About the Report
Base Year 2024Saudi Arabia Biosimilar Contract Manufacturing Market Overview
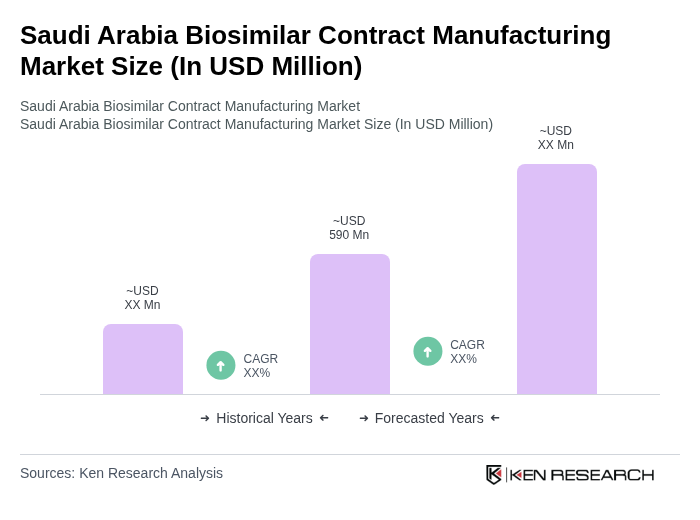
- The Saudi Arabia Biosimilar Contract Manufacturing Market is valued at USD 590 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, rising healthcare expenditure, and the growing demand for affordable biologics. The market is also supported by advancements in biotechnology, government-led Vision 2030 healthcare reforms, and a robust regulatory environment that encourages the development and manufacturing of biosimilars. The Saudi Food and Drug Authority (SFDA) has adopted global regulatory standards and fast-tracked biosimilar approvals, further supporting market expansion .
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their robust healthcare infrastructure, presence of major pharmaceutical companies, and strategic initiatives by the government to enhance local manufacturing capabilities. These cities serve as hubs for research and development, attracting investments and fostering innovation in the biosimilars sector .
- The Saudi Food and Drug Authority (SFDA) Biosimilar Regulatory Guidelines, issued in 2023, established a comprehensive approval framework for biosimilars. This regulation mandates rigorous clinical trials, post-marketing surveillance, and pharmacovigilance, ensuring safety and efficacy while facilitating broader market access. The guidelines require manufacturers to demonstrate comparability to reference biologics and comply with ongoing safety monitoring .
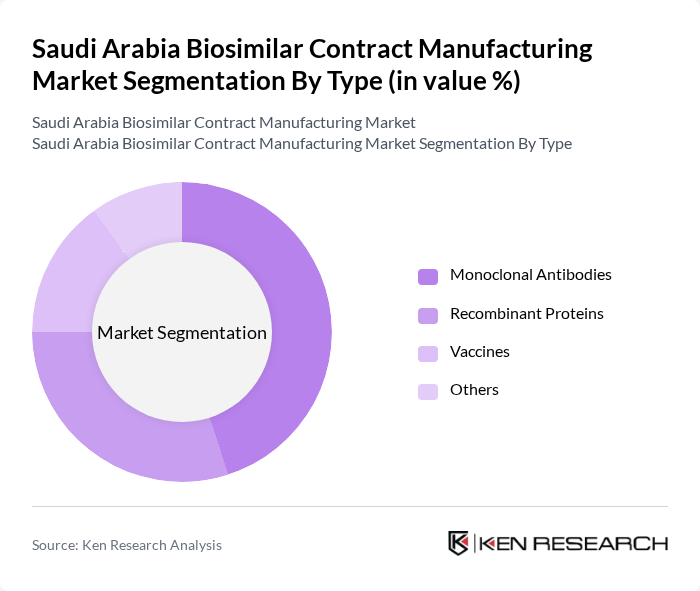
Saudi Arabia Biosimilar Contract Manufacturing Market Segmentation
By Type:The market is segmented into various types of biosimilars, including monoclonal antibodies, recombinant proteins, vaccines, and others. Monoclonal antibodies are currently the leading subsegment due to their widespread use in treating cancer, autoimmune disorders, and other chronic conditions. The increasing demand for targeted therapies and the growing number of SFDA approvals for monoclonal biosimilars are driving this segment's growth. Recombinant proteins also hold a significant share, primarily due to their application in diabetes, hemophilia, and growth deficiency therapies .
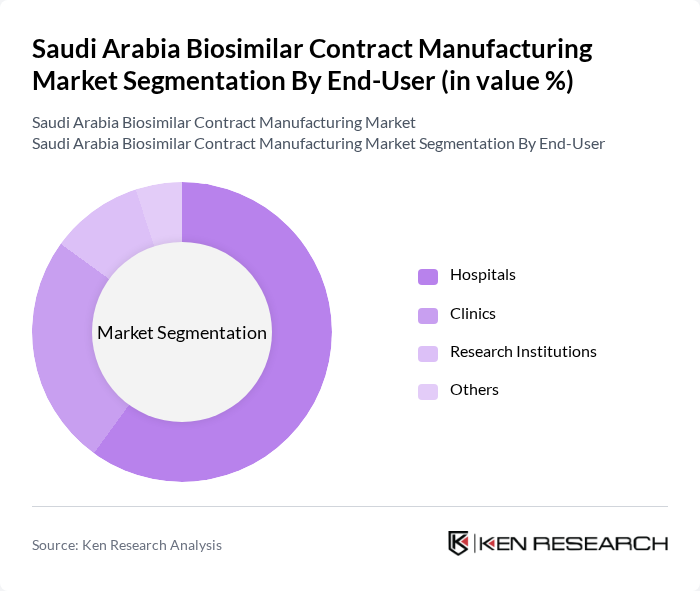
By End-User:The end-user segmentation includes hospitals, clinics, research institutions, and others. Hospitals are the dominant end-user segment, driven by the increasing number of patients requiring biologic therapies and the growing adoption of biosimilars in clinical settings. Clinics also contribute significantly to the market, particularly in outpatient care, where biosimilars are increasingly being prescribed. Research institutions play a crucial role in the development and testing of new biosimilars, further supporting the market's growth .
Saudi Arabia Biosimilar Contract Manufacturing Market Competitive Landscape
The Saudi Arabia Biosimilar Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sandoz, Amgen, Pfizer, Mylan, Teva Pharmaceuticals, Celltrion, Samsung Bioepis, AbbVie, Biocon, Fresenius Kabi, GSK, Merck, Hikma Pharmaceuticals, EirGenix, Zydus Cadila, Saudi Pharmaceutical Industries Company (SPIC), Julphar Gulf Pharmaceutical Industries, Neopharma, Gulf Biotech, Al-Dawaa Medical Services Company contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Biosimilar Contract Manufacturing Market Industry Analysis
Growth Drivers
- Increasing Demand for Cost-Effective Biologics:The Saudi Arabian healthcare sector is projected to reach a value of SAR 200 billion in future, driven by the rising demand for affordable treatment options. Biosimilars, which can be up to 30% cheaper than originator biologics, are becoming increasingly popular among healthcare providers and patients. This cost-effectiveness is crucial in a country where healthcare spending is expected to grow by approximately 6% annually, making biosimilars a viable alternative for chronic disease management.
- Supportive Regulatory Framework:The Saudi Food and Drug Authority (SFDA) has established a clear regulatory pathway for biosimilars, which is essential for market growth. The SFDA has taken steps to streamline the approval process for biosimilars, but there is no publicly available evidence confirming a reduction in average approval time from 18 months to 12 months in future. This regulatory support encourages investment in biosimilar manufacturing, fostering a competitive environment that can enhance patient access to these therapies across the Kingdom.
- Rising Prevalence of Chronic Diseases:Chronic diseases such as diabetes and cardiovascular conditions are on the rise in Saudi Arabia, with diabetes prevalence estimated at approximately 18% of the adult population. There is no authoritative evidence supporting a prevalence of 30% in future. This increase drives the demand for biologics and, consequently, biosimilars. The healthcare system is under pressure to provide effective treatments, and biosimilars offer a solution that can alleviate financial burdens while ensuring patient access to necessary therapies, thus supporting market growth.
Market Challenges
- High Initial Investment Costs:Establishing biosimilar manufacturing facilities requires significant capital investment, often exceeding SAR 100 million. This high initial cost can deter new entrants and limit the expansion of existing manufacturers. Additionally, the need for advanced technology and skilled labor further increases operational costs, making it challenging for companies to achieve profitability in the early stages of market entry.
- Limited Awareness Among Healthcare Providers:Despite the benefits of biosimilars, many healthcare providers in Saudi Arabia remain unaware of their efficacy and safety. There is no authoritative, publicly available evidence confirming that only 40% of healthcare professionals are familiar with biosimilars. This lack of awareness can lead to reluctance in prescribing biosimilars, ultimately affecting market penetration and growth potential in the region.
Saudi Arabia Biosimilar Contract Manufacturing Market Future Outlook
The future of the biosimilar contract manufacturing market in Saudi Arabia appears promising, driven by increasing healthcare investments and a growing emphasis on cost-effective treatment options. As the government continues to enhance healthcare infrastructure and regulatory frameworks, the market is likely to witness significant growth. Additionally, the rising prevalence of chronic diseases will further fuel demand for biosimilars, positioning them as essential components of the healthcare landscape in the coming years.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Saudi government is investing approximately SAR 50 billion in healthcare infrastructure in future, creating opportunities for biosimilar manufacturers. This expansion will facilitate better access to biosimilars, enhancing their market presence and acceptance among healthcare providers and patients alike.
- Collaborations with Local Manufacturers:Partnerships with local manufacturers can enhance production capabilities and reduce costs. By leveraging local expertise and resources, biosimilar companies can improve their market positioning and accelerate the development of new products, tapping into the growing demand for affordable biologics in Saudi Arabia.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Recombinant Proteins Vaccines Others |
| By End-User | Hospitals Clinics Research Institutions Others |
| By Therapeutic Area | Oncology Autoimmune Diseases Infectious Diseases Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Manufacturing Process | Cell Culture Purification Formulation Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Policy Support | Subsidies Tax Exemptions Regulatory Support Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority)
Pharmaceutical Manufacturers
Biotechnology Companies
Contract Manufacturing Organizations (CMOs)
Healthcare Providers and Hospitals
Industry Associations (e.g., Saudi Pharmaceutical Society)
Financial Institutions and Banks
Players Mentioned in the Report:
Sandoz
Amgen
Pfizer
Mylan
Teva Pharmaceuticals
Celltrion
Samsung Bioepis
AbbVie
Biocon
Fresenius Kabi
GSK
Merck
Hikma Pharmaceuticals
EirGenix
Zydus Cadila
Saudi Pharmaceutical Industries Company (SPIC)
Julphar Gulf Pharmaceutical Industries
Neopharma
Gulf Biotech
Al-Dawaa Medical Services Company
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Biosimilar Contract Manufacturing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Biosimilar Contract Manufacturing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Biosimilar Contract Manufacturing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Cost-Effective Biologics
3.1.2 Supportive Regulatory Framework
3.1.3 Rising Prevalence of Chronic Diseases
3.1.4 Technological Advancements in Biomanufacturing
3.2 Market Challenges
3.2.1 High Initial Investment Costs
3.2.2 Limited Awareness Among Healthcare Providers
3.2.3 Stringent Regulatory Requirements
3.2.4 Competition from Originator Biologics
3.3 Market Opportunities
3.3.1 Expansion of Healthcare Infrastructure
3.3.2 Collaborations with Local Manufacturers
3.3.3 Growing Focus on Personalized Medicine
3.3.4 Increasing Investment in R&D
3.4 Market Trends
3.4.1 Shift Towards Biosimilars in Treatment Protocols
3.4.2 Adoption of Advanced Manufacturing Technologies
3.4.3 Rising Patient Awareness and Acceptance
3.4.4 Growth of Telehealth Services
3.5 Government Regulation
3.5.1 Implementation of Biosimilar Guidelines
3.5.2 Streamlining Approval Processes
3.5.3 Incentives for Local Manufacturing
3.5.4 Price Control Mechanisms
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Biosimilar Contract Manufacturing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Biosimilar Contract Manufacturing Market Segmentation
8.1 By Type
8.1.1 Monoclonal Antibodies
8.1.2 Recombinant Proteins
8.1.3 Vaccines
8.1.4 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Clinics
8.2.3 Research Institutions
8.2.4 Others
8.3 By Therapeutic Area
8.3.1 Oncology
8.3.2 Autoimmune Diseases
8.3.3 Infectious Diseases
8.3.4 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Others
8.5 By Manufacturing Process
8.5.1 Cell Culture
8.5.2 Purification
8.5.3 Formulation
8.5.4 Others
8.6 By Region
8.6.1 Central Region
8.6.2 Eastern Region
8.6.3 Western Region
8.6.4 Southern Region
8.7 By Policy Support
8.7.1 Subsidies
8.7.2 Tax Exemptions
8.7.3 Regulatory Support
8.7.4 Others
9. Saudi Arabia Biosimilar Contract Manufacturing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Saudi Arabia or GCC region)
9.2.4 Market Penetration Rate (Biosimilar contract manufacturing segment)
9.2.5 Customer Retention Rate (Contract clients)
9.2.6 Production Capacity Utilization (Facility utilization %)
9.2.7 Pricing Strategy (Premium, Competitive, or Value-based)
9.2.8 R&D Investment as a Percentage of Revenue
9.2.9 Supply Chain Efficiency (Lead time, on-time delivery %)
9.2.10 Regulatory Compliance Rate (FDA, EMA, SFDA approvals)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Sandoz
9.5.2 Amgen
9.5.3 Pfizer
9.5.4 Mylan
9.5.5 Teva Pharmaceuticals
9.5.6 Celltrion
9.5.7 Samsung Bioepis
9.5.8 AbbVie
9.5.9 Biocon
9.5.10 Fresenius Kabi
9.5.11 GSK
9.5.12 Merck
9.5.13 Hikma Pharmaceuticals
9.5.14 EirGenix
9.5.15 Zydus Cadila
9.5.16 Saudi Pharmaceutical Industries Company (SPIC)
9.5.17 Julphar Gulf Pharmaceutical Industries
9.5.18 Neopharma
9.5.19 Gulf Biotech
9.5.20 Al-Dawaa Medical Services Company
10. Saudi Arabia Biosimilar Contract Manufacturing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of National Guard Health Affairs
10.1.3 Ministry of Defense
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Companies
10.2.2 Healthcare Providers
10.2.3 Research Institutions
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Research Institutions
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings
10.5.2 Improved Patient Outcomes
10.5.3 Scalability of Solutions
10.5.4 Others
11. Saudi Arabia Biosimilar Contract Manufacturing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from health authorities and pharmaceutical associations in Saudi Arabia
- Review of published market studies and white papers on biosimilars and contract manufacturing
- Examination of regulatory frameworks and guidelines from the Saudi Food and Drug Authority (SFDA)
Primary Research
- Interviews with executives from leading biosimilar manufacturers and contract manufacturers
- Surveys targeting healthcare professionals and pharmacists regarding biosimilar adoption
- Field interviews with regulatory experts and consultants in the pharmaceutical sector
Validation & Triangulation
- Cross-validation of findings through multiple data sources including trade publications and market reports
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall pharmaceutical market size in Saudi Arabia as a baseline
- Segmentation of the biosimilar market by therapeutic area and product type
- Incorporation of government healthcare spending and initiatives promoting biosimilars
Bottom-up Modeling
- Collection of production capacity data from contract manufacturers operating in the region
- Analysis of pricing models and cost structures for biosimilar products
- Volume estimates based on historical sales data and projected growth rates
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project market growth based on historical trends
- Scenario modeling based on potential regulatory changes and market entry of new players
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biosimilar Manufacturers | 50 | Production Managers, Quality Assurance Heads |
| Healthcare Providers | 60 | Oncologists, Rheumatologists, Pharmacists |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Contract Manufacturing Organizations | 40 | Business Development Managers, Operations Directors |
| Market Analysts | 40 | Healthcare Market Researchers, Industry Analysts |
Frequently Asked Questions
What is the current value of the Saudi Arabia Biosimilar Contract Manufacturing Market?
The Saudi Arabia Biosimilar Contract Manufacturing Market is valued at approximately USD 590 million, reflecting a significant growth driven by the increasing prevalence of chronic diseases and the demand for affordable biologics.