Region:Middle East
Author(s):Rebecca
Product Code:KRAD2791
Pages:84
Published On:November 2025
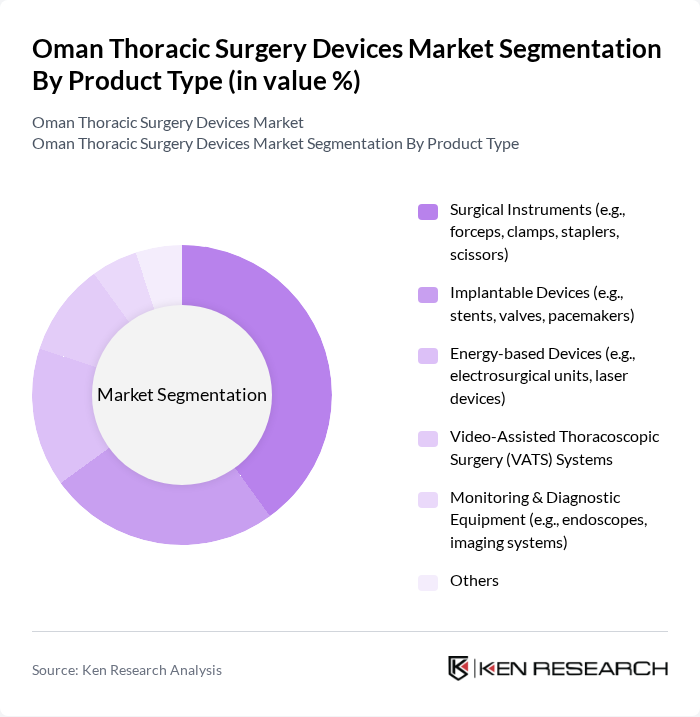
By Product Type:The market is segmented into various product types, including surgical instruments, implantable devices, energy-based devices, video-assisted thoracoscopic surgery (VATS) systems, monitoring & diagnostic equipment, and others. Among these, surgical instruments are currently leading the market due to their essential role in thoracic surgeries and the increasing demand for precision tools. The trend towards minimally invasive surgeries has also boosted the adoption of VATS systems, which are gaining popularity for their benefits in recovery time and reduced complications. The adoption of robotic and video-assisted systems is also rising, reflecting global and regional trends toward minimally invasive thoracic procedures .
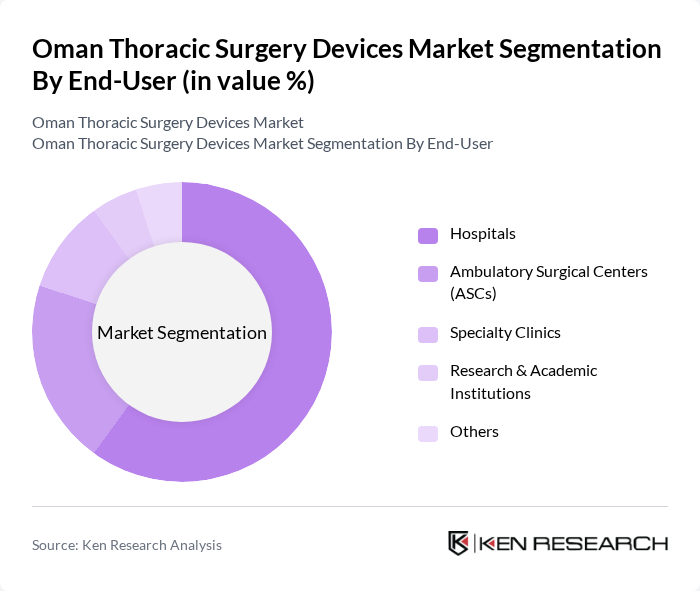
By End-User:The end-user segmentation includes hospitals, ambulatory surgical centers (ASCs), specialty clinics, research & academic institutions, and others. Hospitals dominate this segment due to their comprehensive surgical services and advanced facilities, which are essential for performing complex thoracic surgeries. The increasing number of surgical procedures performed in hospitals, coupled with the growing patient population, continues to drive the demand for thoracic surgery devices in this sector. Ambulatory surgical centers are experiencing rapid growth as minimally invasive and same-day procedures become more common in Oman, reflecting a broader regional shift toward outpatient thoracic interventions .
The Oman Thoracic Surgery Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson (Ethicon, DePuy Synthes), Boston Scientific Corporation, Stryker Corporation, Abbott Laboratories, B. Braun Melsungen AG, Siemens Healthineers AG, Koninklijke Philips N.V. (Philips Healthcare), GE HealthCare Technologies Inc., Olympus Corporation, Cook Medical LLC, Terumo Corporation, Hologic, Inc., Intuitive Surgical, Inc., Zimmer Biomet Holdings, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the thoracic surgery devices market in Oman appears promising, driven by technological advancements and increased healthcare investments. The integration of artificial intelligence in surgical procedures is expected to enhance precision and outcomes. Additionally, the growing trend towards robotic-assisted surgeries will likely reshape surgical practices. As healthcare infrastructure expands, the market is poised for growth, with a focus on improving patient-centric care models and enhancing surgical capabilities across the region.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Surgical Instruments (e.g., forceps, clamps, staplers, scissors) Implantable Devices (e.g., stents, valves, pacemakers) Energy-based Devices (e.g., electrosurgical units, laser devices) Video-Assisted Thoracoscopic Surgery (VATS) Systems Monitoring & Diagnostic Equipment (e.g., endoscopes, imaging systems) Others |
| By End-User | Hospitals Ambulatory Surgical Centers (ASCs) Specialty Clinics Research & Academic Institutions Others |
| By Application | Lung Surgery (e.g., lobectomy, wedge resection) Cardiac Surgery (e.g., valve repair, coronary artery bypass) Esophageal Surgery Mediastinal Surgery Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Muscat Dhofar Al Batinah Others |
| By Technology | Minimally Invasive Techniques (e.g., VATS, robotic-assisted surgery) Robotic Surgery Traditional/Open Surgery Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Thoracic Surgeons | 60 | Consultant Surgeons, Surgical Residents |
| Hospital Procurement Managers | 50 | Procurement Officers, Supply Chain Managers |
| Medical Device Distributors | 40 | Sales Representatives, Distribution Managers |
| Healthcare Policy Makers | 40 | Health Administrators, Policy Analysts |
| Clinical Researchers | 45 | Research Scientists, Clinical Trial Coordinators |
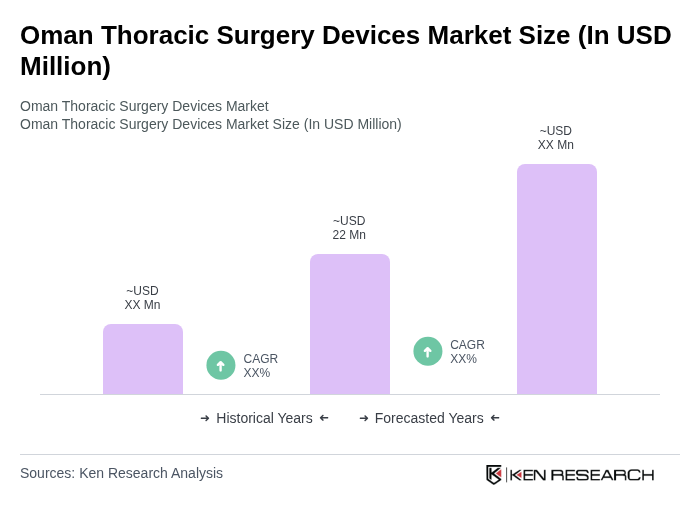
The Oman Thoracic Surgery Devices Market is valued at approximately USD 22 million, reflecting a five-year historical analysis. This valuation is influenced by the rising prevalence of respiratory diseases and advancements in surgical technologies.