Region:Asia
Author(s):Dev
Product Code:KRAC3473
Pages:90
Published On:October 2025
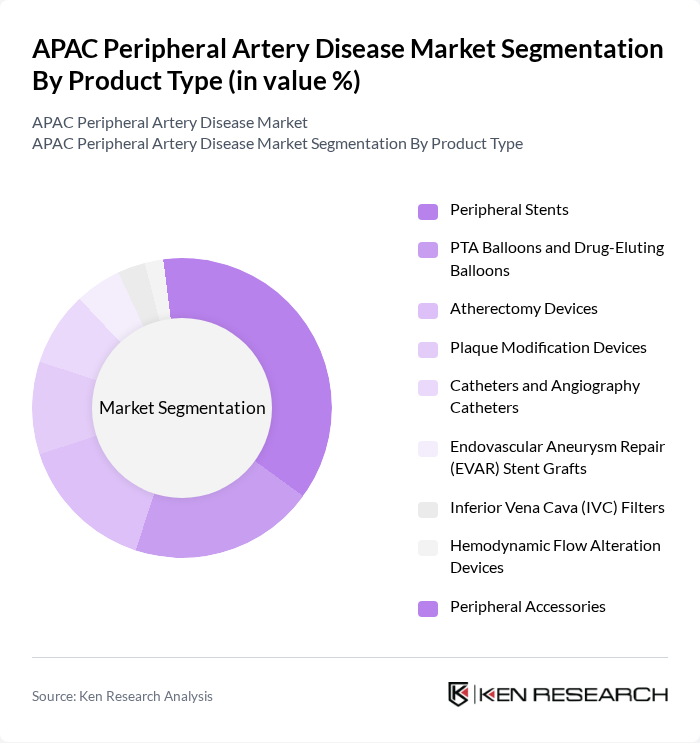
By Product Type:The product type segmentation includes various devices used in the treatment and management of peripheral artery disease. The subsegments are Peripheral Stents, PTA Balloons and Drug-Eluting Balloons, Atherectomy Devices, Plaque Modification Devices, Catheters and Angiography Catheters, Endovascular Aneurysm Repair (EVAR) Stent Grafts, Inferior Vena Cava (IVC) Filters, Hemodynamic Flow Alteration Devices, and Peripheral Accessories. Among these, Peripheral Stents are leading the market due to their effectiveness in treating blockages in peripheral arteries, which is a common complication of peripheral artery disease. Plaque Modification Devices are the fastest-growing segment, driven by technological advancements and increasing adoption in complex vascular interventions.
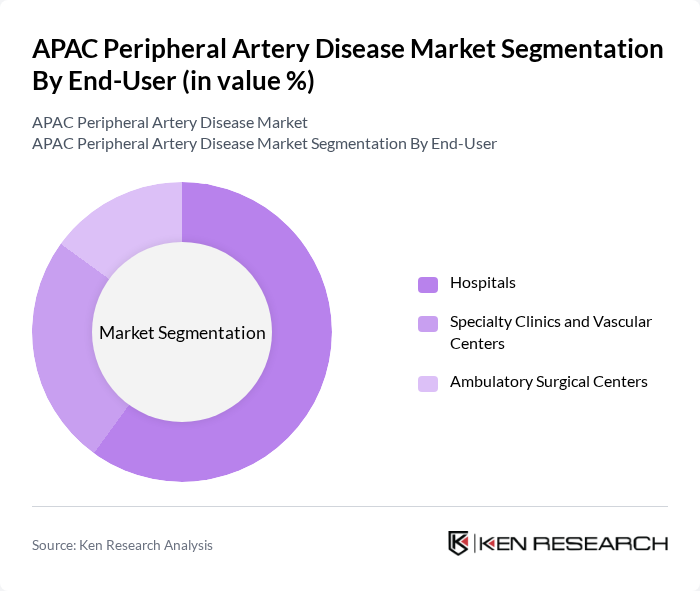
By End-User:The end-user segmentation includes Hospitals, Specialty Clinics and Vascular Centers, and Ambulatory Surgical Centers. Hospitals are the dominant end-user segment due to their comprehensive facilities and resources for treating complex cases of peripheral artery disease. The increasing number of patients seeking treatment in hospitals, coupled with advancements in surgical techniques and technologies, has further solidified their leading position in the market. Ambulatory surgical centers are also experiencing growth, supported by favorable reimbursement policies and the trend toward outpatient care for less complex procedures.
The APAC Peripheral Artery Disease Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic PLC, Boston Scientific Corporation, Abbott Laboratories, Cook Group Incorporated, Terumo Corporation, B. Braun Melsungen AG, Edwards Lifesciences Corporation, Philips Healthcare, Teleflex Incorporated, St. Jude Medical (Abbott), Avinger, Inc., Inari Medical, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
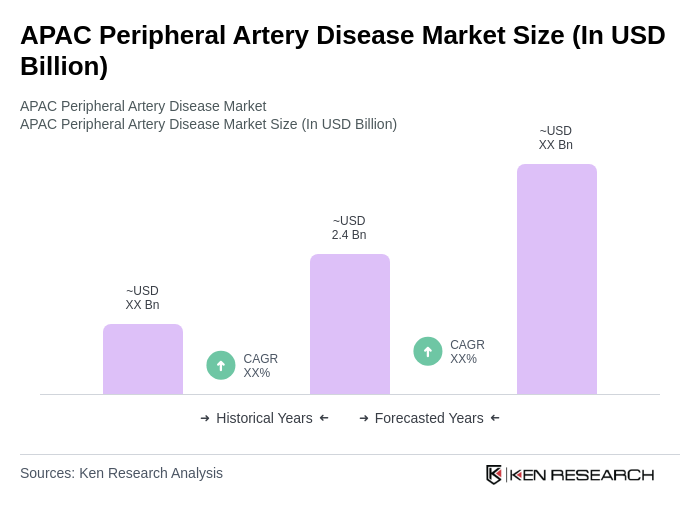
**Market Size Note:** The Asia-Pacific peripheral vascular devices market was valued at approximately USD 2.38 billion in 2023. The peripheral artery disease device segment, a major component of this market, is estimated at approximately USD 2.4 billion, reflecting normalization based on available regional device revenue data. This figure is consistent with the broader device market dynamics in the region and aligns with the growth trajectory driven by demographic and technological factors. **Growth Drivers Update:** The market is further propelled by rising healthcare spending, improved insurance penetration, and government initiatives to modernize healthcare infrastructure. The shift toward minimally invasive treatments and the introduction of novel drug-coated devices and robotic navigation systems are expanding the addressable patient population and improving procedural outcomes. The increasing burden of diabetes and hypertension, coupled with lifestyle changes, continues to elevate the risk profile for peripheral artery disease across APAC, sustaining demand for advanced diagnostic and therapeutic solutions. **Regulation Update:** The National Health Mission (NHM) in India, issued by the Ministry of Health and Family Welfare, Government of India, is the primary framework for non-communicable disease prevention and management, including peripheral artery disease. The NHM mandates the integration of screening, early diagnosis, and management of cardiovascular diseases into primary healthcare services, with compliance required for all state health departments. The mission supports the procurement and deployment of advanced medical devices in public health facilities and promotes public-private partnerships to enhance service delivery.
The future of the APAC Peripheral Artery Disease market appears promising, driven by technological advancements and an increasing focus on preventive healthcare. As healthcare systems adapt to the growing prevalence of cardiovascular diseases, the integration of artificial intelligence in diagnostics and treatment planning is expected to enhance patient outcomes. Furthermore, the shift towards personalized medicine will likely lead to more effective treatment strategies tailored to individual patient needs, fostering a more proactive approach to managing PAD.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Peripheral Stents PTA Balloons and Drug-Eluting Balloons Atherectomy Devices Plaque Modification Devices Catheters and Angiography Catheters Endovascular Aneurysm Repair (EVAR) Stent Grafts Inferior Vena Cava (IVC) Filters Hemodynamic Flow Alteration Devices Peripheral Accessories |
| By End-User | Hospitals Specialty Clinics and Vascular Centers Ambulatory Surgical Centers |
| By Country | India China Japan South Korea Australia Thailand Malaysia Vietnam Indonesia Singapore New Zealand Philippines |
| By Application | Diagnosis and Imaging Treatment and Intervention Monitoring and Follow-up |
| By Distribution Channel | Direct Sales to Hospitals Medical Device Distributors Online and E-commerce Channels |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologists and Vascular Surgeons | 100 | Medical Practitioners, Specialists in PAD |
| Healthcare Administrators | 60 | Hospital Managers, Clinic Directors |
| Patients with Peripheral Artery Disease | 80 | Individuals diagnosed with PAD, Caregivers |
| Medical Device Suppliers | 50 | Sales Representatives, Product Managers |
| Health Policy Experts | 40 | Healthcare Analysts, Policy Makers |
The APAC Peripheral Artery Disease market is valued at approximately USD 2.4 billion, reflecting a significant growth driven by the increasing prevalence of cardiovascular diseases, an aging population, and advancements in medical technology.