Region:Middle East
Author(s):Geetanshi
Product Code:KRAC4432
Pages:94
Published On:October 2025
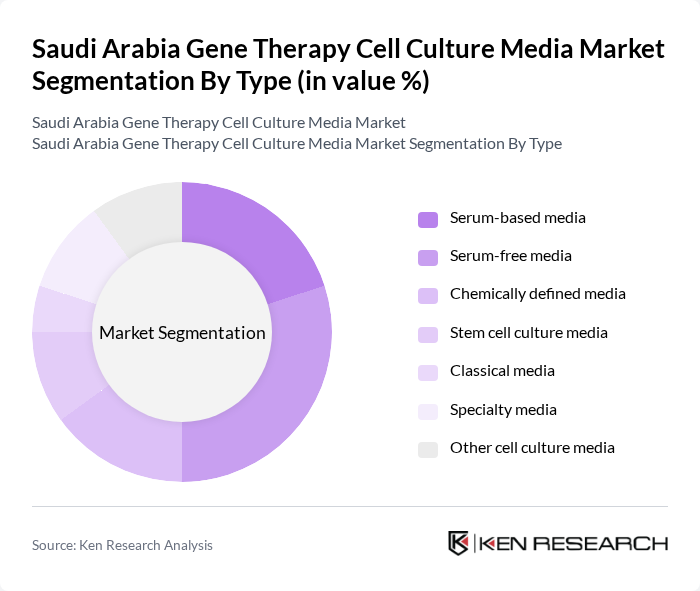
By Type:The market is segmented into various types of cell culture media, including serum-based media, serum-free media, chemically defined media, stem cell culture media, classical media, specialty media, and other cell culture media. Among these, serum-free media is gaining traction due to its ability to support the growth of specific cell types without the variability associated with serum, making it a preferred choice for researchers and biopharmaceutical companies .
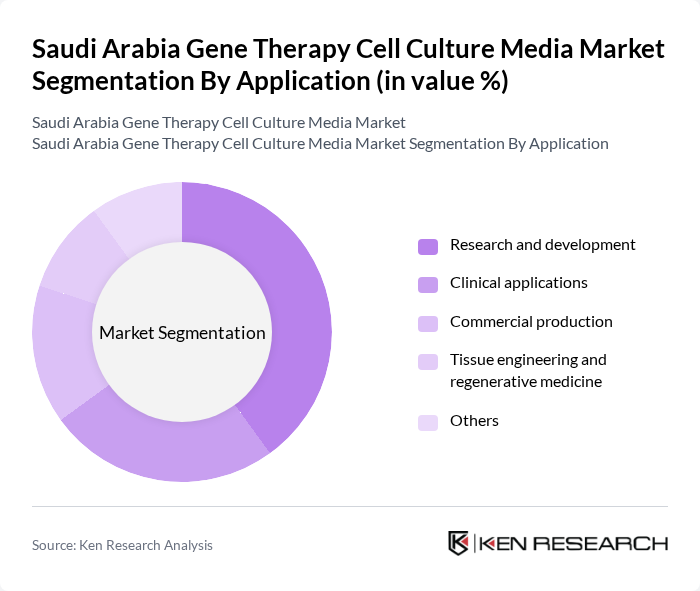
By Application:The applications of cell culture media in the market include research and development, clinical applications, commercial production, tissue engineering and regenerative medicine, and others. The research and development segment is particularly dominant, driven by the increasing focus on innovative therapies and the need for advanced research methodologies in biotechnology and pharmaceuticals .
The Saudi Arabia Gene Therapy Cell Culture Media Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Cytiva (formerly GE Healthcare Life Sciences), Corning Incorporated, Becton, Dickinson and Company (BD), FUJIFILM Irvine Scientific, Sigma-Aldrich (now part of Merck KGaA), CellGenix GmbH, PromoCell GmbH, Sartorius AG, EMD Millipore (Merck Millipore), Bio-Techne Corporation, InVivoGen, STEMCELL Technologies Inc., Danaher Corporation (Cytiva parent), FUJIFILM Holdings Corporation, King Faisal Specialist Hospital & Research Centre (KFSHRC), King Abdullah International Medical Research Center (KAIMRC), King Abdullah University of Science and Technology (KAUST) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabia gene therapy cell culture media market appears promising, driven by ongoing advancements in biotechnology and increasing healthcare investments. As the government continues to support research initiatives, the market is expected to witness a surge in innovative therapies. Additionally, the growing focus on personalized medicine will likely create new opportunities for specialized cell culture media, enhancing treatment efficacy and patient outcomes. Overall, the market is set for significant growth in the future.
| Segment | Sub-Segments |
|---|---|
| By Type | Serum-based media Serum-free media Chemically defined media Stem cell culture media Classical media Specialty media Other cell culture media |
| By Application | Research and development Clinical applications Commercial production Tissue engineering and regenerative medicine Others |
| By End-User | Pharmaceutical and biotechnology companies Academic and research institutions Contract research organizations (CROs) Hospitals and clinics Others |
| By Distribution Channel | Direct sales Distributors Online sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region Others |
| By Price Range | Low price range Mid price range High price range |
| By Packaging Type | Single-use packaging Multi-use packaging Bulk packaging Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Research Institutions | 60 | Research Scientists, Lab Directors |
| Biotechnology Firms | 40 | Product Managers, R&D Heads |
| Pharmaceutical Companies | 40 | Clinical Development Managers, Regulatory Affairs Specialists |
| Healthcare Providers | 40 | Oncologists, Genetic Counselors |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
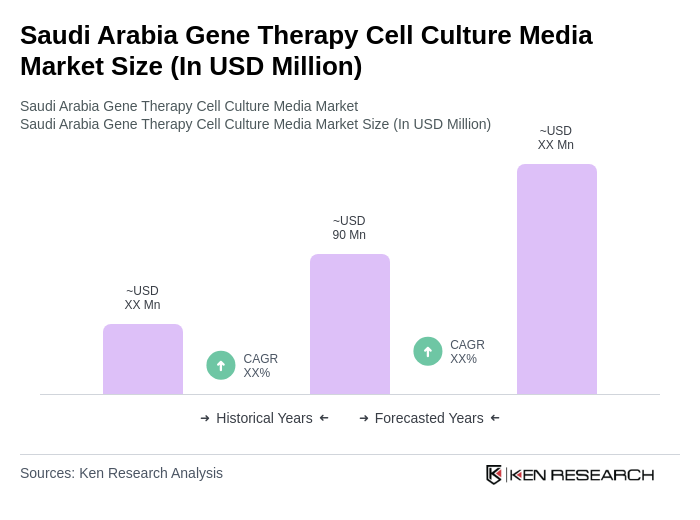
The Saudi Arabia Gene Therapy Cell Culture Media Market is valued at approximately USD 90 million, reflecting significant growth driven by advancements in biotechnology and increasing investments in healthcare under the Vision 2030 initiative.