About the Report
Base Year 2024Qatar Bioburden Testing Market Overview
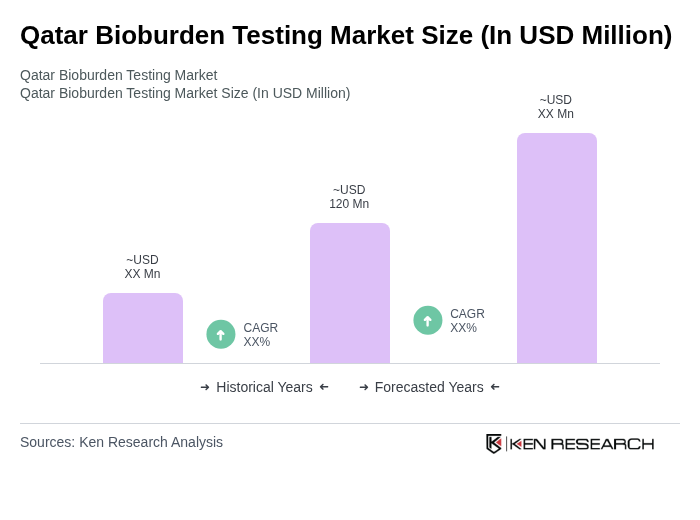
- The Qatar Bioburden Testing Market is valued at USD 120 million, based on a five-year historical analysis. Growth is driven by rising demand for diagnostic accuracy in healthcare, expanding laboratory infrastructure, and increased prevalence of chronic diseases, which together underpin the need for robust microbial contamination testing in medical and pharmaceutical contexts.
- Key players in this market include diagnostic service providers and laboratory equipment suppliers who support quality control and compliance with testing standards in the broader diagnostics ecosystem. Healthcare institutions and diagnostic laboratories in Doha serve as hubs for testing services due to advanced healthcare infrastructure and research capabilities.
- A key policy influencing this environment is the Clinical Laboratories Licensing and Accreditation Regulation, 2023, which mandates proficiency testing participation for all clinical laboratories to ensure test reliability, indirectly reinforcing demand for bioburden testing services.
Qatar Bioburden Testing Market Segmentation
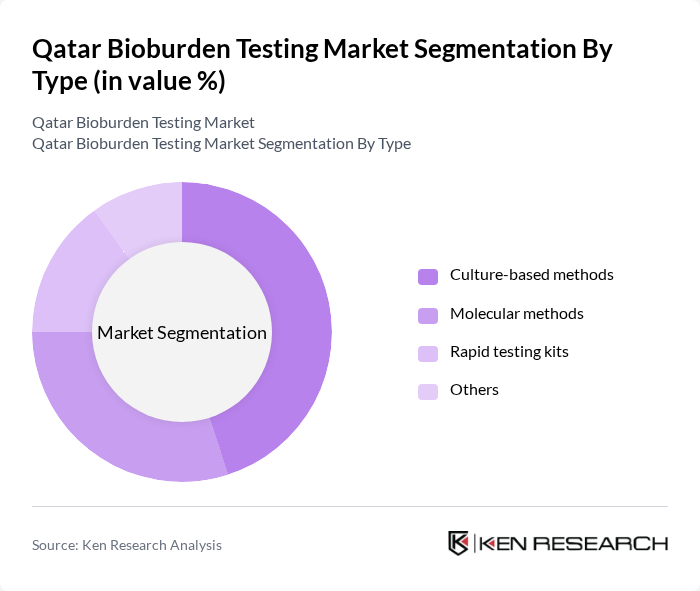
By Type:The bioburden testing market is segmented into various types, including culture-based methods, molecular methods, rapid testing kits, and others. Among these, culture-based methods dominate the market due to their reliability and established protocols in microbial testing. The preference for these methods is driven by their ability to provide accurate results over a longer incubation period, which is crucial for industries like pharmaceuticals and biotechnology. Molecular methods are gaining traction due to their speed and sensitivity, but culture-based methods remain the gold standard.
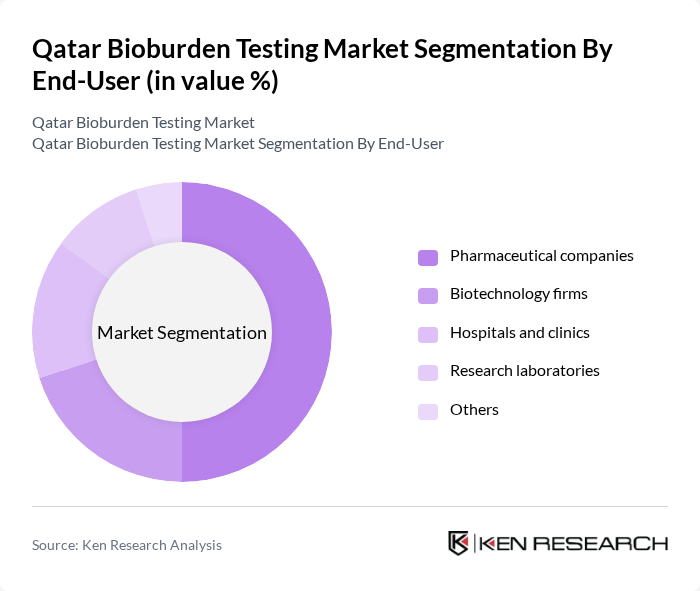
By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, hospitals and clinics, research laboratories, and others. Pharmaceutical companies are the leading end-users, primarily due to stringent regulatory requirements for sterility and quality control in drug manufacturing. The increasing focus on biopharmaceuticals and personalized medicine is further driving the demand for bioburden testing in this sector. Hospitals and clinics also contribute significantly, as they require reliable testing for infection control and patient safety.
Qatar Bioburden Testing Market Competitive Landscape
The Qatar Bioburden Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as BioMerieux, Thermo Fisher Scientific, Charles River Laboratories, Merck KGaA, Becton, Dickinson and Company, 3M Company, Sartorius AG, EMD Millipore, Pall Corporation, Nelson Labs, SGS SA, Intertek Group plc, Eurofins Scientific, Labcorp, Q2 Solutions contribute to innovation, geographic expansion, and service delivery in this space.
Qatar Bioburden Testing Market Industry Analysis
Growth Drivers
- High Healthcare Spending per Capita:In future, Qatar's healthcare spending per capita reached USD 2,000, reflecting a strong commitment to health services. This elevated expenditure indicates a robust demand for advanced laboratory services, including bioburden testing, as healthcare facilities strive to meet stringent quality standards. The increasing focus on patient safety and effective sterilization processes further drives the need for reliable testing solutions in the healthcare sector.
- Strong Macroeconomic Output and Income Levels:Qatar's GDP in future was approximately USD 250 billion, with a GDP per capita of around USD 85,000. This significant economic output and high income levels create a favorable environment for investment in quality control testing services across various sectors, including healthcare and pharmaceuticals. The wealth generated supports the adoption of advanced bioburden testing technologies, enhancing product safety and compliance.
- Comprehensive Digital Infrastructure:As of early future, Qatar had 3.00 million internet users, representing a 100% penetration rate, and 5.00 million mobile connections, equating to 180% of the population. This advanced digital infrastructure facilitates efficient laboratory information systems and data transfer, enabling the adoption of digital-biotech platforms. Such capabilities are essential for enhancing bioburden testing operations, improving accuracy, and streamlining processes in laboratories.
Market Challenges
- Economic Dependence on Hydrocarbons and Fiscal Volatility:Qatar's real GDP growth is projected to be modest at 2.0% in future, with a central government fiscal surplus narrowing to 0.5% of GDP. This economic landscape, characterized by low growth and limited fiscal buffers, poses challenges for public and private sector investments in specialized testing services, including bioburden testing. Such fiscal constraints may hinder the expansion of necessary laboratory infrastructure.
- High Costs and Skilled Labor Shortage in Tech Infrastructure:The growing data center and AI infrastructure in Qatar face significant challenges, including high implementation costs and a shortage of skilled professionals. Bioburden testing providers require qualified lab technologists and digital systems experts to operate effectively. The scarcity of skilled labor in these areas can impede the scaling of bioburden testing services, limiting market growth potential.
Qatar Bioburden Testing Market Future Outlook
The future of the Qatar bioburden testing market appears promising, driven by trends such as digital transformation and the integration of AI technologies in laboratory operations. As healthcare facilities increasingly adopt automated testing solutions, the demand for efficient and accurate bioburden testing will rise. Additionally, public-private collaborations under the Third National Development Strategy (NDS3) are expected to enhance investment in healthcare R&D, fostering innovation and growth in the bioburden testing sector.
Market Opportunities
- Expansion in Healthcare Testing Segment:With healthcare spending per capita at USD 2,000 in future, there is significant potential to develop bioburden testing as part of expanded healthcare offerings. This growth can help meet the increasing quality standards required in pharmaceuticals and hospitals, ensuring patient safety and compliance with regulatory requirements.
- Leveraging Digital Infrastructure for Remote and Automated Testing:Qatar's extensive digital infrastructure, with nearly the entire population online and mobile-connected, presents an opportunity to deploy remote testing services. The planned expansion of data centers can facilitate automated lab operations, enhancing efficiency and accessibility in bioburden testing services across the region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Culture-based methods Molecular methods Rapid testing kits Others |
| By End-User | Pharmaceutical companies Biotechnology firms Hospitals and clinics Research laboratories Others |
| By Application | Sterility testing Environmental monitoring Quality control in manufacturing Others |
| By Region | Doha Al Rayyan Umm Salal Others |
| By Technology | Automated systems Manual testing methods Hybrid approaches Others |
| By Investment Source | Private sector investments Government funding International collaborations Others |
| By Policy Support | Government subsidies for testing facilities Tax incentives for R&D Grants for technology adoption Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Qatar Ministry of Public Health, Qatar Food Safety Department)
Pharmaceutical Manufacturers
Medical Device Manufacturers
Biotechnology Companies
Quality Control Laboratories
Healthcare Providers and Hospitals
Industry Associations (e.g., Qatar Chamber of Commerce)
Players Mentioned in the Report:
BioMerieux
Thermo Fisher Scientific
Charles River Laboratories
Merck KGaA
Becton, Dickinson and Company
3M Company
Sartorius AG
EMD Millipore
Pall Corporation
Nelson Labs
SGS SA
Intertek Group plc
Eurofins Scientific
Labcorp
Q2 Solutions
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Qatar Bioburden Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Qatar Bioburden Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Qatar Bioburden Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for sterilization in healthcare
3.1.2 Rising awareness of bioburden testing in pharmaceuticals
3.1.3 Stringent regulatory requirements
3.1.4 Technological advancements in testing methods
3.2 Market Challenges
3.2.1 High costs associated with testing equipment
3.2.2 Limited availability of skilled professionals
3.2.3 Variability in regulatory standards
3.2.4 Competition from alternative testing methods
3.3 Market Opportunities
3.3.1 Expansion of the pharmaceutical industry in Qatar
3.3.2 Growth in the biotechnology sector
3.3.3 Increasing investments in healthcare infrastructure
3.3.4 Collaboration opportunities with research institutions
3.4 Market Trends
3.4.1 Shift towards automated testing solutions
3.4.2 Integration of digital technologies in testing
3.4.3 Focus on sustainability in testing processes
3.4.4 Rising demand for rapid testing methods
3.5 Government Regulation
3.5.1 Implementation of GMP (Good Manufacturing Practices)
3.5.2 Compliance with ISO standards
3.5.3 Regulatory oversight by the Qatar Ministry of Public Health
3.5.4 Guidelines for bioburden testing in medical devices
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Qatar Bioburden Testing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Qatar Bioburden Testing Market Segmentation
8.1 By Type
8.1.1 Culture-based methods
8.1.2 Molecular methods
8.1.3 Rapid testing kits
8.1.4 Others
8.2 By End-User
8.2.1 Pharmaceutical companies
8.2.2 Biotechnology firms
8.2.3 Hospitals and clinics
8.2.4 Research laboratories
8.2.5 Others
8.3 By Application
8.3.1 Sterility testing
8.3.2 Environmental monitoring
8.3.3 Quality control in manufacturing
8.3.4 Others
8.4 By Region
8.4.1 Doha
8.4.2 Al Rayyan
8.4.3 Umm Salal
8.4.4 Others
8.5 By Technology
8.5.1 Automated systems
8.5.2 Manual testing methods
8.5.3 Hybrid approaches
8.5.4 Others
8.6 By Investment Source
8.6.1 Private sector investments
8.6.2 Government funding
8.6.3 International collaborations
8.6.4 Others
8.7 By Policy Support
8.7.1 Government subsidies for testing facilities
8.7.2 Tax incentives for R&D
8.7.3 Grants for technology adoption
8.7.4 Others
9. Qatar Bioburden Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Innovation Rate
9.2.8 Operational Efficiency
9.2.9 Customer Satisfaction Score
9.2.10 Market Share Percentage
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 BioMerieux
9.5.2 Thermo Fisher Scientific
9.5.3 Charles River Laboratories
9.5.4 Merck KGaA
9.5.5 Becton, Dickinson and Company
9.5.6 3M Company
9.5.7 Sartorius AG
9.5.8 EMD Millipore
9.5.9 Pall Corporation
9.5.10 Nelson Labs
9.5.11 SGS SA
9.5.12 Intertek Group plc
9.5.13 Eurofins Scientific
9.5.14 Labcorp
9.5.15 Q2 Solutions
10. Qatar Bioburden Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for bioburden testing
10.1.2 Decision-making processes
10.1.3 Preferred suppliers and vendors
10.1.4 Compliance requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in testing facilities
10.2.2 Spending on technology upgrades
10.2.3 Budget for training and development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in meeting regulatory standards
10.3.2 Issues with testing accuracy
10.3.3 Delays in testing processes
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of bioburden testing benefits
10.4.2 Training needs for staff
10.4.3 Infrastructure readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of testing effectiveness
10.5.2 Expansion into new applications
10.5.3 Long-term cost savings
10.5.4 Others
11. Qatar Bioburden Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps and opportunities
1.2 Value proposition development
1.3 Revenue model identification
1.4 Customer segmentation
1.5 Key partnerships
1.6 Cost structure analysis
1.7 Competitive advantage
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target market identification
2.4 Communication strategies
2.5 Digital marketing approaches
2.6 Customer engagement tactics
2.7 Performance metrics
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales approaches
3.5 Partnership with local distributors
3.6 Logistics and supply chain management
3.7 Performance evaluation
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Value-based pricing models
4.6 Discount strategies
4.7 Pricing optimization techniques
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments analysis
5.3 Emerging trends identification
5.4 Product development opportunities
5.5 Customer feedback mechanisms
5.6 Market research insights
5.7 Future demand forecasting
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer support strategies
6.4 Feedback collection methods
6.5 Relationship management tools
6.6 Community engagement initiatives
6.7 Performance tracking
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling points
7.4 Customer-centric approaches
7.5 Competitive differentiation
7.6 Value delivery mechanisms
7.7 Performance metrics
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Training and development
8.5 Market research activities
8.6 Performance evaluation
8.7 Continuous improvement processes
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries identification
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
10.6 Strategic fit analysis
10.7 Performance metrics
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Budget allocation
11.4 Resource planning
11.5 Financial projections
11.6 Risk management strategies
11.7 Performance tracking
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnership dynamics
12.3 Risk mitigation strategies
12.4 Performance evaluation
12.5 Long-term sustainability
12.6 Strategic alignment
12.7 Decision-making frameworks
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Financial health indicators
13.4 Revenue growth projections
13.5 Cost management strategies
13.6 Performance metrics
13.7 Market positioning
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Collaboration opportunities
14.6 Performance evaluation
14.7 Risk assessment
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for key activities
15.2.2 Performance tracking
15.2.3 Resource allocation
15.2.4 Risk management
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from local health authorities and biopharmaceutical associations
- Review of scientific literature on bioburden testing methodologies and standards
- Examination of market trends through publications from regulatory bodies such as the Qatar Ministry of Public Health
Primary Research
- Interviews with laboratory managers in hospitals and pharmaceutical companies
- Surveys targeting quality assurance professionals in the bioburden testing sector
- Field visits to testing facilities to gather insights on operational practices
Validation & Triangulation
- Cross-validation of findings with data from industry conferences and workshops
- Triangulation of insights from primary interviews with secondary data sources
- Sanity checks through expert panel discussions with microbiologists and regulatory experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the bioburden testing market size based on healthcare expenditure in Qatar
- Segmentation of the market by end-user applications such as pharmaceuticals, medical devices, and research laboratories
- Incorporation of growth rates from related sectors, including biotechnology and pharmaceuticals
Bottom-up Modeling
- Collection of data on the number of bioburden tests conducted annually across various sectors
- Estimation of average pricing for bioburden testing services and products
- Calculation of market size based on volume of tests multiplied by average service costs
Forecasting & Scenario Analysis
- Development of forecasts using historical growth trends and emerging market dynamics
- Scenario analysis based on potential regulatory changes and technological advancements
- Creation of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing | 100 | Quality Control Managers, Production Supervisors |
| Medical Device Testing | 80 | Regulatory Affairs Specialists, Lab Technicians |
| Research Laboratories | 70 | Research Scientists, Lab Managers |
| Healthcare Facilities | 90 | Infection Control Officers, Microbiologists |
| Biotechnology Firms | 60 | Product Development Managers, Quality Assurance Leads |
Frequently Asked Questions
What is the current value of the Qatar Bioburden Testing Market?
The Qatar Bioburden Testing Market is valued at approximately USD 120 million, reflecting a robust demand for microbial contamination testing driven by the need for diagnostic accuracy in healthcare and the increasing prevalence of chronic diseases.