About the Report
Base Year 2024Saudi Arabia Multiple Sclerosis Therapeutics Market Overview
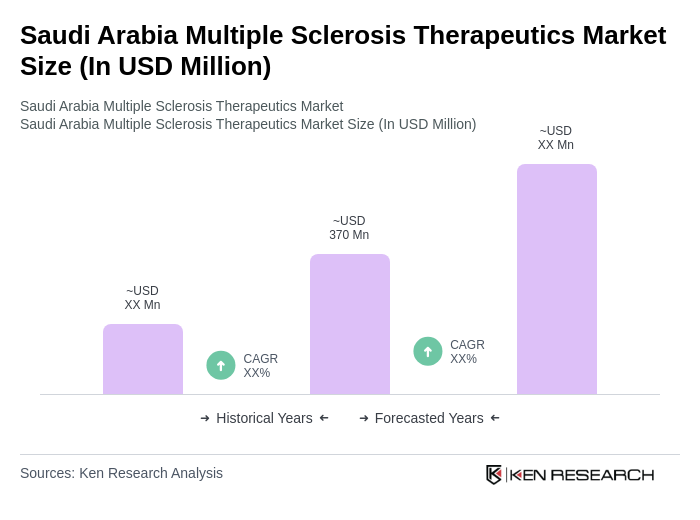
- The Saudi Arabia Multiple Sclerosis Therapeutics Market is valued at USD 370 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of multiple sclerosis (MS) in the region, advancements in disease-modifying therapies, and heightened awareness regarding early diagnosis and treatment. The market is also supported by rising healthcare expenditure, government initiatives to improve healthcare infrastructure, and the introduction of innovative digital health tools for MS management .
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their advanced healthcare facilities and concentration of specialized medical professionals. These urban centers are equipped with state-of-the-art hospitals and clinics that offer comprehensive MS treatment options, including access to the latest disease-modifying therapies and personalized care plans, making them pivotal in the delivery of healthcare services for MS patients .
- In 2023, the Saudi Arabian government enacted the “Unified Health Insurance Policy for Cooperative Health Insurance,” issued by the Council of Cooperative Health Insurance, mandating the inclusion of multiple sclerosis treatments in the national health insurance scheme. This regulation requires all licensed health insurance providers to cover essential MS therapies, enhancing patient access to advanced treatments and ensuring timely, effective care for MS patients across the Kingdom .
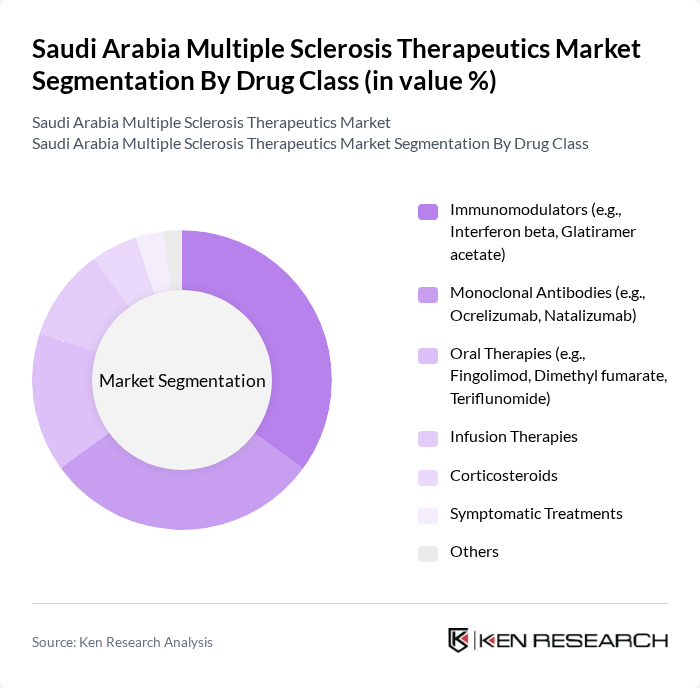
Saudi Arabia Multiple Sclerosis Therapeutics Market Segmentation
By Drug Class:The market is segmented into various drug classes, each addressing specific therapeutic needs of MS patients. The primary drug classes include Immunomodulators, Monoclonal Antibodies, Oral Therapies, Infusion Therapies, Corticosteroids, Symptomatic Treatments, and Others. Among these, Immunomodulators and Monoclonal Antibodies lead due to their proven effectiveness in reducing relapse rates and slowing disease progression. Recent trends highlight the adoption of oral therapies and monoclonal antibodies, reflecting a shift toward patient convenience and improved efficacy .
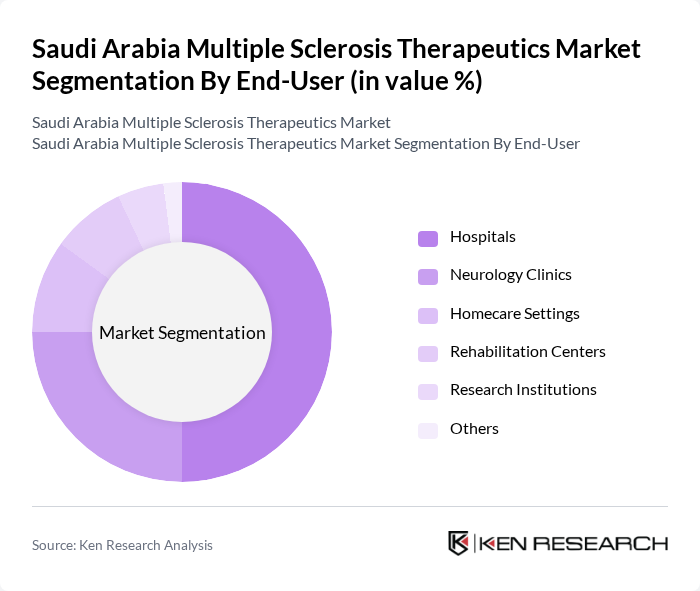
By End-User:The end-user segmentation includes Hospitals, Neurology Clinics, Homecare Settings, Rehabilitation Centers, Research Institutions, and Others. Hospitals and Neurology Clinics remain the primary end-users, as they provide specialized care and advanced treatment protocols for MS patients, supported by multidisciplinary teams and access to the latest therapies. The growing role of homecare and rehabilitation centers reflects the increasing emphasis on long-term disease management and quality of life improvements .
Saudi Arabia Multiple Sclerosis Therapeutics Market Competitive Landscape
The Saudi Arabia Multiple Sclerosis Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Biogen Inc., Merck KGaA, Sanofi S.A., Teva Pharmaceutical Industries Ltd., Roche Holding AG, AbbVie Inc., Bayer AG, Celgene Corporation (now part of Bristol Myers Squibb), EMD Serono (Merck KGaA's US/Canada biopharma division), Genzyme Corporation (Sanofi Genzyme), Acorda Therapeutics, Inc., Sandoz International GmbH, Hikma Pharmaceuticals PLC, UCB S.A., Takeda Pharmaceutical Company Limited, Pfizer Inc., Johnson & Johnson Services, Inc., Neuraxpharm Group contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Multiple Sclerosis Therapeutics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Multiple Sclerosis:The prevalence of Multiple Sclerosis (MS) in Saudi Arabia is estimated to be around 50 cases per 100,000 people, with approximately 8,000 to 10,000 diagnosed patients as of in future. This rising incidence is attributed to genetic, environmental, and lifestyle factors. The increasing number of diagnosed cases drives demand for effective therapeutics, leading to a projected increase in market growth as healthcare providers seek to address this growing health concern.
- Advancements in Therapeutic Options:The Saudi healthcare sector has seen significant advancements in MS treatment options, with more than 10 new therapies approved in the last five years. These include disease-modifying therapies (DMTs) that have shown improved efficacy and safety profiles. The introduction of innovative treatments enhances patient outcomes and encourages healthcare providers to adopt these therapies, thereby expanding the market for MS therapeutics in the region.
- Growing Healthcare Expenditure:Saudi Arabia's healthcare expenditure is projected to reach approximately SAR 189 billion (USD 50.4 billion) in future, reflecting a commitment to improving healthcare services. This increase in funding supports the development and accessibility of MS therapeutics, enabling better patient care and treatment options. Enhanced healthcare spending is expected to facilitate the introduction of advanced therapies, further driving market growth in the MS therapeutics sector.
Market Challenges
- High Cost of Treatment:The average annual cost of MS treatment in Saudi Arabia can exceed SAR 100,000 (USD 26,600), which poses a significant financial burden on patients and healthcare systems. This high cost limits access to necessary therapies, particularly for those without comprehensive insurance coverage. Consequently, the financial constraints can hinder the overall growth of the MS therapeutics market as many patients may delay or forgo treatment.
- Limited Access to Specialized Care:Access to specialized MS care is limited, particularly in rural areas of Saudi Arabia, where only about one-third of neurologists are located. This geographical disparity results in inadequate diagnosis and treatment options for patients. The lack of specialized care can lead to delayed treatment initiation, negatively impacting patient outcomes and overall market growth for MS therapeutics in the region.
Saudi Arabia Multiple Sclerosis Therapeutics Market Future Outlook
The future of the Saudi Arabia Multiple Sclerosis therapeutics market appears promising, driven by ongoing advancements in treatment options and increased healthcare investments. As the prevalence of MS continues to rise, healthcare providers are likely to adopt innovative therapies, including oral medications and biologics. Additionally, the integration of digital health solutions will enhance patient monitoring and adherence, ultimately improving treatment outcomes and expanding market potential in the coming years.
Market Opportunities
- Expansion of Telemedicine Services:The growth of telemedicine in Saudi Arabia presents a significant opportunity for MS management. With over 98% of the population having access to the internet, telemedicine can facilitate remote consultations and follow-ups, improving patient access to specialists and enhancing treatment adherence, ultimately benefiting the MS therapeutics market.
- Development of Personalized Medicine:The shift towards personalized medicine in MS treatment offers substantial market opportunities. By leveraging genetic and biomarker data, healthcare providers can tailor therapies to individual patient profiles, improving efficacy and minimizing side effects. This approach is expected to gain traction, driving innovation and growth in the MS therapeutics market in Saudi Arabia.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Drug Class | Immunomodulators (e.g., Interferon beta, Glatiramer acetate) Monoclonal Antibodies (e.g., Ocrelizumab, Natalizumab) Oral Therapies (e.g., Fingolimod, Dimethyl fumarate, Teriflunomide) Infusion Therapies Corticosteroids Symptomatic Treatments Others |
| By End-User | Hospitals Neurology Clinics Homecare Settings Rehabilitation Centers Research Institutions Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Direct Sales Others |
| By Route of Administration | Oral Injectable (Intramuscular, Subcutaneous) Intravenous Others |
| By Patient Demographics | Age Group (Pediatric, Adult, Geriatric) Gender Socioeconomic Status Geographic Distribution Others |
| By Treatment Duration | Short-term Treatment Long-term Treatment Maintenance Therapy Others |
| By Pricing Tier | Premium Mid-range Economy Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Pharmaceutical Manufacturers
Biotechnology Companies
Healthcare Providers and Hospitals
Insurance Companies
Patient Advocacy Groups
Clinical Research Organizations
Players Mentioned in the Report:
Novartis AG
Biogen Inc.
Merck KGaA
Sanofi S.A.
Teva Pharmaceutical Industries Ltd.
Roche Holding AG
AbbVie Inc.
Bayer AG
Celgene Corporation (now part of Bristol Myers Squibb)
EMD Serono (Merck KGaA's US/Canada biopharma division)
Genzyme Corporation (Sanofi Genzyme)
Acorda Therapeutics, Inc.
Sandoz International GmbH
Hikma Pharmaceuticals PLC
UCB S.A.
Takeda Pharmaceutical Company Limited
Pfizer Inc.
Johnson & Johnson Services, Inc.
Neuraxpharm Group
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Multiple Sclerosis Therapeutics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Multiple Sclerosis Therapeutics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Multiple Sclerosis Therapeutics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of Multiple Sclerosis
3.1.2 Advancements in therapeutic options
3.1.3 Growing healthcare expenditure
3.1.4 Rising awareness and diagnosis rates
3.2 Market Challenges
3.2.1 High cost of treatment
3.2.2 Limited access to specialized care
3.2.3 Regulatory hurdles
3.2.4 Variability in patient adherence to treatment
3.3 Market Opportunities
3.3.1 Expansion of telemedicine services
3.3.2 Development of personalized medicine
3.3.3 Collaborations with research institutions
3.3.4 Increasing investment in healthcare infrastructure
3.4 Market Trends
3.4.1 Shift towards oral therapies
3.4.2 Integration of digital health solutions
3.4.3 Focus on patient-centric care models
3.4.4 Rise in biosimilars and generics
3.5 Government Regulation
3.5.1 Approval processes for new therapies
3.5.2 Pricing regulations for pharmaceuticals
3.5.3 Policies promoting research and development
3.5.4 Guidelines for clinical trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Multiple Sclerosis Therapeutics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Multiple Sclerosis Therapeutics Market Segmentation
8.1 By Drug Class
8.1.1 Immunomodulators (e.g., Interferon beta, Glatiramer acetate)
8.1.2 Monoclonal Antibodies (e.g., Ocrelizumab, Natalizumab)
8.1.3 Oral Therapies (e.g., Fingolimod, Dimethyl fumarate, Teriflunomide)
8.1.4 Infusion Therapies
8.1.5 Corticosteroids
8.1.6 Symptomatic Treatments
8.1.7 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Neurology Clinics
8.2.3 Homecare Settings
8.2.4 Rehabilitation Centers
8.2.5 Research Institutions
8.2.6 Others
8.3 By Distribution Channel
8.3.1 Hospital Pharmacies
8.3.2 Retail Pharmacies
8.3.3 Online Pharmacies
8.3.4 Direct Sales
8.3.5 Others
8.4 By Route of Administration
8.4.1 Oral
8.4.2 Injectable (Intramuscular, Subcutaneous)
8.4.3 Intravenous
8.4.4 Others
8.5 By Patient Demographics
8.5.1 Age Group (Pediatric, Adult, Geriatric)
8.5.2 Gender
8.5.3 Socioeconomic Status
8.5.4 Geographic Distribution
8.5.5 Others
8.6 By Treatment Duration
8.6.1 Short-term Treatment
8.6.2 Long-term Treatment
8.6.3 Maintenance Therapy
8.6.4 Others
8.7 By Pricing Tier
8.7.1 Premium
8.7.2 Mid-range
8.7.3 Economy
8.7.4 Others
9. Saudi Arabia Multiple Sclerosis Therapeutics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Share in Saudi Arabia MS Therapeutics
9.2.3 Revenue Growth Rate (Saudi Arabia MS portfolio)
9.2.4 Number of Approved MS Products in Saudi Arabia
9.2.5 Patient Access Programs (availability, reach)
9.2.6 Local Partnerships/Distribution Agreements
9.2.7 R&D Investment Ratio (MS-focused)
9.2.8 Regulatory Approval Speed (Saudi FDA)
9.2.9 Sales Channel Effectiveness (hospital, retail, online)
9.2.10 Brand Recognition among Saudi Neurologists
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Novartis AG
9.5.2 Biogen Inc.
9.5.3 Merck KGaA
9.5.4 Sanofi S.A.
9.5.5 Teva Pharmaceutical Industries Ltd.
9.5.6 Roche Holding AG
9.5.7 AbbVie Inc.
9.5.8 Bayer AG
9.5.9 Celgene Corporation (now part of Bristol Myers Squibb)
9.5.10 EMD Serono (Merck KGaA's US/Canada biopharma division)
9.5.11 Genzyme Corporation (Sanofi Genzyme)
9.5.12 Acorda Therapeutics, Inc.
9.5.13 Sandoz International GmbH
9.5.14 Hikma Pharmaceuticals PLC
9.5.15 UCB S.A.
9.5.16 Takeda Pharmaceutical Company Limited
9.5.17 Pfizer Inc.
9.5.18 Johnson & Johnson Services, Inc.
9.5.19 Neuraxpharm Group
10. Saudi Arabia Multiple Sclerosis Therapeutics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for MS Treatments
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Evaluation Criteria
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Funding for Research Initiatives
10.2.3 Partnerships with Private Sector
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Treatment
10.3.2 Affordability of Medications
10.3.3 Availability of Support Services
10.4 User Readiness for Adoption
10.4.1 Awareness of Treatment Options
10.4.2 Willingness to Engage with New Therapies
10.4.3 Training Needs for Healthcare Providers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Treatment Efficacy
10.5.2 Long-term Patient Outcomes
10.5.3 Expansion into New Patient Segments
11. Saudi Arabia Multiple Sclerosis Therapeutics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Key Partnerships
1.5 Cost Structure Overview
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Partnerships with Healthcare Providers
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends in Patient Needs
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Competitive Advantages
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Innovations
9.2 Export Entry Strategy
9.2.1 Target Countries Analysis
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Market Entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from health organizations and government health ministries in Saudi Arabia
- Review of scientific literature and clinical studies related to multiple sclerosis treatments
- Examination of market reports and industry publications focusing on pharmaceutical and biotechnology sectors
Primary Research
- Interviews with neurologists and healthcare professionals specializing in multiple sclerosis
- Surveys conducted with patients diagnosed with multiple sclerosis to understand treatment preferences
- Discussions with pharmaceutical representatives and distributors regarding market dynamics
Validation & Triangulation
- Cross-validation of findings through multiple data sources including clinical guidelines and treatment protocols
- Triangulation of market data with insights from healthcare professionals and patient feedback
- Sanity checks through expert panel reviews comprising neurologists and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national health expenditure and prevalence rates of multiple sclerosis
- Segmentation of the market by therapeutic categories, including disease-modifying therapies and symptomatic treatments
- Incorporation of government healthcare initiatives and funding for multiple sclerosis research and treatment
Bottom-up Modeling
- Collection of sales data from leading pharmaceutical companies offering multiple sclerosis therapies in Saudi Arabia
- Estimation of treatment costs based on pricing strategies and reimbursement policies
- Volume estimates derived from patient population data and treatment adherence rates
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating demographic trends, healthcare access, and treatment advancements
- Scenario modeling based on potential changes in healthcare policies and drug approval timelines
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Professionals | 100 | Neurologists, General Practitioners |
| Patients with Multiple Sclerosis | 80 | Individuals diagnosed with multiple sclerosis |
| Pharmaceutical Representatives | 50 | Sales Managers, Product Managers |
| Healthcare Policy Makers | 40 | Health Ministry Officials, Regulatory Authorities |
| Market Analysts | 40 | Healthcare Market Researchers, Industry Analysts |
Frequently Asked Questions
What is the current value of the Saudi Arabia Multiple Sclerosis Therapeutics Market?
The Saudi Arabia Multiple Sclerosis Therapeutics Market is valued at approximately USD 370 million, reflecting a significant growth driven by the increasing prevalence of multiple sclerosis, advancements in therapies, and rising healthcare expenditure in the region.