Region:Middle East
Author(s):Rebecca
Product Code:KRAA9221
Pages:96
Published On:November 2025
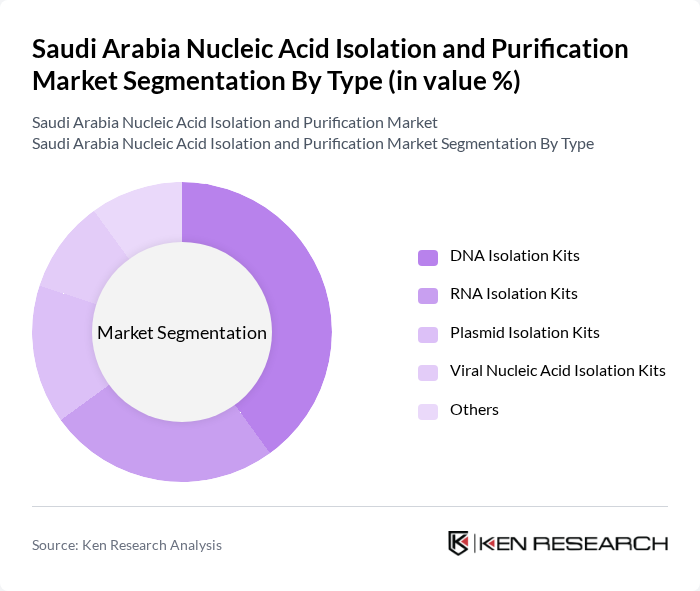
By Type:The market is segmented into various types of nucleic acid isolation kits, each catering to specific needs in research and diagnostics. The primary subsegments include DNA Isolation Kits, RNA Isolation Kits, Plasmid Isolation Kits, Viral Nucleic Acid Isolation Kits, and Others. Among these, DNA Isolation Kits are the most widely used due to their critical role in genetic research and diagnostics, driven by the increasing focus on genomics, automation in sample processing, and personalized medicine .
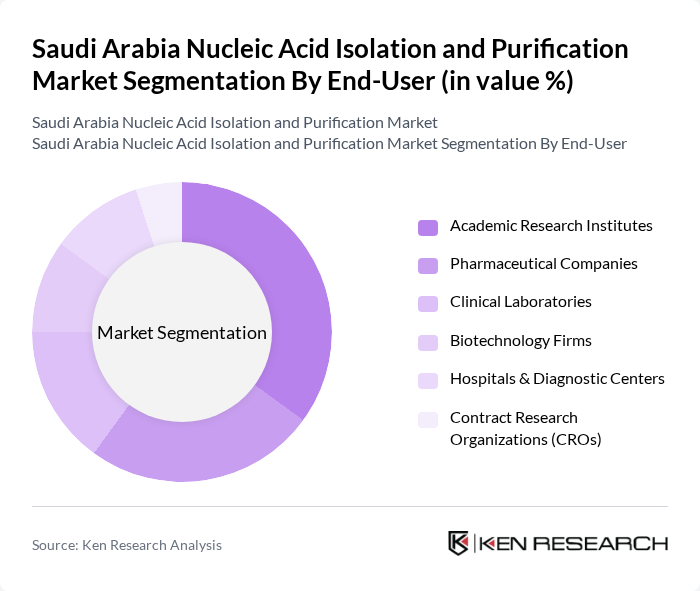
By End-User:The end-user segmentation includes Academic Research Institutes, Pharmaceutical Companies, Clinical Laboratories, Biotechnology Firms, Hospitals & Diagnostic Centers, Contract Research Organizations (CROs), and Others. Academic Research Institutes are the leading end-users, driven by the increasing number of research projects focusing on genomics and molecular biology, which require reliable nucleic acid isolation methods. The growing presence of biotechnology firms and clinical laboratories is also contributing to market expansion, especially with the adoption of high-throughput and automated nucleic acid extraction systems .
The Saudi Arabia Nucleic Acid Isolation and Purification Market is characterized by a dynamic mix of regional and international players. Leading participants such as Qiagen N.V., Thermo Fisher Scientific Inc., Roche Diagnostics, Bio-Rad Laboratories, Inc., Agilent Technologies, Inc., Merck KGaA, New England Biolabs, Inc., Promega Corporation, Takara Bio Inc., Zymo Research Corporation, GenScript Biotech Corporation, Abcam plc, Bioneer Corporation, Eppendorf AG, SeraCare Life Sciences, Inc., Illumina, Inc., PerkinElmer, Inc., Saudi Biotechnology Manufacturing Co., Al-Dawaa Pharmacies (Life Sciences Division), Al Jeel Medical & Trading Co. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the nucleic acid isolation and purification market in Saudi Arabia appears promising, driven by ongoing advancements in biotechnology and increasing government support for healthcare initiatives. As the demand for genetic testing continues to rise, laboratories are likely to adopt more automated and efficient technologies. Additionally, the integration of artificial intelligence in nucleic acid analysis is expected to enhance accuracy and speed, further propelling market growth. The focus on personalized medicine will also create new avenues for innovation and investment in this sector.
| Segment | Sub-Segments |
|---|---|
| By Type | DNA Isolation Kits RNA Isolation Kits Plasmid Isolation Kits Viral Nucleic Acid Isolation Kits Others |
| By End-User | Academic Research Institutes Pharmaceutical Companies Clinical Laboratories Biotechnology Firms Hospitals & Diagnostic Centers Contract Research Organizations (CROs) Others |
| By Application | Diagnostics Drug Development Forensic Science Genomics & Transcriptomics Research Others |
| By Technology | Column-Based Technology Magnetic Bead Technology Spin Column Technology Automated Liquid Handling Systems Others |
| By Source | Plant-Based Sources Animal-Based Sources Microbial Sources Viral Sources Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Academic Research Institutions | 120 | Research Scientists, Lab Technicians |
| Clinical Laboratories | 90 | Laboratory Managers, Quality Control Officers |
| Biotechnology Companies | 60 | Product Development Managers, R&D Directors |
| Healthcare Providers | 50 | Clinical Pathologists, Molecular Diagnostics Specialists |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
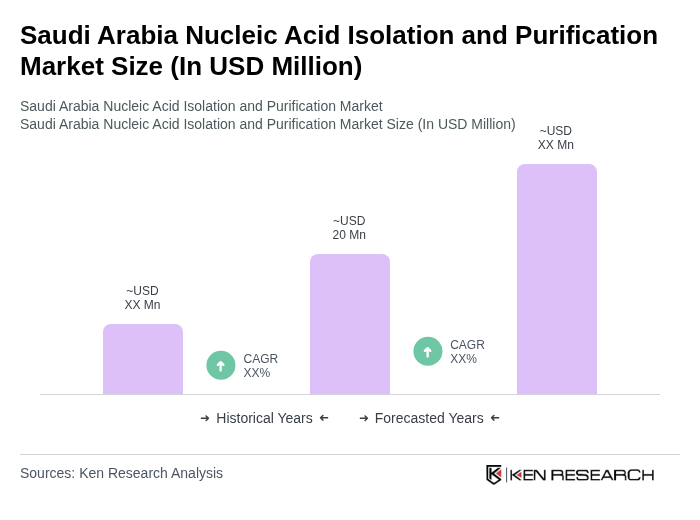
The Saudi Arabia Nucleic Acid Isolation and Purification Market is valued at approximately USD 20 million, reflecting a five-year historical analysis driven by advancements in biotechnology and increasing demand for genetic testing and diagnostics.