Region:Middle East
Author(s):Rebecca
Product Code:KRAD5046
Pages:95
Published On:December 2025
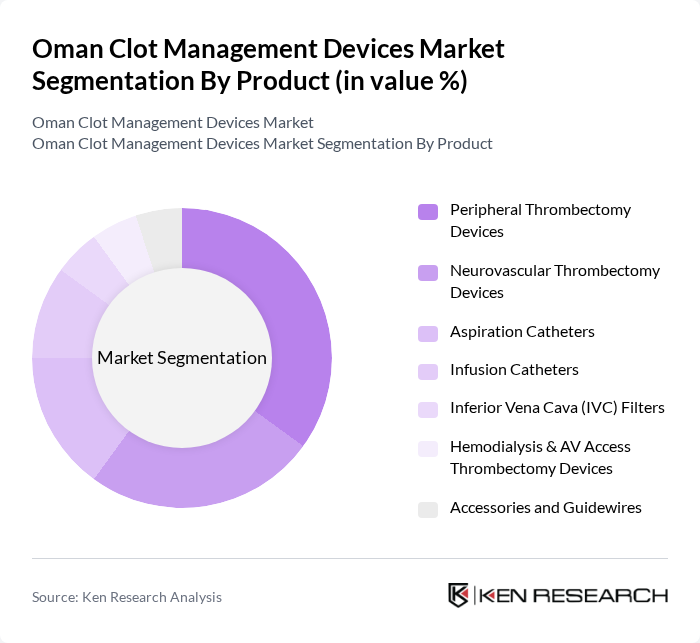
By Product:The product segmentation of the Oman Clot Management Devices Market includes various types of devices designed for effective clot management. The key subsegments are Peripheral Thrombectomy Devices, Neurovascular Thrombectomy Devices, Aspiration Catheters, Infusion Catheters, Inferior Vena Cava (IVC) Filters, Hemodialysis & AV Access Thrombectomy Devices, and Accessories and Guidewires. Among these, Peripheral Thrombectomy Devices are leading the market due to their widespread application in treating deep vein thrombosis, pulmonary embolism, and other peripheral vascular conditions, particularly as minimally invasive mechanical thrombectomy gains preference over systemic thrombolysis for selected patients. The increasing incidence of these conditions, coupled with advancements in device technology such as large?bore aspiration systems, rotational and rheolytic thrombectomy platforms, and improved trackability of catheters, has significantly boosted the demand for these devices.
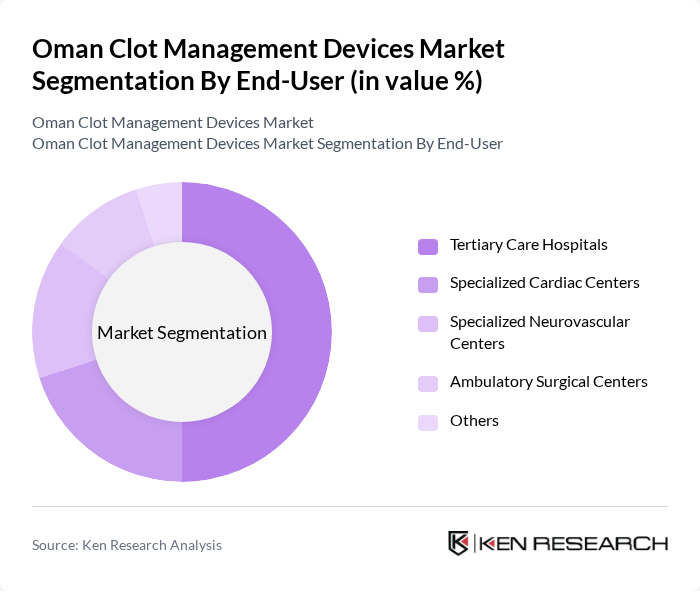
By End-User:The end-user segmentation of the Oman Clot Management Devices Market includes Tertiary Care Hospitals, Specialized Cardiac Centers, Specialized Neurovascular Centers, Ambulatory Surgical Centers, and Others. Tertiary Care Hospitals are the leading end-users due to their comprehensive services, interventional radiology and cardiology suites, intensive care capacity, and advanced treatment capabilities. These facilities are equipped with the latest imaging technologies, catheterization laboratories, and skilled multidisciplinary teams, making them the preferred choice for patients requiring complex clot management procedures such as pulmonary embolism thrombectomy, limb ischemia interventions, and endovascular stroke treatment. The increasing number of patients seeking treatment in these hospitals, supported by national strategies to strengthen noncommunicable disease and stroke care pathways, is driving the demand for clot management devices.
The Oman Clot Management Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, Johnson & Johnson (Cerenovus), Penumbra, Inc., Stryker Corporation, Teleflex Incorporated, AngioDynamics, Inc., Merit Medical Systems, Inc., Cook Medical LLC, Terumo Corporation, Cordis, Philips Image Guided Therapy (Philips Healthcare), Acandis GmbH, MicroPort Scientific Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman clot management devices market appears promising, driven by ongoing technological advancements and government support for healthcare improvements. As the healthcare infrastructure expands, the demand for innovative and effective treatment options is expected to rise. Additionally, the integration of artificial intelligence in medical devices is likely to enhance diagnostic accuracy and treatment efficacy, further propelling market growth. The focus on patient-centric care will also shape future developments in this sector.
| Segment | Sub-Segments |
|---|---|
| By Product | Peripheral Thrombectomy Devices Neurovascular Thrombectomy Devices Aspiration Catheters Infusion Catheters Inferior Vena Cava (IVC) Filters Hemodialysis & AV Access Thrombectomy Devices Accessories and Guidewires |
| By End-User | Tertiary Care Hospitals Specialized Cardiac Centers Specialized Neurovascular Centers Ambulatory Surgical Centers Others |
| By Clinical Application | Deep Vein Thrombosis (DVT) Pulmonary Embolism (PE) Acute Ischemic Stroke Peripheral Arterial & Venous Thrombosis Hemodialysis Access Management Others |
| By Procedure Type | Mechanical Thrombectomy Catheter-Directed Thrombolysis Aspiration Thrombectomy Hybrid / Combination Procedures |
| By Distribution Channel | Direct Institutional Sales Local Distributors / Importers Group Purchasing Organizations (GPOs) Online / E-Procurement Portals |
| By Province | Muscat Dhofar Al Batinah North & South Al Dakhiliyah Al Sharqiyah North & South Others |
| By Patient Demographics | Adults (18–64 Years) Pediatrics (<18 Years) Geriatrics (65+ Years) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Hospitals | 100 | Cardiologists, Interventional Radiologists |
| Healthcare Procurement Units | 80 | Procurement Managers, Supply Chain Directors |
| Medical Device Distributors | 60 | Sales Managers, Product Specialists |
| Clinical Research Organizations | 50 | Clinical Researchers, Regulatory Affairs Managers |
| Patient Advocacy Groups | 40 | Patient Representatives, Healthcare Advocates |
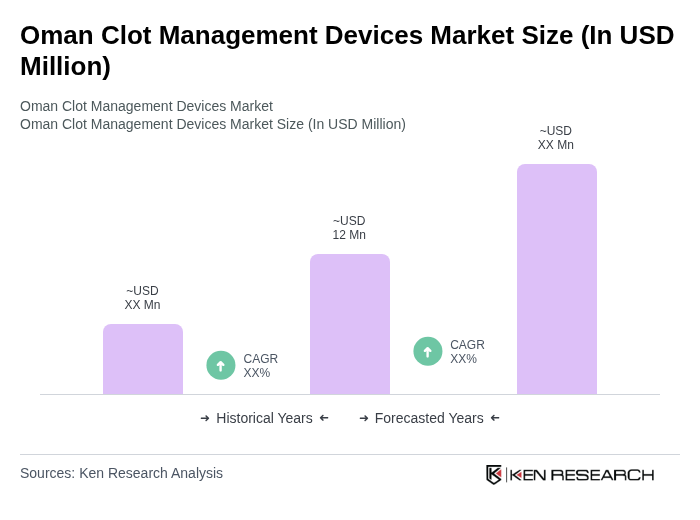
The Oman Clot Management Devices Market is valued at approximately USD 12 million, reflecting a five-year historical analysis and trends in local anticoagulant spending, driven by the rising prevalence of cardiovascular diseases and advancements in healthcare infrastructure.