Region:Middle East
Author(s):Rebecca
Product Code:KRAD2309
Pages:92
Published On:January 2026
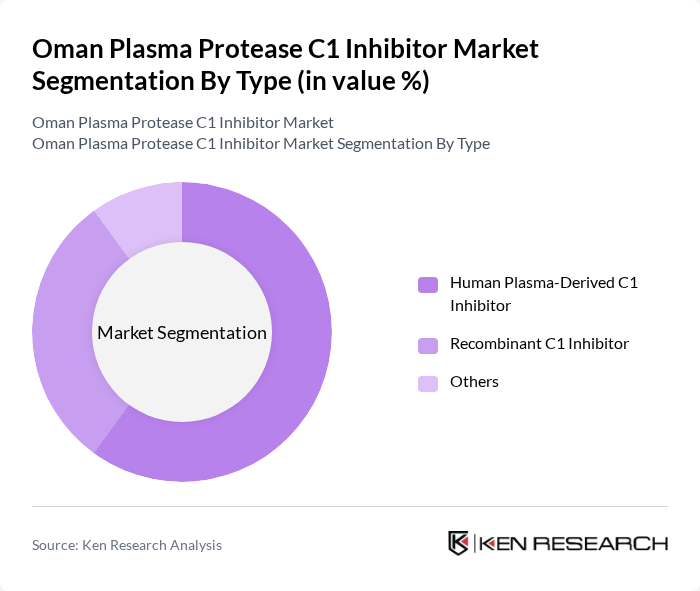
By Type:The market is segmented into three main types: Human Plasma-Derived C1 Inhibitor, Recombinant C1 Inhibitor, and Others. Among these, Human Plasma-Derived C1 Inhibitor is the leading subsegment due to its established efficacy and safety profile, making it the preferred choice for treating patients with hereditary angioedema. The growing acceptance of this treatment option among healthcare providers and patients contributes significantly to its dominance in the market.
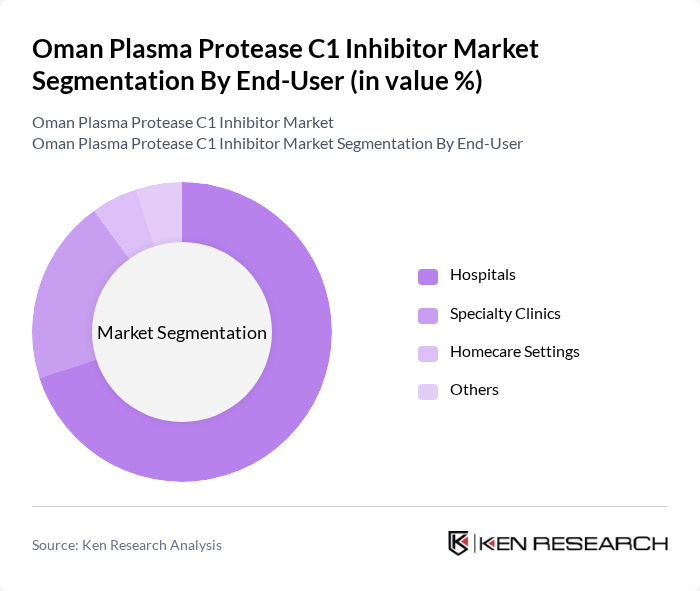
By End-User:The end-user segmentation includes Hospitals, Specialty Clinics, Homecare Settings, and Others. Hospitals dominate this segment, accounting for a significant share of the market. This is primarily due to the high patient volume and the availability of advanced medical facilities that can provide comprehensive care for patients requiring plasma protease C1 inhibitors. The trend towards specialized treatment centers further supports the growth of this subsegment.
The Oman Plasma Protease C1 Inhibitor Market is characterized by a dynamic mix of regional and international players. Leading participants such as CSL Behring, Takeda Pharmaceutical Company, Grifols, Octapharma, Bio Products Laboratory, Kedrion Biopharma, LFB Group, Emergent BioSolutions, Sobi, Haffkine Bio-Pharmaceutical Corporation, Biotest AG, Sanofi, Pfizer, Amgen, UCB contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman plasma protease C1 inhibitor market appears promising, driven by ongoing advancements in healthcare and increasing patient awareness. As the government continues to invest in healthcare infrastructure, the accessibility of specialized treatments is expected to improve. Additionally, the integration of digital health solutions will facilitate better patient management. These trends indicate a positive trajectory for the market, with potential for enhanced patient outcomes and increased demand for innovative therapies in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Human Plasma-Derived C1 Inhibitor Recombinant C1 Inhibitor Others |
| By End-User | Hospitals Specialty Clinics Homecare Settings Others |
| By Distribution Channel | Direct Sales Distributors Online Pharmacies Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Treatment Type | Prophylactic Treatment On-Demand Treatment Others |
| By Region | Muscat Salalah Sohar Others |
| By Policy Support | Subsidies for Rare Disease Treatments Tax Incentives for Pharmaceutical Companies Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Hematologists, Immunologists |
| Hospital Procurement Departments | 80 | Procurement Managers, Supply Chain Officers |
| Clinical Researchers | 60 | Research Scientists, Clinical Trial Coordinators |
| Patient Advocacy Groups | 50 | Patient Representatives, Health Advocates |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Policy Makers |
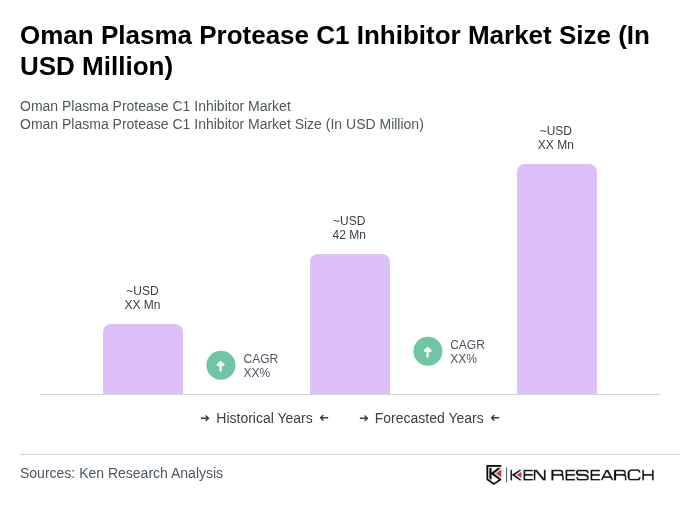
The Oman Plasma Protease C1 Inhibitor Market is valued at approximately USD 2 million, reflecting a five-year historical analysis. This valuation is influenced by the increasing prevalence of hereditary angioedema and the demand for effective treatment options.