Region:Middle East
Author(s):Geetanshi
Product Code:KRAA3466
Pages:86
Published On:January 2026
By Type:
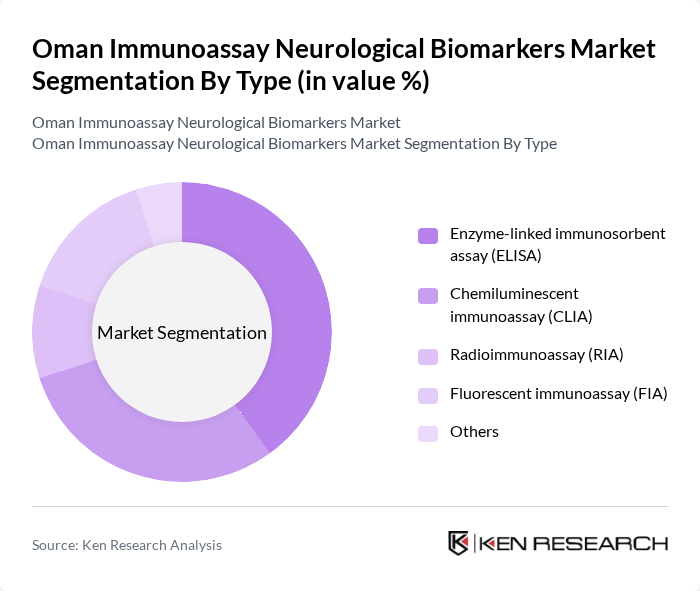
The market is segmented into various types of immunoassays, including Enzyme-linked Immunosorbent Assay (ELISA), Chemiluminescent Immunoassay (CLIA), Radioimmunoassay (RIA), Fluorescent Immunoassay (FIA), and Others. Among these, ELISA is the leading sub-segment due to its widespread use in clinical laboratories for diagnosing neurological disorders. Its cost-effectiveness, ease of use, and reliability make it a preferred choice for healthcare providers. The increasing adoption of ELISA in research and clinical settings is driving its dominance in the market.
By End-User:
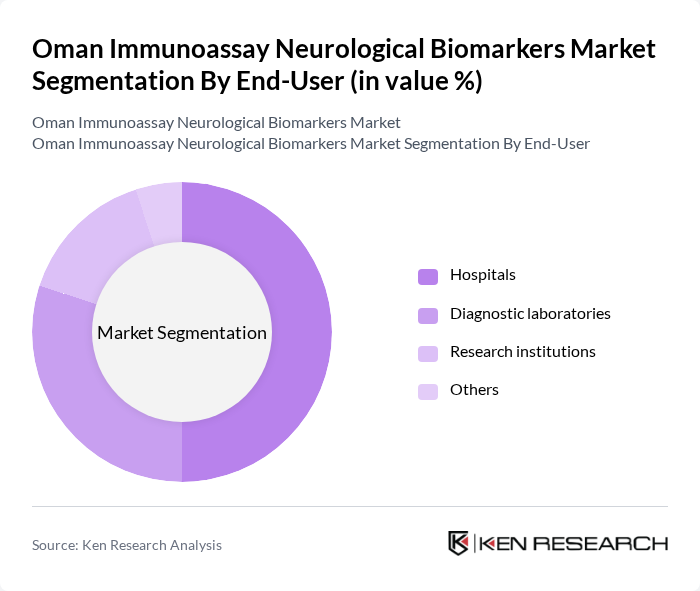
The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, and Others. Hospitals are the dominant end-user segment, accounting for a significant share of the market. This is primarily due to the increasing number of patients seeking neurological diagnostics and the availability of advanced diagnostic facilities in hospitals. The growing trend of hospitals investing in state-of-the-art diagnostic equipment further solidifies their leading position in the market.
The Oman Immunoassay Neurological Biomarkers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, Ortho Clinical Diagnostics, Thermo Fisher Scientific, Quidel Corporation, Fujirebio, PerkinElmer, Mindray Medical International, Sysmex Corporation, Hologic, Inc., DiaSorin S.p.A., Grifols S.A., ACON Laboratories, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman immunoassay neurological biomarkers market appears promising, driven by ongoing advancements in technology and increasing healthcare investments. The integration of artificial intelligence in diagnostics is expected to enhance accuracy and efficiency, while the shift towards personalized medicine will cater to individual patient needs. As healthcare infrastructure expands, more patients will gain access to essential diagnostic tools, ultimately improving early diagnosis and treatment outcomes for neurological disorders in Oman.
| Segment | Sub-Segments |
|---|---|
| By Type | Enzyme-linked Immunosorbent Assay (ELISA) Chemiluminescent Immunoassay (CLIA) Radioimmunoassay (RIA) Fluorescent Immunoassay (FIA) Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Others |
| By Application | Alzheimer's Disease Parkinson's Disease Multiple Sclerosis Others |
| By Biomarker Type | Amyloid Beta Tau Protein Neurofilament Light Chain Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Muscat Salalah Sohar Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurologists and Specialists | 45 | Neurologists, Neuropsychologists |
| Laboratory Managers | 80 | Laboratory Directors, Quality Control Managers |
| Healthcare Policy Makers | 50 | Health Ministry Officials, Regulatory Affairs Managers |
| Patient Advocacy Groups | 60 | Patient Representatives, Caregivers |
| Immunoassay Manufacturers | 70 | Product Managers, Sales Executives |
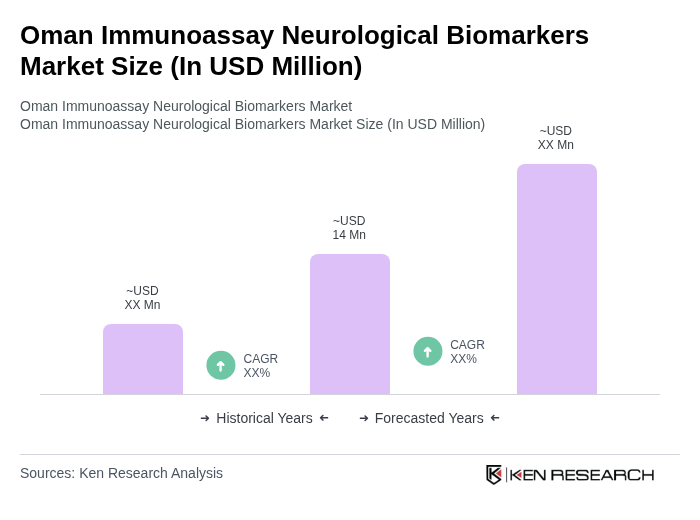
The Oman Immunoassay Neurological Biomarkers Market is valued at approximately USD 12 million, reflecting a significant growth driven by the rising prevalence of neurological disorders and advancements in diagnostic technologies.