About the Report
Base Year 2024Middle East Traumatic Brain Injury Biomarkers Market Overview
- The Middle East Traumatic Brain Injury Biomarkers Market is valued at USD 1.1 billion, based on a five-year historical analysis. Growth is primarily driven by the increasing prevalence of traumatic brain injuries, advances in biomarker research, and rising demand for accurate diagnostic tools. Additional contributors include the integration of innovative healthcare technologies, expansion of point-of-care diagnostics, and heightened awareness of brain health across the region. Notably, rapid blood-based diagnostic platforms and expanded clinical trial activity are accelerating market adoption .
- Key players in this market includeSaudi Arabia, the United Arab Emirates, and Egypt. These countries lead due to advanced healthcare infrastructure, substantial investments in medical research, and a high incidence of road traffic accidents, which contribute to traumatic brain injury prevalence. The presence of leading healthcare institutions and research facilities, combined with government-backed initiatives for neurological diagnostics, further strengthens their market position .
- In 2023, the UAE government implemented theNational Strategy for Brain Health, issued by the UAE Ministry of Health and Prevention. This binding instrument mandates the establishment of specialized centers for brain injury diagnosis and treatment, allocates funding for biomarker research and development, and sets operational standards for clinical validation and reporting. The strategy requires compliance with diagnostic protocols and reporting thresholds for traumatic brain injury cases, thereby fostering innovation and enhancing healthcare delivery in the region .
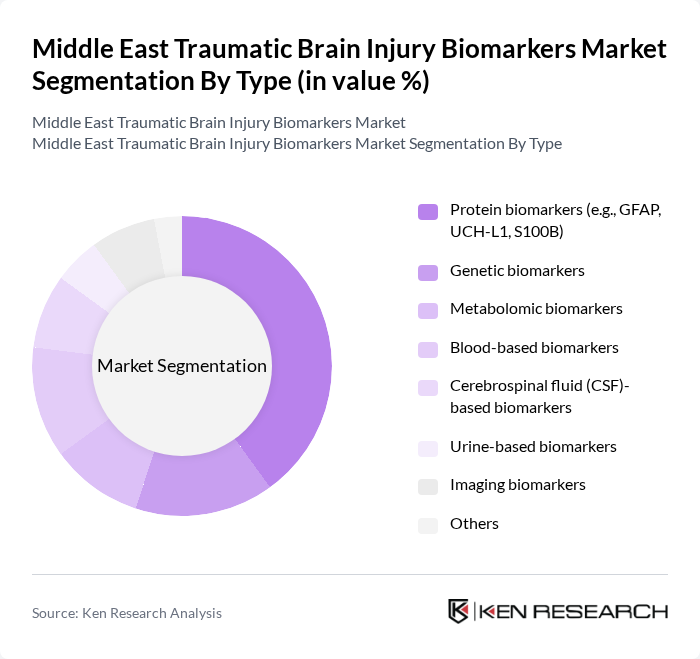
Middle East Traumatic Brain Injury Biomarkers Market Segmentation
By Type:The market is segmented into protein biomarkers, genetic biomarkers, metabolomic biomarkers, blood-based biomarkers, cerebrospinal fluid (CSF)-based biomarkers, urine-based biomarkers, imaging biomarkers, and others.Protein biomarkers, including GFAP, UCH-L1, and S100B, are the leading segment due to their established clinical utility, rapid response post-injury, and high specificity and sensitivity for traumatic brain injury diagnosis. Blood-based biomarkers are increasingly adopted for their ease of integration into point-of-care diagnostics, while genetic and metabolomic biomarkers are gaining traction for personalized medicine applications .
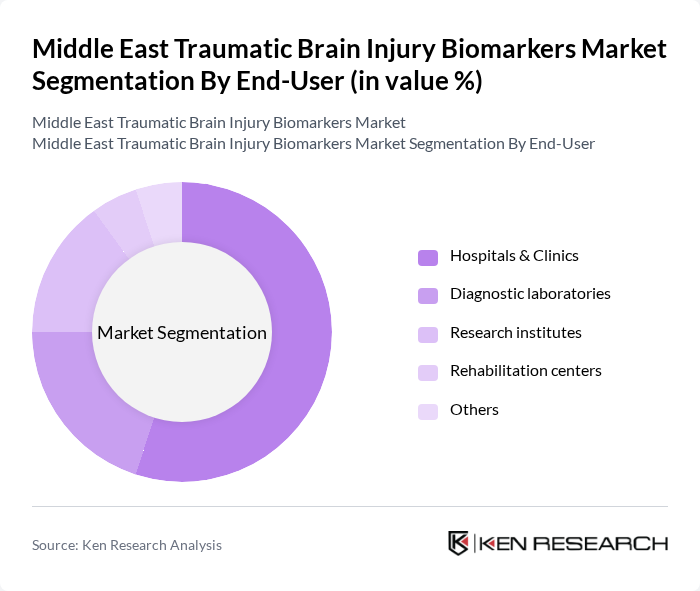
By End-User:The market is segmented by end-users, including hospitals & clinics, diagnostic laboratories, research institutes, rehabilitation centers, and others.Hospitals and clinicsdominate due to the high volume of acute TBI cases requiring immediate diagnosis and treatment, and the growing adoption of advanced diagnostic technologies. Diagnostic laboratories and research institutes are expanding their roles through clinical validation studies and biomarker assay development .
Middle East Traumatic Brain Injury Biomarkers Market Competitive Landscape
The Middle East Traumatic Brain Injury Biomarkers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Quanterix Corporation, BioDirection, Inc., Banyan Biomarkers, Inc., Siemens Healthineers, Roche Diagnostics, Medicortex Finland Oy, BrainBox Solutions, Inc., Thermo Fisher Scientific, BioMérieux, Randox Laboratories, PerkinElmer, Inc., Myriad Genetics, Erytech Pharma, Celerion contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Traumatic Brain Injury Biomarkers Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Traumatic Brain Injuries:The Middle East has witnessed a significant rise in traumatic brain injuries, with an estimated 1.6 million cases reported annually. According to the World Health Organization, road traffic accidents contribute to approximately 32% of these injuries. This alarming trend necessitates advanced diagnostic tools, including biomarkers, to improve patient outcomes and drive market growth. The increasing incidence of sports-related injuries further exacerbates this issue, highlighting the urgent need for effective biomarker solutions.
- Advancements in Biomarker Research and Technology:The region has seen substantial investments in biomarker research, with funding exceeding $250 million. Innovations in genomics and proteomics are paving the way for the development of novel biomarkers that enhance diagnostic accuracy. Collaborations between universities and healthcare institutions are fostering a conducive environment for research, leading to breakthroughs in identifying and validating biomarkers for traumatic brain injuries, thus propelling market growth.
- Rising Awareness and Diagnosis Rates:Increased public awareness regarding traumatic brain injuries has led to a surge in diagnosis rates, with a reported 30% increase in hospital admissions for head injuries in the last year. Educational campaigns by health authorities have emphasized the importance of early detection and intervention. This heightened awareness is driving demand for biomarker testing, as healthcare providers seek reliable methods to diagnose and monitor brain injuries effectively.
Market Challenges
- High Costs of Biomarker Testing:The financial burden associated with biomarker testing remains a significant challenge, with average costs ranging from $600 to $1,800 per test. This high price point limits accessibility, particularly in low-income regions of the Middle East. As healthcare budgets are strained, many facilities may prioritize essential services over advanced diagnostic testing, hindering market growth and patient access to necessary biomarker evaluations.
- Regulatory Hurdles in Biomarker Approval:The approval process for new biomarkers is often lengthy and complex, with an average timeline of 4 to 6 years for regulatory clearance. Stricter guidelines imposed by health authorities can delay the introduction of innovative biomarker tests into the market. This regulatory environment can stifle innovation and deter investment in biomarker research, ultimately impacting the availability of advanced diagnostic solutions for traumatic brain injuries.
Middle East Traumatic Brain Injury Biomarkers Market Future Outlook
The future of the Middle East traumatic brain injury biomarkers market appears promising, driven by ongoing advancements in technology and increased healthcare investments. As the region shifts towards personalized medicine, the demand for tailored biomarker solutions is expected to rise. Additionally, the integration of artificial intelligence in biomarker analysis will enhance diagnostic accuracy and efficiency, paving the way for innovative healthcare delivery models that prioritize patient outcomes and preventive care strategies.
Market Opportunities
- Development of Point-of-Care Testing Solutions:The demand for point-of-care testing solutions is on the rise, with an estimated market potential of $150 million. These solutions offer rapid and accurate biomarker testing, enabling timely diagnosis and treatment. The convenience of on-site testing can significantly improve patient outcomes, particularly in remote areas with limited access to advanced healthcare facilities.
- Expansion of Telemedicine Services:The telemedicine market in the Middle East is projected to reach $1.5 billion, driven by increased smartphone penetration and internet access. This growth presents an opportunity for integrating biomarker testing into telehealth platforms, allowing patients to receive timely consultations and diagnostic services from the comfort of their homes, thereby enhancing healthcare accessibility and efficiency.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Protein biomarkers (e.g., GFAP, UCH-L1, S100B) Genetic biomarkers Metabolomic biomarkers Blood-based biomarkers Cerebrospinal fluid (CSF)-based biomarkers Urine-based biomarkers Imaging biomarkers Others |
| By End-User | Hospitals & Clinics Diagnostic laboratories Research institutes Rehabilitation centers Others |
| By Application | Diagnosis Prognosis Monitoring treatment response Drug development Research and development Others |
| By Biomarker Class | Neurotransmitters Cytokines Growth factors Enzymes Others |
| By Region | GCC Countries (e.g., Saudi Arabia, UAE) Levant Region (e.g., Jordan, Lebanon) North Africa (e.g., Egypt, Morocco) Others |
| By Testing Method | ELISA Mass spectrometry PCR Imaging techniques Microfluidics-based assays Others |
| By Distribution Channel | Direct sales Online sales Distributors Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, UAE; Saudi Food and Drug Authority)
Biomarker Manufacturers and Producers
Healthcare Providers and Hospitals
Pharmaceutical Companies
Medical Device Manufacturers
Industry Associations (e.g., Middle East Healthcare Association)
Health Insurance Companies
Players Mentioned in the Report:
Abbott Laboratories
Quanterix Corporation
BioDirection, Inc.
Banyan Biomarkers, Inc.
Siemens Healthineers
Roche Diagnostics
Medicortex Finland Oy
BrainBox Solutions, Inc.
Thermo Fisher Scientific
BioMerieux
Randox Laboratories
PerkinElmer, Inc.
Myriad Genetics
Erytech Pharma
Celerion
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Traumatic Brain Injury Biomarkers Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Traumatic Brain Injury Biomarkers Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Traumatic Brain Injury Biomarkers Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of traumatic brain injuries
3.1.2 Advancements in biomarker research and technology
3.1.3 Rising awareness and diagnosis rates
3.1.4 Government initiatives and funding for research
3.2 Market Challenges
3.2.1 High costs of biomarker testing
3.2.2 Limited access to advanced healthcare facilities
3.2.3 Regulatory hurdles in biomarker approval
3.2.4 Lack of standardized testing protocols
3.3 Market Opportunities
3.3.1 Development of point-of-care testing solutions
3.3.2 Expansion of telemedicine services
3.3.3 Collaborations with research institutions
3.3.4 Growing demand for personalized medicine
3.4 Market Trends
3.4.1 Increasing investment in neuroprotective therapies
3.4.2 Integration of AI in biomarker analysis
3.4.3 Shift towards preventive healthcare
3.4.4 Rise in patient-centric healthcare models
3.5 Government Regulation
3.5.1 Stricter guidelines for biomarker validation
3.5.2 Incentives for research and development
3.5.3 Regulations on data privacy and patient consent
3.5.4 Policies promoting public health initiatives
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Traumatic Brain Injury Biomarkers Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Traumatic Brain Injury Biomarkers Market Segmentation
8.1 By Type
8.1.1 Protein biomarkers (e.g., GFAP, UCH-L1, S100B)
8.1.2 Genetic biomarkers
8.1.3 Metabolomic biomarkers
8.1.4 Blood-based biomarkers
8.1.5 Cerebrospinal fluid (CSF)-based biomarkers
8.1.6 Urine-based biomarkers
8.1.7 Imaging biomarkers
8.1.8 Others
8.2 By End-User
8.2.1 Hospitals & Clinics
8.2.2 Diagnostic laboratories
8.2.3 Research institutes
8.2.4 Rehabilitation centers
8.2.5 Others
8.3 By Application
8.3.1 Diagnosis
8.3.2 Prognosis
8.3.3 Monitoring treatment response
8.3.4 Drug development
8.3.5 Research and development
8.3.6 Others
8.4 By Biomarker Class
8.4.1 Neurotransmitters
8.4.2 Cytokines
8.4.3 Growth factors
8.4.4 Enzymes
8.4.5 Others
8.5 By Region
8.5.1 GCC Countries (e.g., Saudi Arabia, UAE)
8.5.2 Levant Region (e.g., Jordan, Lebanon)
8.5.3 North Africa (e.g., Egypt, Morocco)
8.5.4 Others
8.6 By Testing Method
8.6.1 ELISA
8.6.2 Mass spectrometry
8.6.3 PCR
8.6.4 Imaging techniques
8.6.5 Microfluidics-based assays
8.6.6 Others
8.7 By Distribution Channel
8.7.1 Direct sales
8.7.2 Online sales
8.7.3 Distributors
8.7.4 Others
9. Middle East Traumatic Brain Injury Biomarkers Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (regional and segment-specific)
9.2.4 Market Penetration Rate (Middle East TBI biomarker adoption)
9.2.5 Regulatory Approval Status (e.g., CE Mark, local MOH approval)
9.2.6 Product Portfolio Breadth (number and diversity of TBI biomarker assays)
9.2.7 Clinical Validation Strength (number of peer-reviewed studies, clinical trial participation)
9.2.8 Strategic Partnerships (regional hospital, research institute collaborations)
9.2.9 Distribution Network Coverage (Middle East reach, local distributor presence)
9.2.10 Customer Satisfaction Score (regional healthcare provider feedback)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 List of Major Companies
9.5.1 Abbott Laboratories
9.5.2 Quanterix Corporation
9.5.3 BioDirection, Inc.
9.5.4 Banyan Biomarkers, Inc.
9.5.5 Siemens Healthineers
9.5.6 Roche Diagnostics
9.5.7 Medicortex Finland Oy
9.5.8 BrainBox Solutions, Inc.
9.5.9 Thermo Fisher Scientific
9.5.10 BioMérieux
9.5.11 Randox Laboratories
9.5.12 PerkinElmer, Inc.
9.5.13 Myriad Genetics
9.5.14 Erytech Pharma
9.5.15 Celerion
10. Middle East Traumatic Brain Injury Biomarkers Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for healthcare
10.1.2 Decision-making processes
10.1.3 Evaluation criteria for biomarker testing
10.1.4 Collaboration with private sector
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare infrastructure
10.2.2 Funding for research initiatives
10.2.3 Partnerships with technology providers
10.2.4 Allocation for training and development
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to advanced diagnostic tools
10.3.2 Cost of biomarker testing
10.3.3 Integration of new technologies
10.3.4 Availability of skilled personnel
10.4 User Readiness for Adoption
10.4.1 Awareness of biomarker benefits
10.4.2 Training needs for healthcare providers
10.4.3 Infrastructure readiness
10.4.4 Patient acceptance of new testing methods
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of clinical outcomes
10.5.2 Cost savings analysis
10.5.3 Expansion into new applications
10.5.4 Long-term sustainability assessments
11. Middle East Traumatic Brain Injury Biomarkers Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Risk assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience definition
2.4 Communication strategy
2.5 Digital marketing approach
2.6 Event marketing plans
2.7 Feedback mechanisms
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales force deployment
3.5 Partnerships with healthcare providers
3.6 Logistics and supply chain management
3.7 Performance metrics
4. Channel & Pricing Gaps
4.1 Underserved routes analysis
4.2 Pricing bands evaluation
4.3 Competitor pricing comparison
4.4 Customer willingness to pay
4.5 Discount strategies
4.6 Bundling options
4.7 Price elasticity assessment
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends exploration
5.4 Feedback from healthcare professionals
5.5 Future needs forecasting
5.6 Innovation opportunities
5.7 Market entry barriers
6. Customer Relationship
6.1 Loyalty programs design
6.2 After-sales service strategies
6.3 Customer engagement initiatives
6.4 Feedback collection methods
6.5 Relationship management tools
6.6 Community building efforts
6.7 Customer education programs
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling points
7.4 Customer-centric approaches
7.5 Competitive advantages
7.6 Long-term value creation
7.7 Market differentiation strategies
8. Key Activities
8.1 Regulatory compliance measures
8.2 Branding efforts
8.3 Distribution setup
8.4 Training and development programs
8.5 Marketing campaigns
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries selection
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model evaluation
10.5 Risk assessment
10.6 Market entry timelines
10.7 Resource allocation
11. Capital and Timeline Estimation
11.1 Capital requirements analysis
11.2 Timelines for market entry
11.3 Funding sources identification
11.4 Financial projections
11.5 Budget allocation strategies
11.6 Cost management plans
11.7 Risk mitigation strategies
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
12.3 Risk management frameworks
12.4 Control mechanisms
12.5 Strategic alignment
12.6 Performance metrics
12.7 Long-term sustainability
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability assessments
13.3 Profit margin projections
13.4 Revenue growth forecasts
13.5 Cost structure analysis
13.6 Financial health indicators
13.7 Investment return expectations
14. Potential Partner List
14.1 Distributors identification
14.2 Joint Ventures opportunities
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Collaboration with research institutions
14.6 Networking opportunities
14.7 Partnership evaluation criteria
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup<
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published studies on traumatic brain injury (TBI) biomarkers in the Middle East
- Review of market reports from healthcare organizations and academic institutions
- Examination of government health statistics and policy documents related to TBI
Primary Research
- Interviews with neurologists and specialists in TBI treatment across leading hospitals
- Surveys with laboratory managers involved in biomarker testing and analysis
- Focus groups with healthcare professionals to understand market needs and trends
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and literature reviews
- Triangulation of data from clinical studies, market reports, and expert opinions
- Sanity checks through feedback from a panel of TBI research experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the TBI patient population in the Middle East based on regional health data
- Analysis of healthcare expenditure trends related to TBI diagnosis and treatment
- Incorporation of government initiatives aimed at improving TBI care and research funding
Bottom-up Modeling
- Collection of data on the number of diagnostic tests performed annually in hospitals
- Estimation of average costs associated with biomarker testing and treatment
- Volume x cost analysis to derive revenue potential for TBI biomarkers
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering population growth and healthcare advancements
- Scenario modeling based on potential changes in healthcare policies and funding
- Baseline, optimistic, and pessimistic projections for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurology Clinics | 100 | Neurologists, TBI Specialists |
| Diagnostic Laboratories | 70 | Laboratory Managers, Biomedical Scientists |
| Healthcare Policy Makers | 50 | Health Administrators, Policy Analysts |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Patient Representatives |
| Research Institutions | 60 | Research Scientists, Academic Professors |
Frequently Asked Questions
What is the current value of the Middle East Traumatic Brain Injury Biomarkers Market?
The Middle East Traumatic Brain Injury Biomarkers Market is valued at approximately USD 1.1 billion, reflecting a significant growth driven by the rising prevalence of traumatic brain injuries and advancements in biomarker research and diagnostic tools.