Region:Middle East
Author(s):Geetanshi
Product Code:KRAC9466
Pages:88
Published On:November 2025
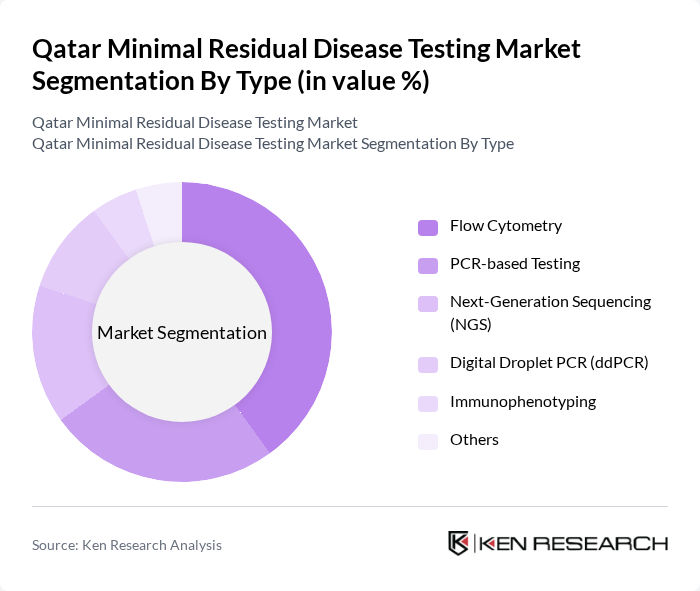
By Type:The market is segmented into various types of testing methods, including PCR-based Testing, Flow Cytometry, Next-Generation Sequencing (NGS), Digital Droplet PCR (ddPCR), Immunophenotyping, and Others. Among these,Flow Cytometryis the most widely used due to its established clinical utility, high throughput, and accessibility in hospital laboratories, making it a preferred choice for detecting minimal residual disease in hematological malignancies.
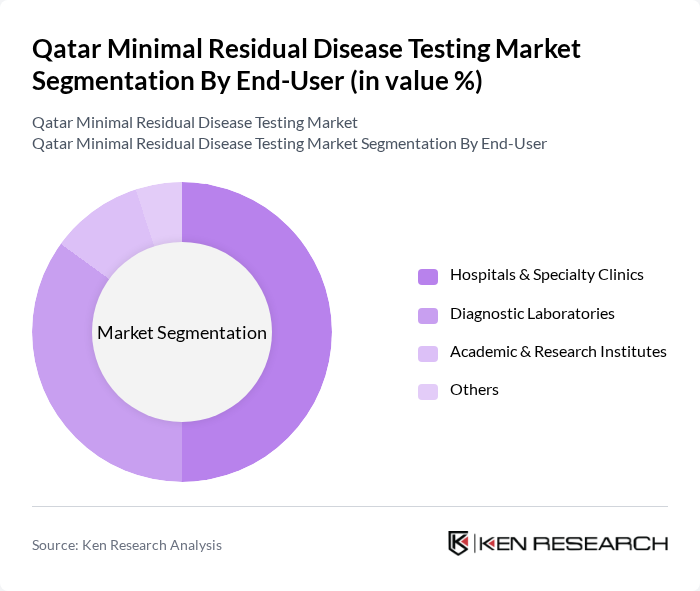
By End-User:The end-user segmentation includes Hospitals & Specialty Clinics, Diagnostic Laboratories, Academic & Research Institutes, and Others.Hospitals & Specialty Clinicsdominate this segment due to their advanced facilities, access to cutting-edge diagnostic technologies, and the central role they play in comprehensive cancer care and clinical research.
The Qatar Minimal Residual Disease Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Qatar Biomedical Research Institute (QBRI), Hamad Medical Corporation, Sidra Medicine, Al Ahli Hospital, Doha Clinic Hospital, Qatar University, Gulf Laboratory and Radiology, MedLabs, Qatar Medical Center, Al Emadi Hospital, Aster DM Healthcare, Dallah Hospital, Qatari Red Crescent Society, Qatar Cancer Society, Weill Cornell Medicine – Qatar, Roche Diagnostics Middle East, Thermo Fisher Scientific (Qatar distributor), QIAGEN (Qatar representative office), Bio-Rad Laboratories (Qatar distributor), Adaptive Biotechnologies (regional presence) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the minimal residual disease testing market in Qatar appears promising, driven by ongoing advancements in technology and increasing healthcare investments. The government is expected to enhance healthcare infrastructure, facilitating better access to diagnostic services. Additionally, the integration of artificial intelligence in diagnostics is anticipated to improve testing accuracy and efficiency. These trends will likely foster a more robust market environment, encouraging innovation and collaboration among healthcare providers and researchers.
| Segment | Sub-Segments |
|---|---|
| By Type | PCR-based Testing Flow Cytometry Next-Generation Sequencing (NGS) Digital Droplet PCR (ddPCR) Immunophenotyping Others |
| By End-User | Hospitals & Specialty Clinics Diagnostic Laboratories Academic & Research Institutes Others |
| By Application | Hematological Malignancies (Leukemia, Lymphoma, Myeloma) Solid Tumor Monitoring Treatment Response Assessment Relapse Detection Clinical Trials Others |
| By Technology | Real-Time PCR Next-Generation Sequencing (NGS) Flow Cytometry Digital PCR Microarray Analysis Others |
| By Sample Type | Blood Samples Bone Marrow Samples Peripheral Blood Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Doha Al Rayyan Umm Salal Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Clinics | 120 | Oncologists, Clinical Researchers |
| Diagnostic Laboratories | 100 | Laboratory Managers, Technicians |
| Patient Advocacy Groups | 80 | Patient Representatives, Caregivers |
| Healthcare Policy Makers | 60 | Health Administrators, Policy Analysts |
| Pharmaceutical Companies | 90 | Product Managers, Market Access Specialists |
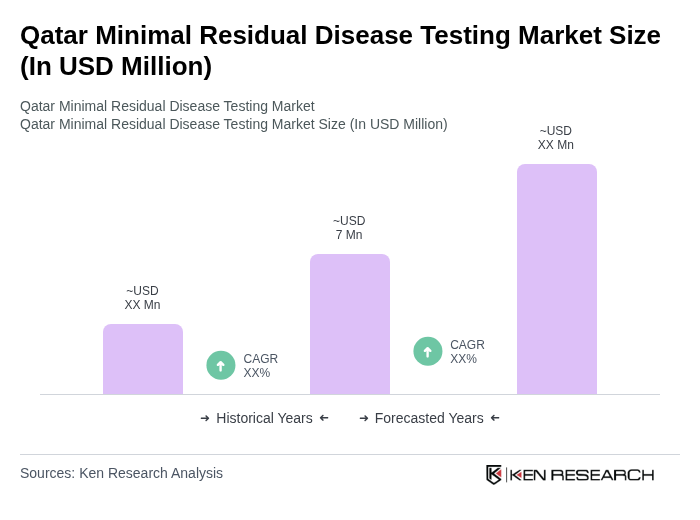
The Qatar Minimal Residual Disease Testing Market is valued at approximately USD 7 million, driven by the rising prevalence of hematological malignancies and advancements in diagnostic technologies such as next-generation sequencing and digital PCR.