Region:Middle East
Author(s):Dev
Product Code:KRAC4801
Pages:90
Published On:October 2025
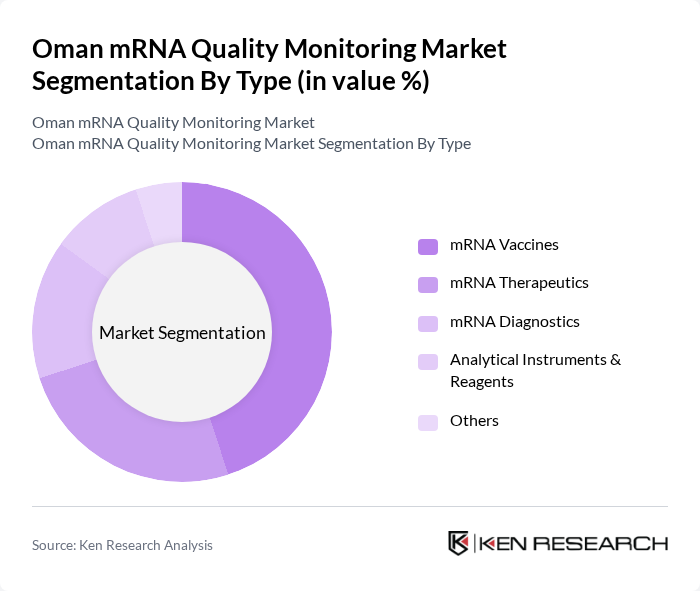
By Type:The market is segmented into various types, including mRNA Vaccines, mRNA Therapeutics, mRNA Diagnostics, Analytical Instruments & Reagents, and Others. Among these, mRNA Vaccines are currently leading the market due to their critical role in combating infectious diseases, particularly during the COVID-19 pandemic. The rapid development and deployment of mRNA vaccines have significantly increased their demand, driving innovation and investment in this segment. The adoption of advanced analytical instruments and reagents is also accelerating, supporting the quality control requirements for both vaccines and therapeutics.
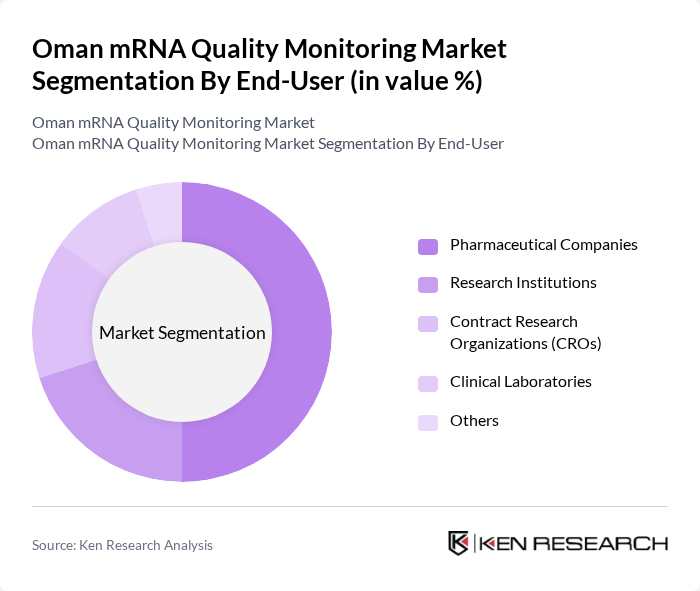
By End-User:The end-user segmentation includes Pharmaceutical Companies, Research Institutions, Contract Research Organizations (CROs), Clinical Laboratories, and Others. Pharmaceutical Companies dominate this segment, driven by their need for high-quality mRNA products for vaccine development and therapeutic applications. The increasing collaboration between pharmaceutical firms and research institutions, as well as the outsourcing of quality control services to specialized CROs, further enhances the demand for mRNA quality monitoring services.
The Oman mRNA Quality Monitoring Market is characterized by a dynamic mix of regional and international players. Leading participants such as Moderna, Inc., BioNTech SE, CureVac N.V., GSK plc, Pfizer Inc., Sanofi S.A., Novartis AG, Merck KGaA, Agilent Technologies, Inc., Bio-Rad Laboratories, Inc., Waters Corporation, Revvity, Inc., GenScript Biotech Corporation, TriLink BioTechnologies (Maravai LifeSciences), Creative Diagnostics contribute to innovation, geographic expansion, and service delivery in this space.
The Oman mRNA quality monitoring market is poised for significant growth, driven by technological advancements and increased government support. As the healthcare sector embraces personalized medicine, the demand for rigorous quality assurance will intensify. Furthermore, collaborations with global biotech firms are expected to enhance local capabilities, fostering innovation. The integration of AI in monitoring processes will streamline operations, ensuring compliance and efficiency. Overall, the market is set to evolve, addressing both local and global healthcare challenges effectively.
| Segment | Sub-Segments |
|---|---|
| By Type | mRNA Vaccines mRNA Therapeutics mRNA Diagnostics Analytical Instruments & Reagents Others |
| By End-User | Pharmaceutical Companies Research Institutions Contract Research Organizations (CROs) Clinical Laboratories Others |
| By Application | Infectious Diseases Cancer Treatment Genetic Disorders Vaccine Quality Control Therapeutic Product Release Testing Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Muscat Salalah Sohar Nizwa Others |
| By Research Phase | Preclinical Clinical Trials Commercialization |
| By Investment Source | Government Funding Private Investments International Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| mRNA Vaccine Production Facilities | 60 | Production Managers, Quality Assurance Officers |
| Healthcare Providers Utilizing mRNA Therapies | 50 | Physicians, Pharmacists, Clinical Researchers |
| Regulatory Bodies and Health Authorities | 40 | Regulatory Affairs Specialists, Policy Makers |
| Biotechnology Research Institutions | 45 | Research Scientists, Lab Directors |
| Public Health Organizations | 40 | Public Health Officials, Epidemiologists |
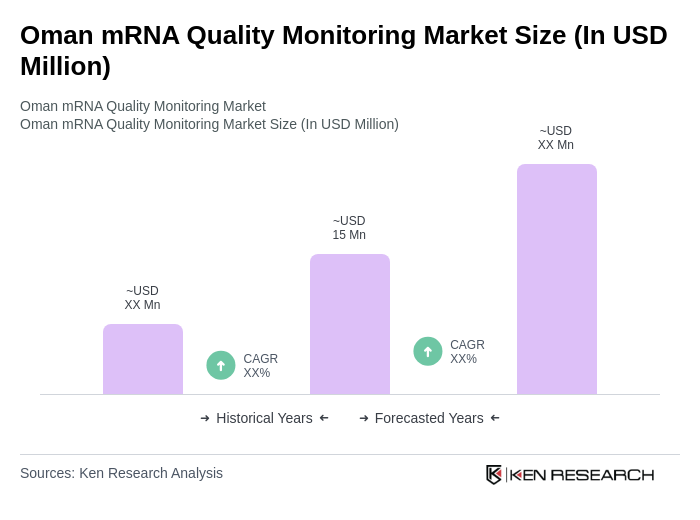
The Oman mRNA Quality Monitoring Market is valued at approximately USD 15 million, reflecting a significant growth driven by the rising demand for mRNA-based therapeutics and vaccines, particularly in response to the COVID-19 pandemic.