Region:Middle East
Author(s):Geetanshi
Product Code:KRAB1654
Pages:91
Published On:January 2026
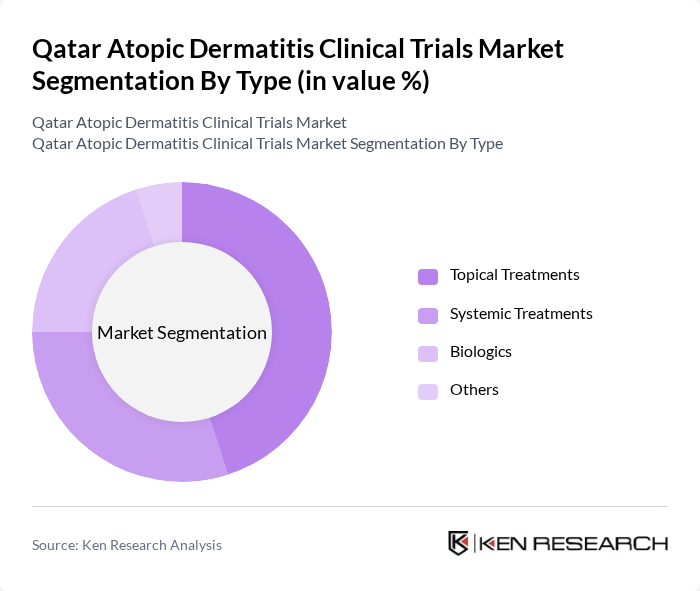
By Type:The market is segmented into various treatment types, including topical treatments, systemic treatments, biologics, and others. Among these, topical treatments are currently leading the market due to their widespread use and effectiveness in managing mild to moderate atopic dermatitis. Systemic treatments and biologics are gaining traction as they offer advanced therapeutic options for severe cases, reflecting a shift in treatment paradigms.
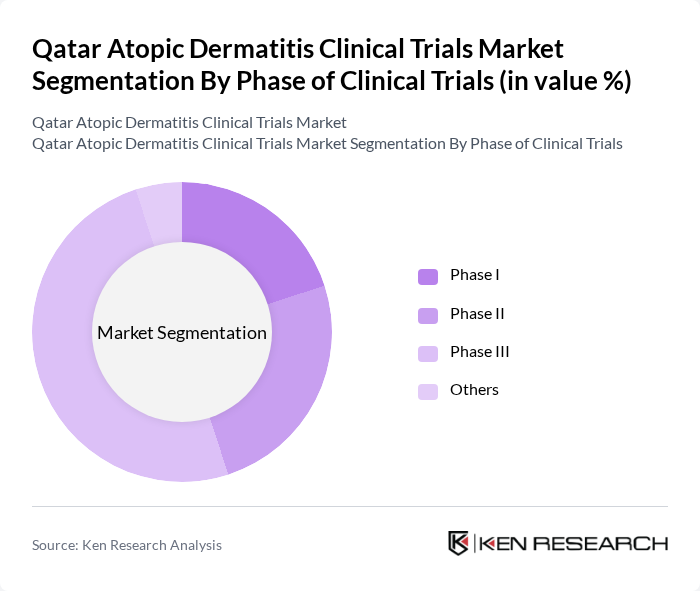
By Phase of Clinical Trials:The clinical trials are categorized into Phase I, Phase II, Phase III, and others. Phase III trials dominate the market as they are crucial for determining the efficacy and safety of new treatments before they receive regulatory approval. The increasing number of participants in these trials reflects the growing interest in developing new therapies for atopic dermatitis.
The Qatar Atopic Dermatitis Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Qatar Biomedical Research Institute, Hamad Medical Corporation, Qatar University, Doha Healthcare, Al Ahli Hospital, Sidra Medicine, Qatar Pharma, Gulf Medical University, Qatar Clinical Research Center, Qatar Foundation, Weill Cornell Medicine-Qatar, Qatar University of Science and Technology, Qatar Medical Center, Al Emadi Hospital, Dallah Hospital contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Qatar atopic dermatitis clinical trials market appears promising, driven by increasing healthcare investments and a growing focus on personalized medicine. As the prevalence of atopic dermatitis continues to rise, innovative trial designs and digital health technologies are expected to enhance patient engagement and streamline processes. Furthermore, collaborations between pharmaceutical companies and academic institutions will likely foster the development of novel therapies, addressing unmet medical needs and improving patient outcomes in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Topical Treatments Systemic Treatments Biologics Others |
| By Phase of Clinical Trials | Phase I Phase II Phase III Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Geography | Doha Al Rayyan Umm Salal Others |
| By Clinical Trial Design | Randomized Controlled Trials Observational Studies Adaptive Trials Others |
| By Funding Source | Government Funding Private Sector Funding Non-Profit Organizations Others |
| By Duration of Trials | Short-term Trials Long-term Trials Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Dermatology Clinics | 100 | Dermatologists, Clinic Managers |
| Pharmaceutical Companies | 80 | Product Managers, Clinical Research Associates |
| Patient Advocacy Groups | 60 | Patient Representatives, Healthcare Advocates |
| Healthcare Policy Makers | 50 | Health Economists, Policy Analysts |
| Clinical Research Organizations | 70 | Clinical Trial Managers, Regulatory Affairs Specialists |
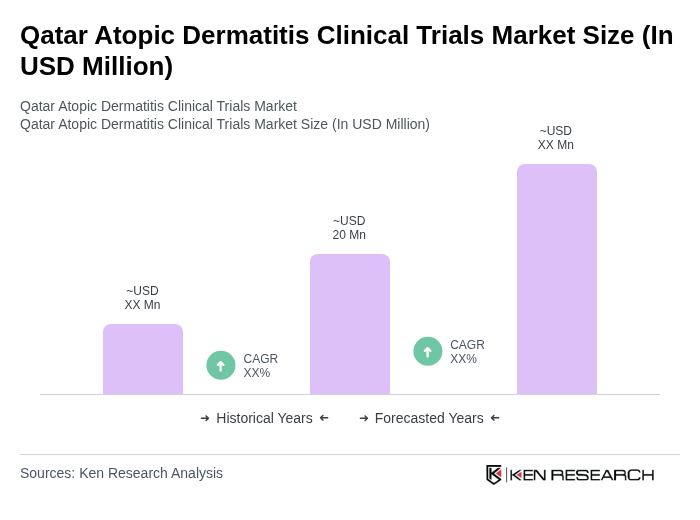
The Qatar Atopic Dermatitis Clinical Trials Market is valued at approximately USD 140 million, reflecting a significant increase driven by the rising prevalence of atopic dermatitis and advancements in clinical research methodologies.