About the Report
Base Year 2024Saudi Arabia Epinephrine Market Overview
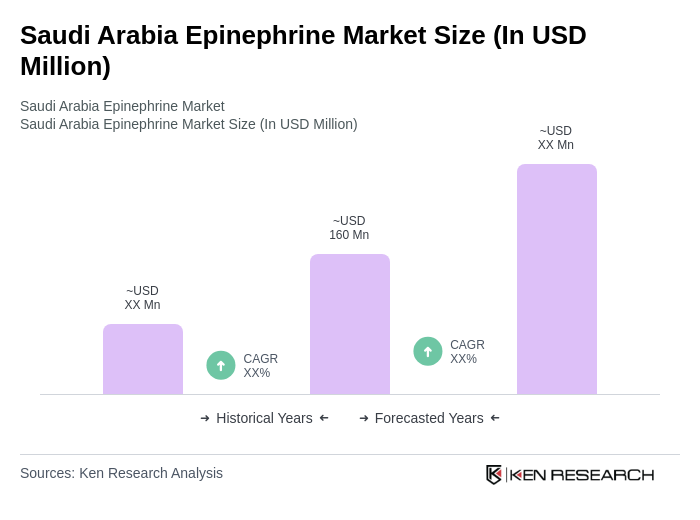
- The Saudi Arabia Epinephrine Market is valued at USD 160 million, based on a five-year historical analysis and proportional allocation from the Middle East & Africa regional market, which was valued at USD 305 million for the most recent period. Growth is driven by the increasing prevalence of allergic reactions and anaphylaxis, rising awareness of immediate treatment options, and advancements in drug delivery systems such as auto-injectors that enhance patient compliance and accessibility. The market is further supported by the adoption of innovative delivery mechanisms and the expansion of emergency preparedness programs in healthcare facilities .
- Key cities dominating the market include Riyadh, Jeddah, and Dammam. These urban centers are characterized by high population density and a well-established healthcare infrastructure, facilitating the distribution and availability of epinephrine products. The presence of major hospitals and clinics in these cities contributes to increased demand for epinephrine in emergency medical situations .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Regulation of Labeling and Packaging of Pharmaceutical Products, 2023” issued by the Saudi Food and Drug Authority, mandating that all epinephrine auto-injectors must include clear usage instructions and expiration dates on their packaging. This regulation aims to enhance patient safety and ensure that individuals at risk of anaphylaxis have access to effective and reliable treatment options .
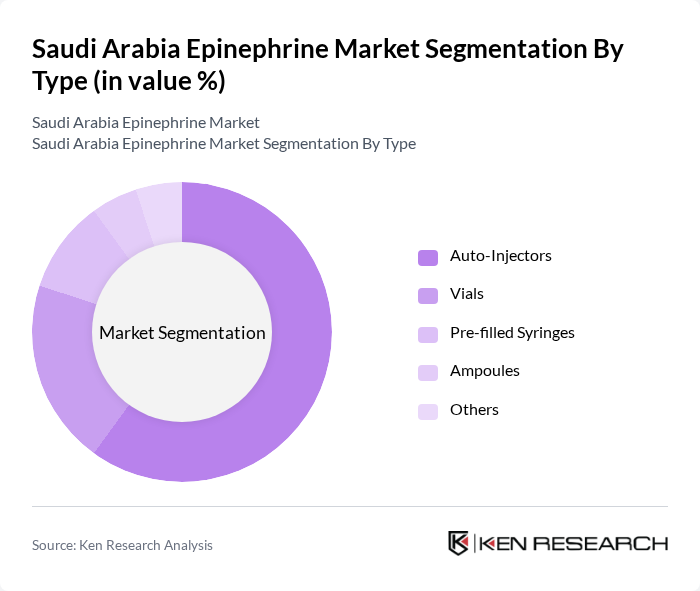
Saudi Arabia Epinephrine Market Segmentation
By Type:The market is segmented into various types, including Auto-Injectors, Vials, Pre-filled Syringes, Ampoules, and Others. Among these, Auto-Injectors are the most dominant due to their ease of use and convenience, particularly for patients who may experience severe allergic reactions. The demand for Vials and Pre-filled Syringes is also significant, especially in hospital settings where healthcare professionals administer the medication. The trend towards self-administration, increased public education on anaphylaxis management, and the integration of epinephrine into emergency kits are driving the preference for Auto-Injectors .
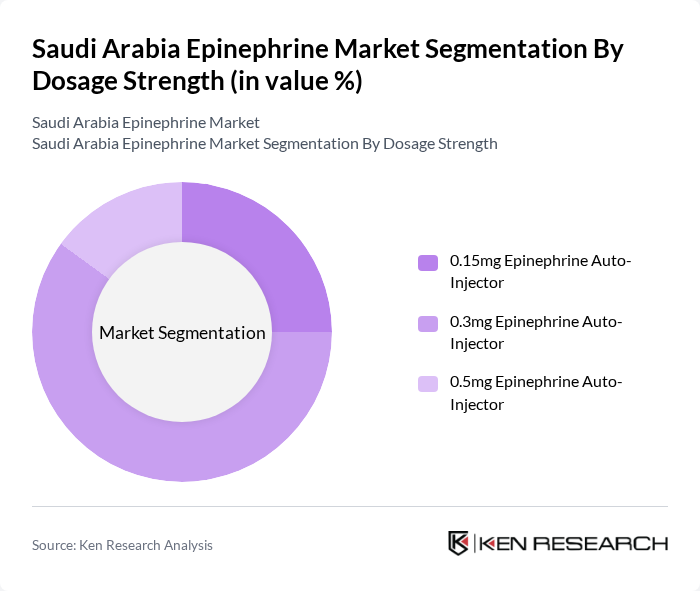
By Dosage Strength:The market is categorized by dosage strength into 0.15mg, 0.3mg, and 0.5mg Epinephrine Auto-Injectors. The 0.3mg dosage is the most widely used, as it is the standard recommended dose for adults and children weighing more than 30 kg. The 0.15mg dosage is gaining traction among pediatric patients, while the 0.5mg dosage is typically reserved for severe cases. The increasing incidence of allergic reactions and the expansion of pediatric allergy diagnosis are driving the demand for these dosage forms .
Saudi Arabia Epinephrine Market Competitive Landscape
The Saudi Arabia Epinephrine Market is characterized by a dynamic mix of regional and international players. Leading participants such as Viatris Inc. (EpiPen), Sanofi S.A., Teva Pharmaceutical Industries Ltd., Kaleo, Inc., Amneal Pharmaceuticals LLC, Pfizer Inc., ALK-Abelló A/S, Adamis Pharmaceuticals Corporation, ARS Pharmaceuticals Inc., Hikma Pharmaceuticals PLC, Sandoz International GmbH, Aurobindo Pharma Ltd., Sun Pharmaceutical Industries Ltd., Bausch Health Companies Inc., Tabuk Pharmaceuticals Manufacturing Co., Jamjoom Pharma, SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation) contribute to innovation, geographic expansion, and service delivery in this space .
Saudi Arabia Epinephrine Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Allergic Reactions:The prevalence of allergic reactions in Saudi Arabia has been rising, with approximately 30% of the population experiencing some form of allergy. According to the Saudi Ministry of Health, around 1.6 million people suffer from severe allergies, necessitating the use of epinephrine auto-injectors. This growing patient base drives demand for epinephrine products, as timely administration is crucial for managing life-threatening allergic reactions, particularly anaphylaxis.
- Rising Awareness about Anaphylaxis:Awareness campaigns led by health authorities have significantly increased public knowledge about anaphylaxis, with a reported 70% of the population now recognizing its symptoms. The Saudi Food and Drug Authority (SFDA) has implemented educational programs in schools and communities, resulting in a 50% increase in the number of individuals trained to use epinephrine auto-injectors. This heightened awareness is crucial for timely intervention, thereby boosting market demand for epinephrine.
- Expansion of Healthcare Infrastructure:Saudi Arabia's healthcare infrastructure is undergoing rapid expansion, with government spending projected to reach SAR 250 billion in future. This investment includes the establishment of new hospitals and clinics, particularly in underserved areas. Enhanced access to healthcare services facilitates the availability of epinephrine auto-injectors, ensuring that patients can receive timely treatment for allergic reactions, thus driving market growth in the region.
Market Challenges
- High Cost of Epinephrine Auto-Injectors:The cost of epinephrine auto-injectors remains a significant barrier to access, with prices ranging from SAR 350 to SAR 600 per device. This high cost limits affordability for many patients, particularly in lower-income segments. The Saudi Ministry of Health has noted that approximately 30% of patients do not have access to these devices due to financial constraints, hindering effective management of allergic reactions.
- Limited Access in Rural Areas:Access to epinephrine auto-injectors is particularly challenging in rural regions of Saudi Arabia, where healthcare facilities are sparse. Reports indicate that only 50% of rural healthcare centers stock epinephrine auto-injectors, leading to delays in treatment during emergencies. This limited availability poses a significant challenge for timely intervention, increasing the risk of severe allergic reactions among rural populations.
Saudi Arabia Epinephrine Market Future Outlook
The future of the Saudi Arabia epinephrine market appears promising, driven by ongoing healthcare reforms and increased public awareness. The government’s commitment to enhancing healthcare access, particularly in rural areas, is expected to improve the distribution of epinephrine auto-injectors. Additionally, the integration of telemedicine for allergy management will facilitate remote consultations, further supporting patient education and timely treatment. These trends indicate a robust growth trajectory for the epinephrine market in the coming years.
Market Opportunities
- Development of New Delivery Systems:Innovations in drug delivery systems, such as needle-free injectors, present significant market opportunities. These advancements can enhance patient compliance and reduce the fear associated with injections, potentially increasing the adoption of epinephrine among patients with severe allergies.
- Collaborations with Healthcare Providers:Strategic partnerships with healthcare providers can enhance distribution channels and improve patient education. Collaborations can lead to more comprehensive training programs for healthcare professionals, ensuring that they are well-equipped to manage anaphylaxis effectively, thereby increasing the overall market penetration of epinephrine products.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Auto-Injectors Vials Pre-filled Syringes Ampoules Others |
| By Dosage Strength | mg Epinephrine Auto-Injector mg Epinephrine Auto-Injector mg Epinephrine Auto-Injector |
| By End-User | Hospitals Clinics Home Care Emergency Services |
| By Distribution Channel | Retail Pharmacies Online Pharmacies Hospital Pharmacies Wholesalers |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Patient Demographics | Children (Under 6 Years) Children (6 to 12 Years) Adolescents & Adults (Over 12 Years) Elderly |
| By Price Range | Low Price Mid Price High Price |
| By Application | Anaphylaxis Treatment Asthma Management Cardiac Arrest Superficial Bleeding Respiratory Disorders Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Pharmaceutical Manufacturers and Producers
Distributors and Wholesalers
Healthcare Providers and Hospitals
Pharmacy Chains and Retail Pharmacies
Medical Device Companies
Health Insurance Companies
Players Mentioned in the Report:
Viatris Inc. (EpiPen)
Sanofi S.A.
Teva Pharmaceutical Industries Ltd.
Kaleo, Inc.
Amneal Pharmaceuticals LLC
Pfizer Inc.
ALK-Abello A/S
Adamis Pharmaceuticals Corporation
ARS Pharmaceuticals Inc.
Hikma Pharmaceuticals PLC
Sandoz International GmbH
Aurobindo Pharma Ltd.
Sun Pharmaceutical Industries Ltd.
Bausch Health Companies Inc.
Tabuk Pharmaceuticals Manufacturing Co.
Jamjoom Pharma
SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Epinephrine Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Epinephrine Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Epinephrine Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of allergic reactions
3.1.2 Rising awareness about anaphylaxis
3.1.3 Expansion of healthcare infrastructure
3.1.4 Government initiatives for emergency medical services
3.2 Market Challenges
3.2.1 High cost of epinephrine auto-injectors
3.2.2 Limited access in rural areas
3.2.3 Regulatory hurdles for new entrants
3.2.4 Competition from alternative treatments
3.3 Market Opportunities
3.3.1 Development of new delivery systems
3.3.2 Increasing demand for personalized medicine
3.3.3 Collaborations with healthcare providers
3.3.4 Expansion into untapped markets
3.4 Market Trends
3.4.1 Growth in telemedicine for allergy management
3.4.2 Shift towards self-administration of epinephrine
3.4.3 Rising focus on patient education
3.4.4 Innovations in drug formulation
3.5 Government Regulation
3.5.1 Mandatory training for epinephrine administration
3.5.2 Regulations on marketing and distribution
3.5.3 Guidelines for emergency response protocols
3.5.4 Policies supporting research and development
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Epinephrine Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Epinephrine Market Segmentation
8.1 By Type
8.1.1 Auto-Injectors
8.1.2 Vials
8.1.3 Pre-filled Syringes
8.1.4 Ampoules
8.1.5 Others
8.2 By Dosage Strength
8.2.1 0.15mg Epinephrine Auto-Injector
8.2.2 0.3mg Epinephrine Auto-Injector
8.2.3 0.5mg Epinephrine Auto-Injector
8.3 By End-User
8.3.1 Hospitals
8.3.2 Clinics
8.3.3 Home Care
8.3.4 Emergency Services
8.4 By Distribution Channel
8.4.1 Retail Pharmacies
8.4.2 Online Pharmacies
8.4.3 Hospital Pharmacies
8.4.4 Wholesalers
8.5 By Region
8.5.1 Central Region
8.5.2 Eastern Region
8.5.3 Western Region
8.5.4 Southern Region
8.6 By Patient Demographics
8.6.1 Children (Under 6 Years)
8.6.2 Children (6 to 12 Years)
8.6.3 Adolescents & Adults (Over 12 Years)
8.6.4 Elderly
8.7 By Price Range
8.7.1 Low Price
8.7.2 Mid Price
8.7.3 High Price
8.8 By Application
8.8.1 Anaphylaxis Treatment
8.8.2 Asthma Management
8.8.3 Cardiac Arrest
8.8.4 Superficial Bleeding
8.8.5 Respiratory Disorders
8.8.6 Others
9. Saudi Arabia Epinephrine Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Saudi Arabia Epinephrine Segment)
9.2.4 Market Penetration Rate (Units Sold/Population at Risk)
9.2.5 Regulatory Approval Status (SFDA, CE, etc.)
9.2.6 Distribution Network Coverage (Number of Pharmacies/Hospitals Served)
9.2.7 Product Portfolio Breadth (Auto-injectors, Vials, Syringes)
9.2.8 Average Selling Price (ASP) per Unit
9.2.9 Local Manufacturing Presence
9.2.10 Brand Recognition Score (Healthcare Professional/Patient Surveys)
9.2.11 Customer Retention Rate
9.2.12 Pricing Strategy (Premium, Value, Tender-based)
9.2.13 Regulatory Compliance Rate
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Viatris Inc. (EpiPen)
9.5.2 Sanofi S.A.
9.5.3 Teva Pharmaceutical Industries Ltd.
9.5.4 Kaleo, Inc.
9.5.5 Amneal Pharmaceuticals LLC
9.5.6 Pfizer Inc.
9.5.7 ALK-Abelló A/S
9.5.8 Adamis Pharmaceuticals Corporation
9.5.9 ARS Pharmaceuticals Inc.
9.5.10 Hikma Pharmaceuticals PLC
9.5.11 Sandoz International GmbH
9.5.12 Aurobindo Pharma Ltd.
9.5.13 Sun Pharmaceutical Industries Ltd.
9.5.14 Bausch Health Companies Inc.
9.5.15 Tabuk Pharmaceuticals Manufacturing Co.
9.5.16 Jamjoom Pharma
9.5.17 SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
10. Saudi Arabia Epinephrine Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Defense
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Emergency Services Funding
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home Care Providers
10.4 User Readiness for Adoption
10.4.1 Training Programs
10.4.2 Awareness Campaigns
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcome Improvements
11. Saudi Arabia Epinephrine Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Timeline
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health authorities and pharmaceutical associations in Saudi Arabia
- Review of published articles and journals focusing on epinephrine usage and market trends
- Examination of government health policies and regulations affecting the epinephrine market
Primary Research
- Interviews with healthcare professionals, including allergists and emergency medicine specialists
- Surveys conducted with pharmacists and hospital procurement managers
- Focus groups with patients who have a history of anaphylaxis to understand their experiences and needs
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and clinical studies
- Triangulation of insights from healthcare professionals, patients, and market data
- Sanity checks through expert panel discussions to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national health expenditure and epinephrine prescriptions
- Segmentation of the market by end-user categories, including hospitals, clinics, and retail pharmacies
- Incorporation of demographic data to assess the prevalence of allergic reactions in the population
Bottom-up Modeling
- Collection of sales data from leading pharmaceutical distributors and manufacturers of epinephrine
- Analysis of pricing strategies and reimbursement policies affecting epinephrine products
- Volume estimates based on historical sales trends and projected growth rates in the healthcare sector
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future demand based on historical data
- Scenario modeling considering factors such as changes in healthcare policies and rising allergy incidences
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Professionals | 100 | Allergists, Emergency Medicine Physicians |
| Pharmacists | 60 | Retail Pharmacists, Hospital Pharmacy Directors |
| Patients with Anaphylaxis | 50 | Individuals with a history of severe allergic reactions |
| Healthcare Administrators | 40 | Hospital Procurement Managers, Health Policy Makers |
| Allergy Support Groups | 40 | Leaders of patient advocacy and support organizations |
Frequently Asked Questions
What is the current value of the Saudi Arabia Epinephrine Market?
The Saudi Arabia Epinephrine Market is valued at approximately USD 160 million, based on a five-year historical analysis and proportional allocation from the broader Middle East & Africa regional market, which is valued at USD 305 million.