Region:Middle East
Author(s):Rebecca
Product Code:KRAA9229
Pages:90
Published On:November 2025

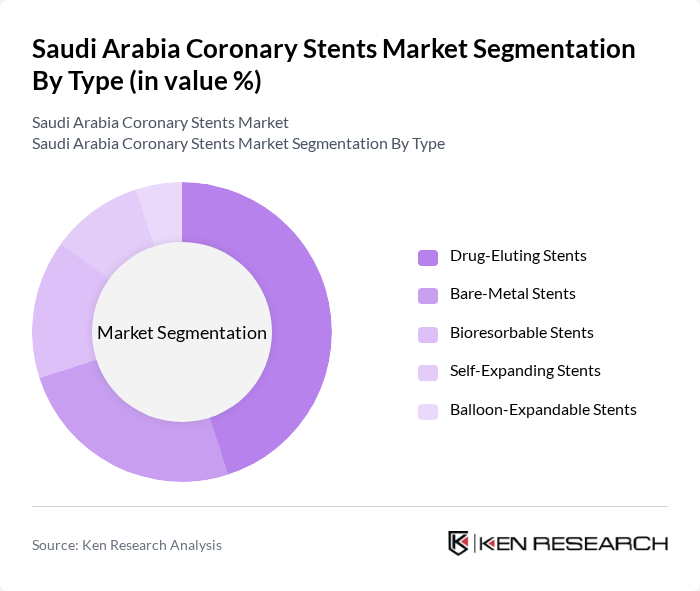
By Type:The market is segmented into various types of coronary stents, including Drug-Eluting Stents, Bare-Metal Stents, Bioresorbable Stents, Self-Expanding Stents, and Balloon-Expandable Stents. Among these, Drug-Eluting Stents are leading the market due to their ability to reduce the risk of restenosis and improve patient outcomes. The increasing preference for minimally invasive procedures and advancements in drug delivery technologies further bolster the demand for this sub-segment.
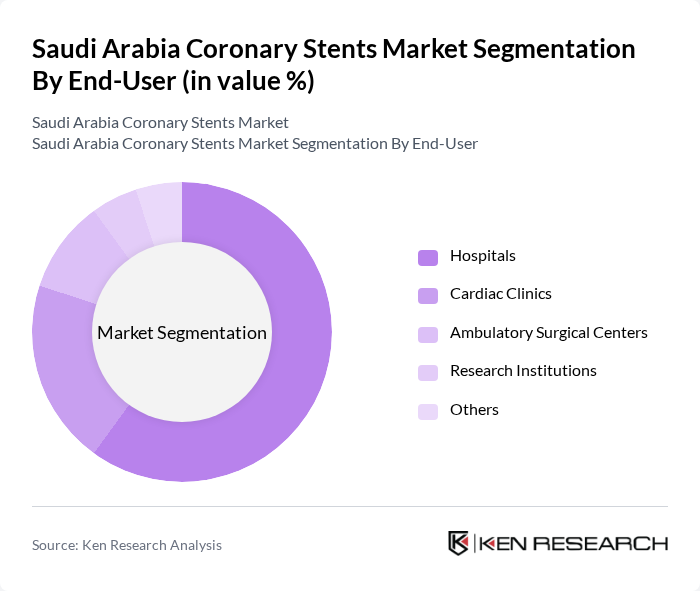
By End-User:The end-user segmentation includes Hospitals, Cardiac Clinics, Ambulatory Surgical Centers, Research Institutions, and Others. Hospitals are the dominant end-user segment, primarily due to their comprehensive cardiac care services and advanced facilities for performing complex procedures. The increasing number of cardiac surgeries and the growing patient population seeking treatment in hospitals contribute significantly to this segment's growth.
The Saudi Arabia Coronary Stents Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Boston Scientific Corporation, Medtronic plc, Johnson & Johnson (Biosense Webster/Cordis), Terumo Corporation, B. Braun Melsungen AG, Cook Medical, Cardinal Health, MicroPort Scientific Corporation, Meril Life Sciences Pvt. Ltd., Al Faisaliah Medical Systems (FMS), Ameco Medical Industries, Biosensors International Group, Ltd., Saham Healthcare, Al-Jeel Medical & Trading Co. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the coronary stents market in Saudi Arabia appears promising, driven by ongoing advancements in medical technology and a supportive regulatory framework. As the healthcare sector continues to evolve, the integration of digital health technologies and personalized medicine will likely enhance patient outcomes. Furthermore, the increasing focus on minimally invasive procedures will drive demand for innovative stent solutions, positioning the market for significant growth in the coming years, particularly as healthcare infrastructure expands.
| Segment | Sub-Segments |
|---|---|
| By Type | Drug-Eluting Stents Bare-Metal Stents Bioresorbable Stents Self-Expanding Stents Balloon-Expandable Stents |
| By End-User | Hospitals Cardiac Clinics Ambulatory Surgical Centers Research Institutions Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Material | Stainless Steel Cobalt Chromium Platinum Chromium Polymer/Biodegradable Materials Others |
| By Procedure Type | Percutaneous Coronary Intervention (PCI) Angioplasty Others |
| By Patient Demographics | Age Group (Adults, Seniors) Gender (Male, Female) Others |
| By Region | Central Region (Riyadh) Eastern Region (Dammam, Khobar) Western Region (Jeddah, Mecca, Medina) Southern Region Northern Region Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologist Insights | 60 | Interventional Cardiologists, Electrophysiologists |
| Hospital Procurement Strategies | 50 | Procurement Managers, Supply Chain Directors |
| Patient Demographics and Preferences | 80 | Patients with coronary artery disease, Caregivers |
| Market Trends and Innovations | 40 | Medical Device Manufacturers, R&D Managers |
| Regulatory and Policy Impact | 40 | Healthcare Policy Makers, Regulatory Affairs Specialists |
The Saudi Arabia Coronary Stents Market is valued at approximately USD 130 million, driven by the rising prevalence of cardiovascular diseases, advancements in stent technology, and increased healthcare expenditure.