Region:Middle East
Author(s):Rebecca
Product Code:KRAD4975
Pages:87
Published On:December 2025
By Type (Needle Length):The market is segmented based on needle length, which includes various categories that cater to different medical needs and preferences. The subsegments include Amniocentesis Needles < 100 mm, Amniocentesis Needles 100–150 mm, Amniocentesis Needles > 150 mm, and Others (specialized/customized needles). Each subsegment serves specific clinical requirements, influencing their market presence.
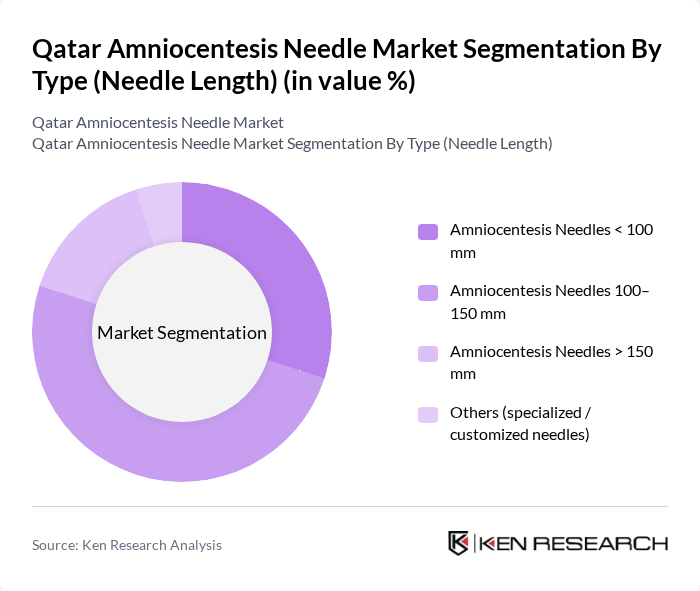
The subsegment of Amniocentesis Needles 100–150 mm dominates the market due to its versatility and suitability for a wide range of ultrasound?guided transabdominal procedures, including routine diagnostic amniocentesis in the second trimester. This needle length is preferred by healthcare professionals for its balance between ease of use, adequate reach to the amniotic cavity in different maternal habitus, and effectiveness in obtaining amniotic fluid samples while minimizing maternal and fetal trauma. The growing trend towards image?guided, minimally invasive prenatal procedures and the increasing number of prenatal tests being conducted in high?risk pregnancies have further solidified the demand for this specific needle length, making it the leading choice among practitioners.
By Procedure:The market is also segmented based on the type of procedure performed using amniocentesis needles. The subsegments include Diagnostic Amniocentesis, Therapeutic Amnioreduction, Fetal Blood Sampling / Cordocentesis, and Amnioinfusion and Others. Each procedure has distinct requirements and applications, influencing the choice of needle type and length, consistent with global utilization patterns where diagnostic amniocentesis remains the predominant indication followed by procedures such as amnioreduction and cordocentesis.
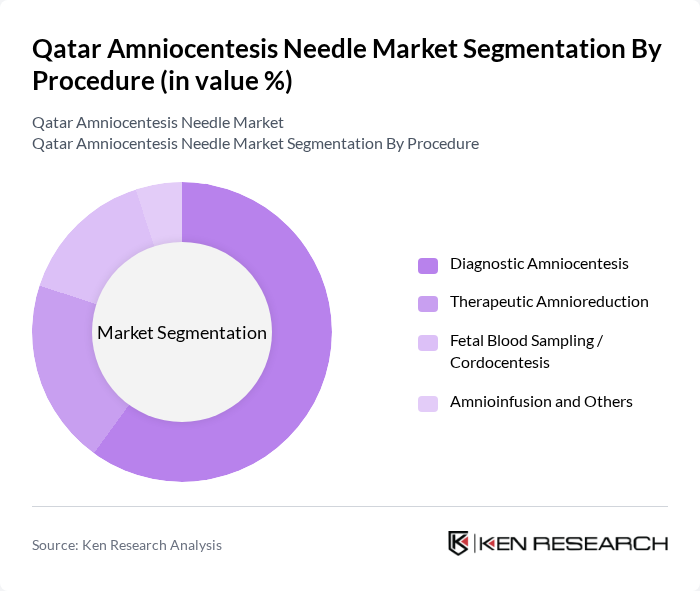
Diagnostic Amniocentesis is the leading procedure in the market, accounting for a significant share due to its critical role in prenatal diagnosis of genetic disorders and chromosomal abnormalities such as trisomy 21, trisomy 18, and neural tube defects. The increasing awareness and acceptance of prenatal testing among expectant mothers, particularly those in advanced maternal age or with high?risk pregnancies, have driven the demand for this procedure even alongside the uptake of non?invasive prenatal testing, which often serves as a screening step before confirmatory invasive diagnostics. Additionally, advancements in ultrasound imaging, improved needle design (such as thinner gauges and enhanced echogenicity), and standardized procedural protocols have further contributed to its dominance in the market by improving safety profiles and procedural success rates.
The Qatar Amniocentesis Needle Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Becton, Dickinson and Company (BD), Cook Medical LLC, Vitrolife AB, RI.MOS. Srl, CooperSurgical, Inc., Smiths Medical (ICU Medical, Inc.), Rocket Medical PLC, Laboratoire C.C.D., Cardinal Health, Inc., Teleflex Incorporated, Hologic, Inc., GE HealthCare Technologies Inc., Siemens Healthineers AG, FUJIFILM Holdings Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Qatar amniocentesis needle market appears promising, driven by ongoing advancements in medical technology and increasing public awareness of prenatal health. As healthcare infrastructure continues to expand, particularly in rural areas, access to amniocentesis is expected to improve. Additionally, the integration of artificial intelligence in prenatal diagnostics is likely to enhance accuracy and efficiency, further encouraging adoption. These trends suggest a robust growth trajectory for the market in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type (Needle Length) | Amniocentesis Needles < 100 mm Amniocentesis Needles 100–150 mm Amniocentesis Needles > 150 mm Others (specialized / customized needles) |
| By Procedure | Diagnostic Amniocentesis Therapeutic Amnioreduction Fetal Blood Sampling / Cordocentesis Amnioinfusion and Others |
| By Application | Chromosomal and Genetic Disorder Testing Fetal Lung Maturity and Infection Assessment Fetal Health Monitoring in High?risk Pregnancies Others |
| By End-User | Public Hospitals and Women’s Health Centers Private Hospitals and Specialty Clinics Diagnostic Laboratories and IVF Centers Academic & Research Institutions |
| By Distribution Channel | Direct Tenders to Hospitals and Government Bodies Local Medical Device Distributors Group Purchasing Organizations (GPOs) Online and Other Channels |
| By Patient Demographics | Maternal Age (< 30 years, 30–35 years, > 35 years) Pregnancy Risk Profile (High?risk vs Low?risk) National vs Expatriate Patients |
| By Technology and Guidance Used | Ultrasound?Guided Amniocentesis Freehand / Landmark?Guided Amniocentesis Image?guided Advanced Techniques (e.g., 3D/4D ultrasound?assisted) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Obstetricians and Gynecologists | 60 | Healthcare Providers, Specialists in Prenatal Care |
| Hospital Administrators | 50 | Healthcare Facility Managers, Operations Directors |
| Expectant Parents | 120 | Pregnant Women, Couples Considering Amniocentesis |
| Healthcare Policy Makers | 40 | Government Officials, Health Program Directors |
| Medical Equipment Suppliers | 40 | Sales Representatives, Product Managers |
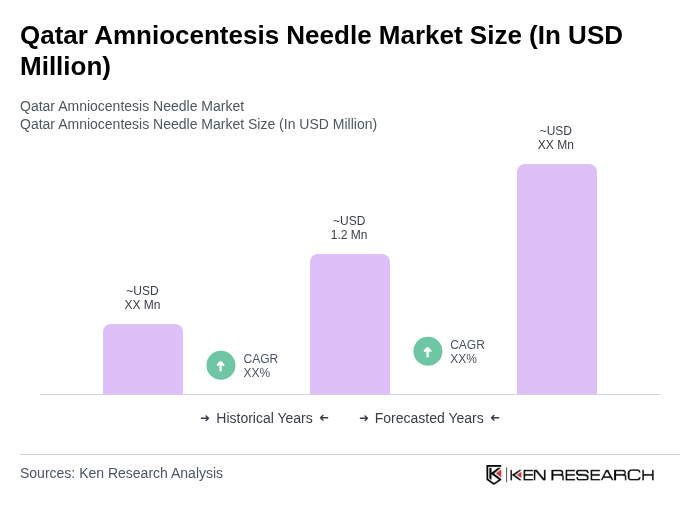
The Qatar Amniocentesis Needle Market is valued at approximately USD 1.2 million, contributing to the broader Middle East and Africa market, which generates around USD 21 million in revenue.