Region:Middle East
Author(s):Dev
Product Code:KRAA6038
Pages:96
Published On:January 2026
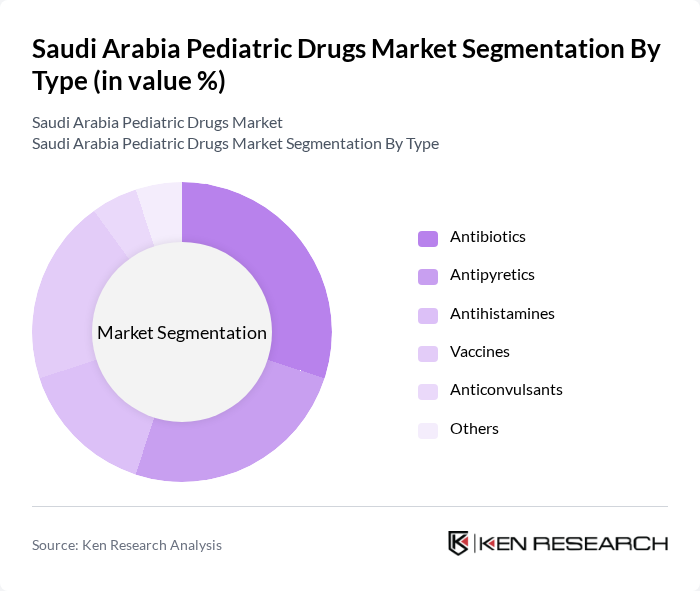
By Type:The pediatric drugs market is segmented into various types, including antibiotics, antipyretics, antihistamines, vaccines, anticonvulsants, and others. Among these, antibiotics and vaccines are the most significant subsegments due to the high prevalence of infectious diseases and the ongoing vaccination programs in the country. The demand for antipyretics and antihistamines is also notable, driven by seasonal illnesses and allergies among children.
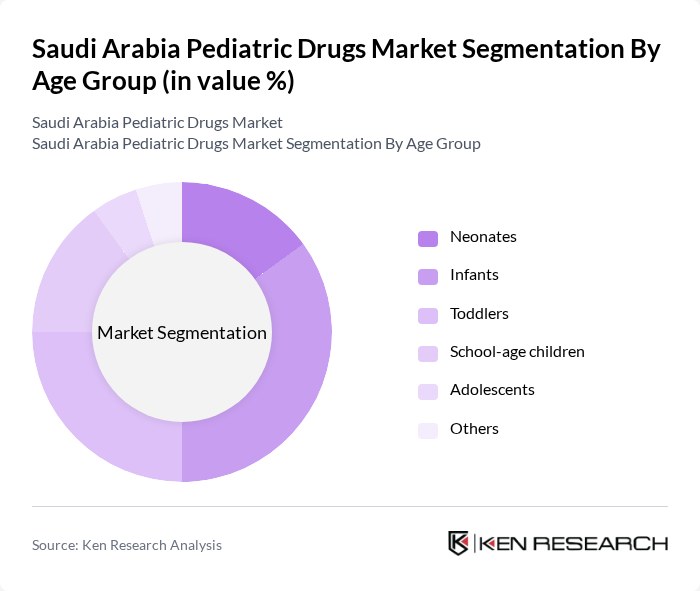
By Age Group:The market is also segmented by age groups, including neonates, infants, toddlers, school-age children, adolescents, and others. Infants and toddlers represent the largest segments due to their higher susceptibility to infections and the need for regular vaccinations. The increasing awareness among parents regarding pediatric health is driving the demand for specialized medications tailored to these age groups.
The Saudi Arabia Pediatric Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer, GlaxoSmithKline, Novartis, Sanofi, Merck & Co., Johnson & Johnson, AstraZeneca, Roche, AbbVie, Bayer, Takeda Pharmaceutical Company, Amgen, Bristol-Myers Squibb, Eli Lilly and Company, Teva Pharmaceutical Industries contribute to innovation, geographic expansion, and service delivery in this space.
The future of the pediatric drugs market in Saudi Arabia appears promising, driven by increasing healthcare investments and a growing focus on child health. The government’s commitment to enhancing pediatric healthcare services is expected to foster innovation in drug development. Additionally, the rise of digital health solutions and telemedicine will likely improve access to pediatric care, facilitating better health outcomes. As awareness of pediatric health issues grows, the demand for specialized medications is anticipated to increase, creating a dynamic market environment.
| Segment | Sub-Segments |
|---|---|
| By Type | Antibiotics Antipyretics Antihistamines Vaccines Anticonvulsants Others |
| By Age Group | Neonates Infants Toddlers School-age children Adolescents Others |
| By Distribution Channel | Hospitals Retail pharmacies Online pharmacies Clinics Others |
| By Formulation | Liquid Tablet Powder Suspension Others |
| By Therapeutic Area | Infectious diseases Respiratory disorders Gastrointestinal disorders Neurological disorders Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Policy Support | Government subsidies Tax incentives Research grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatricians and Child Health Specialists | 100 | Pediatricians, Family Doctors |
| Pharmacists in Retail Pharmacies | 80 | Community Pharmacists, Pharmacy Managers |
| Parents of Pediatric Patients | 150 | Parents, Guardians |
| Healthcare Policy Makers | 50 | Health Administrators, Policy Analysts |
| Hospital Administrators | 70 | Hospital Managers, Clinical Directors |
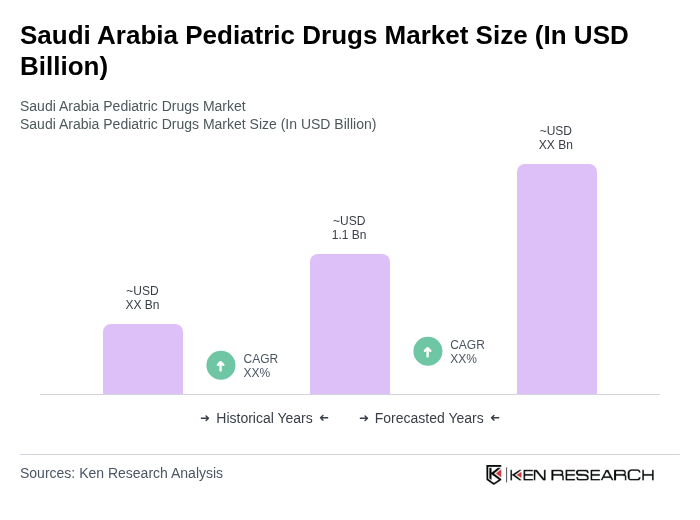
The Saudi Arabia Pediatric Drugs Market is valued at approximately USD 1.1 billion, reflecting a significant growth driven by increased healthcare expenditure, rising awareness of pediatric health issues, and government initiatives aimed at enhancing pediatric healthcare services.