and others. Hospitals are the leading end-user segment, driven by the increasing adoption of smart therapeutic devices to enhance patient care and operational efficiency. The growing trend of telemedicine and remote patient monitoring is also contributing to the rise of home healthcare as a significant segment. The highest market share is held by hospitals, followed by clinics and home healthcare, reflecting the region’s focus on institutional adoption and digital health transformation .
The Middle East Smart Therapeutic Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Abbott Laboratories, Philips Healthcare, Siemens Healthineers, Johnson & Johnson, Boston Scientific, Stryker Corporation, B. Braun Melsungen AG, GE Healthcare, Roche Diagnostics, Zimmer Biomet, Terumo Corporation, Smith & Nephew, Canon Medical Systems, Hologic, Inc., Burjeel Holdings, Hikma Pharmaceuticals, Al Faisaliah Medical Systems, Gulf Medical Co. Ltd., and Axiom Space (regional partnership) contribute to innovation, geographic expansion, and service delivery in this space. Strategic alliances between international medtech companies and regional healthcare institutions are accelerating the integration of advanced smart therapeutic devices into clinical practice, supported by compliance with SFDA, MOHAP, and ISO 13485 standards .
The future of the Middle East smart therapeutic devices market appears promising, driven by technological innovations and increasing healthcare investments. As the region embraces digital health solutions, the integration of AI and machine learning into therapeutic devices will enhance patient care. Additionally, the expansion of telemedicine services is expected to facilitate remote monitoring, improving access to healthcare. These trends indicate a shift towards more efficient, patient-centered care models, positioning the market for significant growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Wearable therapeutic devices (e.g., smartwatches, patches, smart clothing) Implantable therapeutic devices (e.g., neurostimulators, cardiac implants) Smart drug delivery systems (e.g., smart insulin pens, connected inhalers) Remote patient monitoring systems Mobile health applications (mHealth apps for therapy management) Smart respiratory devices (e.g., smart inhalers, CPAP devices) Others (e.g., digital pain management devices) |
| By End-User | Hospitals Clinics Home healthcare Rehabilitation centers Research institutions Others (e.g., sports and fitness centers, assisted living) |
| By Application | Diabetes management Cardiovascular therapy Respiratory therapy Pain management Neurological disorders Others |
| By Distribution Channel | Pharmacies Online platforms Direct-to-consumer (DTC) Hospitals and clinics Others (e.g., specialty retailers, hypermarkets) |
| By Region | GCC Countries (Saudi Arabia, UAE, Kuwait, Qatar, Oman, Bahrain) Levant Region (Jordan, Lebanon, etc.) North Africa (Egypt, Morocco, etc.) Others |
| By Price Range | Low-end devices Mid-range devices High-end devices |
| By User Demographics | Age groups Gender Socioeconomic status Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Smart Wearable Devices | 100 | Healthcare Providers, Device Manufacturers |
| Remote Patient Monitoring Solutions | 80 | Telehealth Coordinators, IT Managers |
| Smart Drug Delivery Systems | 60 | Pharmaceutical Representatives, Clinical Researchers |
| Rehabilitation Robotics | 50 | Physical Therapists, Rehabilitation Center Directors |
| Chronic Disease Management Devices | 70 | End-users, Caregivers, Health Insurance Analysts |
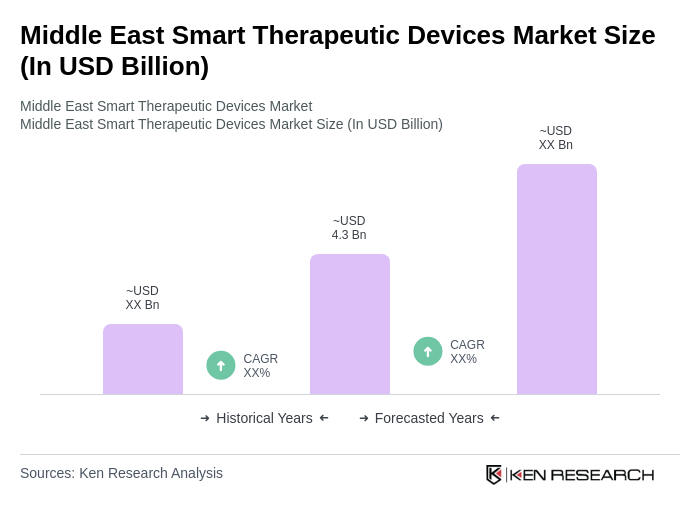
The Middle East Smart Therapeutic Devices Market is valued at approximately USD 4.3 billion, reflecting significant growth driven by the increasing prevalence of chronic diseases and advancements in healthcare technology.