Region:Middle East
Author(s):Shubham
Product Code:KRAC4249
Pages:97
Published On:October 2025
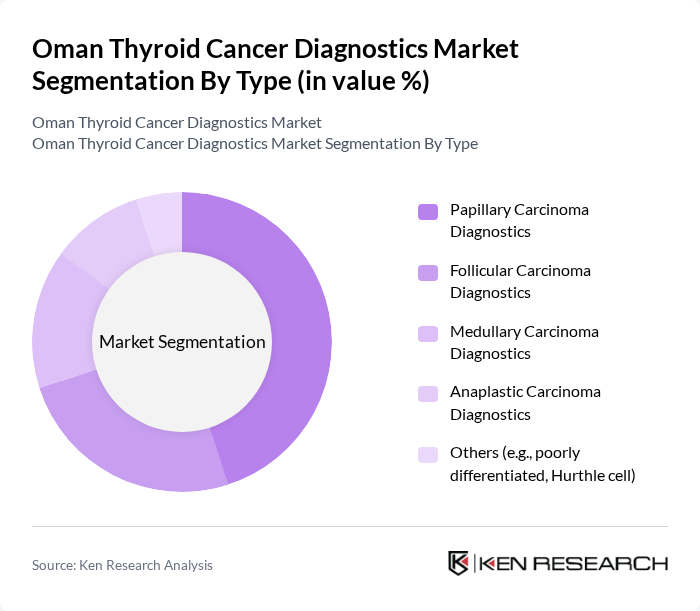
By Type:The market is segmented into various types of thyroid cancer diagnostics, including Papillary Carcinoma Diagnostics, Follicular Carcinoma Diagnostics, Medullary Carcinoma Diagnostics, Anaplastic Carcinoma Diagnostics, and Others (e.g., poorly differentiated, Hurthle cell). Among these, Papillary Carcinoma Diagnostics is the most prominent due to the higher incidence rate of papillary thyroid carcinoma, which accounts for a significant portion of thyroid cancer cases. The increasing awareness and advancements in diagnostic techniques have further propelled this segment's growth.
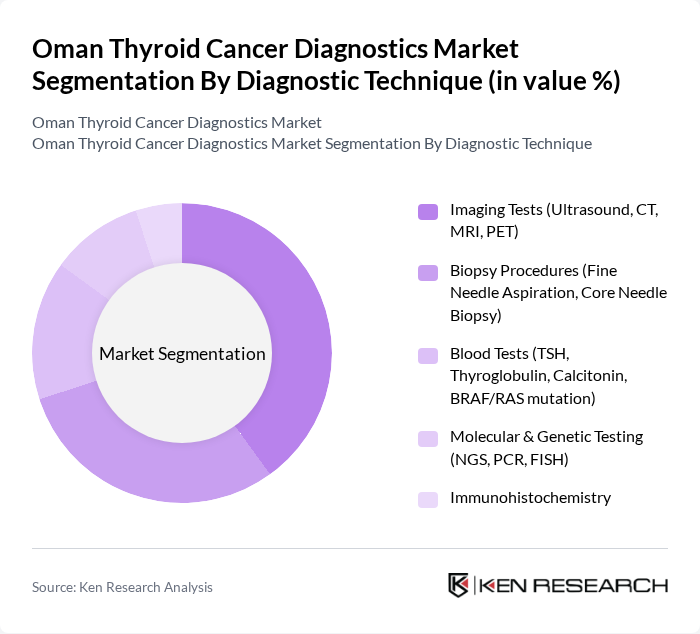
By Diagnostic Technique:The diagnostic techniques used in the market include Imaging Tests (Ultrasound, CT, MRI, PET), Biopsy Procedures (Fine Needle Aspiration, Core Needle Biopsy), Blood Tests (TSH, Thyroglobulin, Calcitonin, BRAF/RAS mutation), Molecular & Genetic Testing (NGS, PCR, FISH), and Immunohistochemistry. Imaging Tests are currently leading the market due to their non-invasive nature and effectiveness in identifying thyroid nodules, which are often precursors to cancer. Molecular and genetic testing, while still a smaller segment, is expected to grow as next-generation sequencing and mutation assays become more accessible.
The Oman Thyroid Cancer Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, Roche Diagnostics, Abbott Laboratories, GE HealthCare, Philips Healthcare, Thermo Fisher Scientific, Bio-Rad Laboratories, PerkinElmer, Agilent Technologies, Becton, Dickinson and Company, Hologic, Inc., Quest Diagnostics, Laboratory Corporation of America Holdings (LabCorp), Sysmex Corporation, Mindray Medical International Limited, Illumina, Inc., QIAGEN N.V., bioMérieux S.A., Toshiba Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Oman thyroid cancer diagnostics market appears promising, driven by ongoing advancements in technology and increased healthcare investments. In the future, the government plans to allocate an additional $10 million towards cancer research and diagnostic infrastructure. Furthermore, the integration of telemedicine is expected to enhance access to diagnostic services, particularly in underserved areas, thereby improving early detection rates and patient outcomes in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Papillary Carcinoma Diagnostics Follicular Carcinoma Diagnostics Medullary Carcinoma Diagnostics Anaplastic Carcinoma Diagnostics Others (e.g., poorly differentiated, Hurthle cell) |
| By Diagnostic Technique | Imaging Tests (Ultrasound, CT, MRI, PET) Biopsy Procedures (Fine Needle Aspiration, Core Needle Biopsy) Blood Tests (TSH, Thyroglobulin, Calcitonin, BRAF/RAS mutation) Molecular & Genetic Testing (NGS, PCR, FISH) Immunohistochemistry |
| By End-User | Hospitals Diagnostic Laboratories Cancer Research Centers Academic & University Laboratories Outpatient Clinics |
| By Distribution Channel | Direct Sales Distributors Online Sales |
| By Application | Screening Diagnosis Recurrence Monitoring Therapy Monitoring |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah |
| By Patient Demographics | Age Group Gender Socioeconomic Status |
| By Technology | Conventional Methods Advanced Diagnostic Technologies (AI-assisted Ultrasound, Multiparametric Imaging) Molecular Diagnostics (NGS, PCR, Liquid Biopsy) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists and Endocrinologists | 60 | Medical Doctors, Cancer Specialists |
| Diagnostic Laboratories | 50 | Laboratory Managers, Technicians |
| Healthcare Policy Makers | 40 | Health Ministry Officials, Public Health Experts |
| Thyroid Cancer Patients | 50 | Patients, Caregivers |
| Medical Equipment Suppliers | 40 | Sales Representatives, Product Managers |
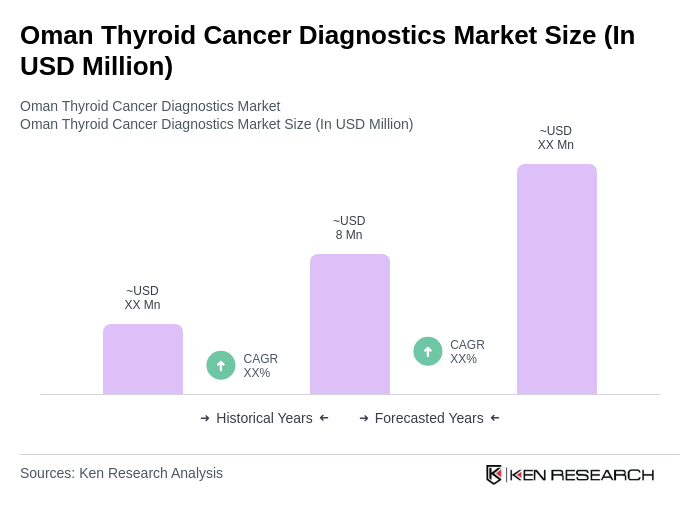
The Oman Thyroid Cancer Diagnostics Market is valued at approximately USD 8 million, reflecting a five-year historical analysis. This valuation is influenced by factors such as increased awareness of thyroid cancer and advancements in diagnostic technologies.