About the Report
Base Year 2024Oman in vitro diagnostics enzymes market Overview
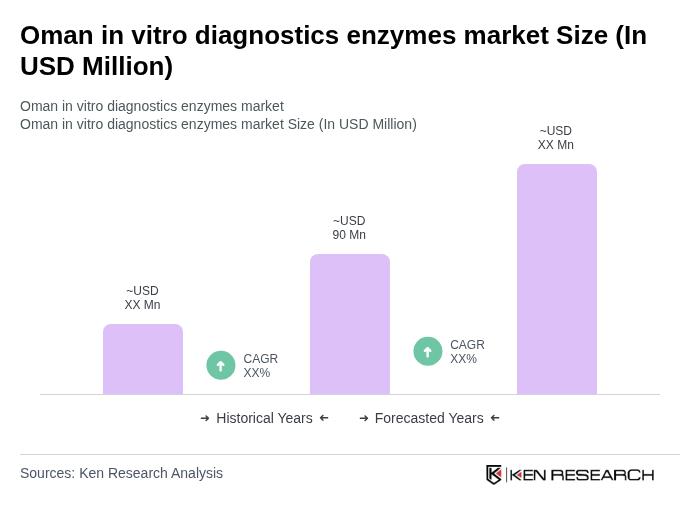
- The Oman in vitro diagnostics enzymes market is valued at USD 90 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases such as diabetes and cardiovascular conditions, rising healthcare expenditure, government investments in healthcare infrastructure, and advancements in diagnostic technologies. The demand for in vitro diagnostics enzymes is further fueled by the growing awareness of early disease detection, the need for efficient diagnostic solutions in healthcare settings, and technological advancements including enzyme engineering for more stable reagents and integration into automated high-throughput platforms.
- Muscat, the capital city, along with other regions like Dhofar and Al Batinah, dominate the Oman in vitro diagnostics enzymes market due to their well-established healthcare infrastructure and the presence of leading diagnostic laboratories. The concentration of healthcare facilities and research institutions in these areas enhances the accessibility and utilization of advanced diagnostic technologies, contributing to market growth.
- The Ministerial Decision No. 51/2017 issued by the Ministry of Health requires registration and certification of in vitro diagnostic devices prior to import, distribution, or use in healthcare facilities. This regulation covers medical devices including IVD reagents and enzymes, mandating compliance with international standards such as ISO 13485 for quality management and specific performance thresholds for diagnostic accuracy. Manufacturers and suppliers must obtain a registration certificate, ensuring traceability, labeling requirements, and post-market surveillance to maintain quality and safety.
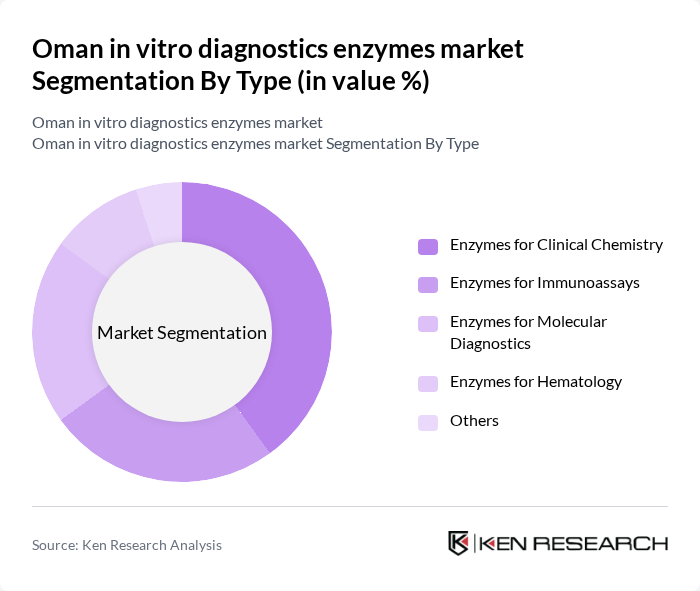
Oman in vitro diagnostics enzymes market Segmentation
By Type:The market is segmented into various types of enzymes used in diagnostics, including enzymes for clinical chemistry, immunoassays, molecular diagnostics, hematology, microbiology, and others. Among these, enzymes for clinical chemistry are the most widely used due to their critical role in routine laboratory tests, which are essential for diagnosing a range of diseases. The increasing demand for accurate and rapid diagnostic results drives the growth of this sub-segment.
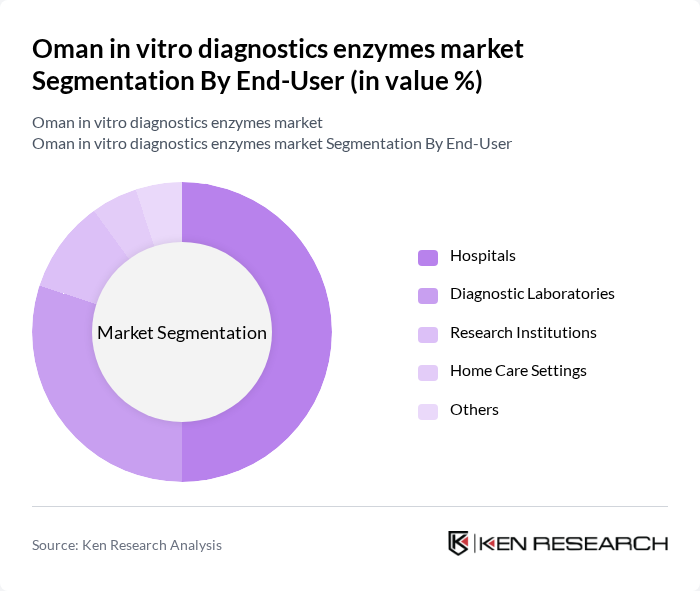
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, research institutions, home care settings, and others. Hospitals are the leading end-users of in vitro diagnostics enzymes, driven by the increasing number of patients requiring diagnostic tests and the need for efficient laboratory services. The growing trend of point-of-care testing in hospitals further enhances the demand for these enzymes.
Oman in vitro diagnostics enzymes market Competitive Landscape
The Oman in vitro diagnostics enzymes market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Danaher Corporation, Ortho Clinical Diagnostics, QIAGEN, PerkinElmer, Sysmex Corporation, Mindray Medical International, Becton, Dickinson and Company, Hologic, Inc., Grifols, DiaSorin S.p.A. contribute to innovation, geographic expansion, and service delivery in this space.
Oman in vitro diagnostics enzymes market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions in Oman is a significant growth driver for the in vitro diagnostics enzymes market. According to the World Health Organization, approximately 30% of the Omani population is affected by chronic diseases, leading to a higher demand for diagnostic testing. This trend is expected to continue, with the Ministry of Health reporting a 20% increase in diabetes cases over the past five years, necessitating advanced diagnostic solutions.
- Rising Demand for Rapid Diagnostic Tests:The demand for rapid diagnostic tests in Oman is surging, driven by the need for timely disease detection and management. The Ministry of Health has noted a 25% increase in the use of rapid tests in healthcare facilities, particularly during health crises like the COVID-19 pandemic. This trend is supported by a growing awareness among patients and healthcare providers about the benefits of quick diagnostics, which can significantly improve patient outcomes and reduce healthcare costs.
- Technological Advancements in Diagnostic Tools:Technological innovations in diagnostic tools are propelling the growth of the in vitro diagnostics enzymes market in Oman. The introduction of automated systems and advanced enzyme-based assays has improved accuracy and efficiency in testing. The Omani government has invested over $60 million in healthcare technology upgrades, facilitating access to state-of-the-art diagnostic equipment. This investment is expected to enhance the capabilities of local laboratories, further driving market growth.
Market Challenges
- High Cost of Advanced Diagnostic Equipment:One of the primary challenges facing the in vitro diagnostics enzymes market in Oman is the high cost associated with advanced diagnostic equipment. Many healthcare facilities struggle to afford these technologies, which can exceed $120,000 per unit. This financial barrier limits the adoption of innovative diagnostic solutions, particularly in rural areas where budgets are constrained, ultimately affecting the overall market growth.
- Limited Awareness Among Healthcare Professionals:Limited awareness and training among healthcare professionals regarding the latest diagnostic technologies pose a significant challenge. A survey conducted by the Omani Medical Association revealed that over 45% of healthcare providers lack adequate knowledge about new diagnostic tools. This gap in understanding can lead to underutilization of advanced diagnostic methods, hindering the market's potential and delaying patient care improvements.
Oman in vitro diagnostics enzymes market Future Outlook
The future of the in vitro diagnostics enzymes market in Oman appears promising, driven by ongoing healthcare reforms and technological advancements. The government’s commitment to enhancing healthcare infrastructure is expected to facilitate the adoption of innovative diagnostic solutions. Additionally, the integration of artificial intelligence in diagnostics is likely to streamline processes and improve accuracy. As healthcare facilities expand and invest in training, the market is poised for significant growth, addressing both current challenges and emerging opportunities.
Market Opportunities
- Expansion of Healthcare Facilities:The ongoing expansion of healthcare facilities in Oman presents a significant opportunity for the in vitro diagnostics enzymes market. With over 20 new hospitals planned in the near future, the demand for advanced diagnostic tools is expected to rise, creating a favorable environment for market players to introduce innovative products and services.
- Collaborations with International Diagnostic Firms:Collaborations with international diagnostic firms can enhance the capabilities of local companies in Oman. By leveraging global expertise and technology, local firms can improve their product offerings and market reach. Such partnerships are anticipated to foster innovation and increase the availability of advanced diagnostic solutions in the region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Enzymes for Clinical Chemistry Enzymes for Immunoassays Enzymes for Molecular Diagnostics Enzymes for Hematology Enzymes for Microbiology Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Others |
| By Application | Disease Diagnosis Drug Development Clinical Research Quality Control in Manufacturing Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Others |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah Others |
| By Technology | Enzyme-Linked Immunosorbent Assay (ELISA) Polymerase Chain Reaction (PCR) Next-Generation Sequencing (NGS) Mass Spectrometry Others |
| By Product Form | Liquid Enzymes Lyophilized Enzymes Reagent Kits Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman)
Manufacturers and Producers of In Vitro Diagnostics Enzymes
Distributors and Retailers of Medical Devices
Healthcare Providers and Laboratories
Technology Providers for Diagnostic Solutions
Industry Associations (e.g., Oman Medical Association)
Financial Institutions and Banks
Players Mentioned in the Report:
Roche Diagnostics
Abbott Laboratories
Siemens Healthineers
Thermo Fisher Scientific
Bio-Rad Laboratories
Danaher Corporation
Ortho Clinical Diagnostics
QIAGEN
PerkinElmer
Sysmex Corporation
Mindray Medical International
Becton, Dickinson and Company
Hologic, Inc.
Grifols
DiaSorin S.p.A.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman in vitro diagnostics enzymes market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman in vitro diagnostics enzymes market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman in vitro diagnostics enzymes market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for rapid diagnostic tests
3.1.3 Technological advancements in diagnostic tools
3.1.4 Government initiatives to improve healthcare infrastructure
3.2 Market Challenges
3.2.1 High cost of advanced diagnostic equipment
3.2.2 Limited awareness among healthcare professionals
3.2.3 Stringent regulatory requirements
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion of healthcare facilities
3.3.2 Collaborations with international diagnostic firms
3.3.3 Growth in personalized medicine
3.3.4 Increasing investment in research and development
3.4 Market Trends
3.4.1 Shift towards point-of-care testing
3.4.2 Integration of AI in diagnostic processes
3.4.3 Focus on sustainable and eco-friendly products
3.4.4 Rise in telemedicine and remote diagnostics
3.5 Government Regulation
3.5.1 Compliance with international quality standards
3.5.2 Licensing requirements for diagnostic products
3.5.3 Regulations on clinical trials and product approvals
3.5.4 Guidelines for the use of in vitro diagnostics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman in vitro diagnostics enzymes market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman in vitro diagnostics enzymes market Segmentation
8.1 By Type
8.1.1 Enzymes for Clinical Chemistry
8.1.2 Enzymes for Immunoassays
8.1.3 Enzymes for Molecular Diagnostics
8.1.4 Enzymes for Hematology
8.1.5 Enzymes for Microbiology
8.1.6 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories
8.2.3 Research Institutions
8.2.4 Home Care Settings
8.2.5 Others
8.3 By Application
8.3.1 Disease Diagnosis
8.3.2 Drug Development
8.3.3 Clinical Research
8.3.4 Quality Control in Manufacturing
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Retail Pharmacies
8.4.5 Others
8.5 By Region
8.5.1 Muscat
8.5.2 Dhofar
8.5.3 Al Batinah
8.5.4 Al Dakhiliyah
8.5.5 Others
8.6 By Technology
8.6.1 Enzyme-Linked Immunosorbent Assay (ELISA)
8.6.2 Polymerase Chain Reaction (PCR)
8.6.3 Next-Generation Sequencing (NGS)
8.6.4 Mass Spectrometry
8.6.5 Others
8.7 By Product Form
8.7.1 Liquid Enzymes
8.7.2 Lyophilized Enzymes
8.7.3 Reagent Kits
8.7.4 Others
9. Oman in vitro diagnostics enzymes market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Innovation Rate
9.2.8 Distribution Efficiency
9.2.9 Brand Recognition
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Abbott Laboratories
9.5.3 Siemens Healthineers
9.5.4 Thermo Fisher Scientific
9.5.5 Bio-Rad Laboratories
9.5.6 Danaher Corporation
9.5.7 Ortho Clinical Diagnostics
9.5.8 QIAGEN
9.5.9 PerkinElmer
9.5.10 Sysmex Corporation
9.5.11 Mindray Medical International
9.5.12 Becton, Dickinson and Company
9.5.13 Hologic, Inc.
9.5.14 Grifols
9.5.15 DiaSorin S.p.A.
10. Oman in vitro diagnostics enzymes market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Defense
10.1.4 Ministry of Commerce and Industry
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Research and Development Funding
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Diagnostic Laboratories
10.3.3 Research Institutions
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance Levels
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Diagnostic Accuracy
10.5.2 Cost Savings Analysis
11. Oman in vitro diagnostics enzymes market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health organizations and industry associations in Oman
- Review of published articles and white papers on in vitro diagnostics (IVD) enzymes
- Examination of government health statistics and regulatory frameworks affecting the IVD market
Primary Research
- Interviews with key opinion leaders in the healthcare and diagnostics sectors
- Surveys with laboratory managers and technicians using IVD enzymes
- Field visits to hospitals and diagnostic centers to gather firsthand insights
Validation & Triangulation
- Cross-validation of findings through multiple data sources including trade publications and expert interviews
- Triangulation of market data with sales figures from leading enzyme manufacturers
- Sanity checks conducted through expert panel discussions to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare expenditure
- Segmentation of the market by enzyme type and application area (clinical, research, etc.)
- Incorporation of growth trends in the healthcare sector and technological advancements in diagnostics
Bottom-up Modeling
- Collection of sales data from major distributors and manufacturers of IVD enzymes
- Estimation of market share based on product offerings and regional presence
- Volume and pricing analysis to derive revenue projections for each enzyme category
Forecasting & Scenario Analysis
- Utilization of historical growth rates to project future market trends through 2030
- Scenario modeling based on potential regulatory changes and healthcare policy shifts
- Development of best-case, worst-case, and most-likely market scenarios
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Managers, Clinical Pathologists |
| Research Institutions | 75 | Research Scientists, Lab Technicians |
| Healthcare Providers | 90 | Healthcare Administrators, Medical Directors |
| Diagnostic Equipment Suppliers | 60 | Sales Managers, Product Specialists |
| Regulatory Bodies | 50 | Regulatory Affairs Managers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Oman in vitro diagnostics enzymes market?
The Oman in vitro diagnostics enzymes market is valued at approximately USD 90 million, reflecting a significant growth driven by the increasing prevalence of chronic diseases, rising healthcare expenditure, and advancements in diagnostic technologies.