Region:Middle East
Author(s):Dev
Product Code:KRAB1893
Pages:90
Published On:January 2026
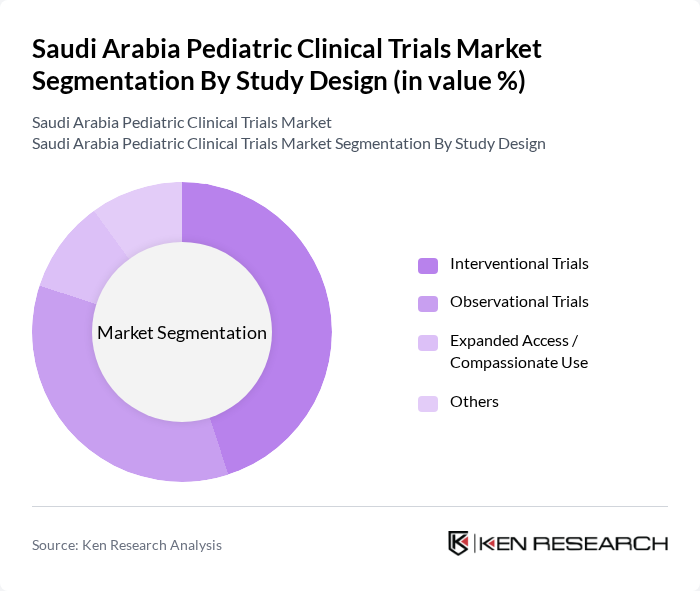
By Study Design:The study design segmentation includes various methodologies employed in clinical trials. The primary subsegments are Interventional Trials, Observational Trials, Expanded Access / Compassionate Use, and Others. Interventional Trials are often the most prevalent due to their focus on testing new treatments, vaccines, and biologics, which are crucial for pediatric populations and are commonly used in sponsored drug and vaccine development programs. Observational Trials follow closely, providing valuable data on the natural progression of diseases in children, treatment patterns, and long-term safety outcomes, and are frequently used in registries and post-marketing safety studies.
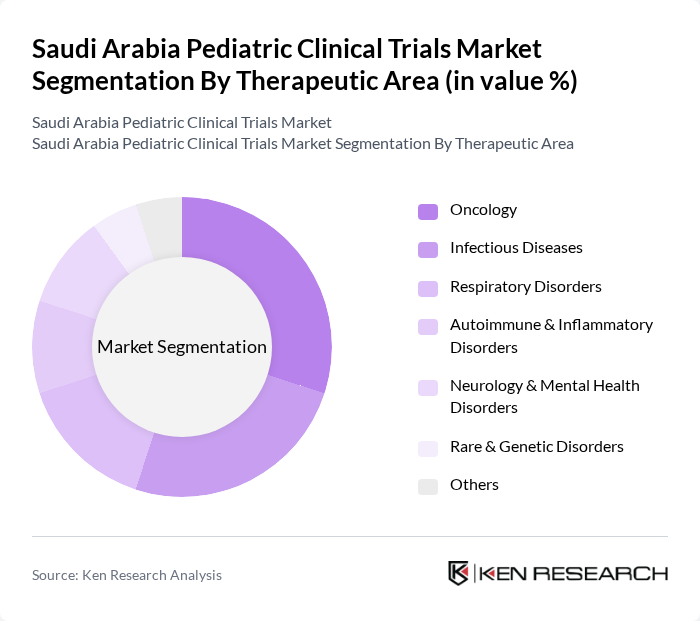
By Therapeutic Area:This segmentation focuses on the various medical conditions addressed in pediatric clinical trials. The key subsegments include Oncology, Infectious Diseases, Respiratory Disorders, Autoimmune & Inflammatory Disorders, Neurology & Mental Health Disorders, Rare & Genetic Disorders, and Others. Oncology trials are particularly significant due to the increasing attention to childhood cancers and hematologic malignancies and the need for targeted therapies and immunotherapies in pediatric oncology. Infectious Diseases also represent a substantial portion of trials, especially vaccine and anti-infective studies driven by global health challenges such as respiratory viral infections and emerging pathogens, which have accelerated pediatric vaccine development programs.
The Saudi Arabia Pediatric Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as King Faisal Specialist Hospital & Research Centre, King Abdullah International Medical Research Center (KAIMRC), Ministry of National Guard Health Affairs, King Saud University Medical City, Saudi Food and Drug Authority (SFDA), Saudi Clinical Trials Registry (SCTR) – Key Role in Pediatric Trials, Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO), Riyadh Pharma, Gulf Pharmaceutical Industries (Julphar) – Saudi Operations, AstraZeneca Saudi Arabia, Novartis Pharmaceuticals Saudi Arabia, Pfizer Saudi Arabia, Merck Sharp & Dohme (MSD) Saudi Arabia, Sanofi Saudi Arabia, GlaxoSmithKline (GSK) Saudi Arabia contribute to innovation, geographic expansion, and service delivery in this space.
The future of the pediatric clinical trials market in Saudi Arabia appears promising, driven by advancements in technology and increasing collaboration between public and private sectors. The integration of digital health technologies is expected to enhance trial efficiency and patient engagement. Additionally, the focus on patient-centric trial designs will likely improve recruitment and retention rates. As the government continues to support research initiatives, the market is poised for growth, with a strong emphasis on innovative therapies tailored for children.
| Segment | Sub-Segments |
|---|---|
| By Study Design | Interventional Trials Observational Trials Expanded Access / Compassionate Use Others |
| By Therapeutic Area | Oncology Infectious Diseases Respiratory Disorders Autoimmune & Inflammatory Disorders Neurology & Mental Health Disorders Rare & Genetic Disorders Others |
| By Phase of Trial | Phase I Phase II Phase III Phase IV |
| By Sponsor Type | Industry (Pharmaceutical & Biopharmaceutical Companies) Contract Research Organizations (CROs) Academic & Research Institutions Government & Non-Government Organizations Others |
| By Age Group | Neonates (0–28 days) Infants (29 days–23 months) Children (2–11 years) Adolescents (12–17 years) |
| By Trial Setting | Hospital-based Trials Community-based Trials Hybrid / Decentralized Trials Others |
| By Region | Central Region Eastern Region Western Region Southern Region Northern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatric Oncology Trials | 100 | Pediatric Oncologists, Clinical Trial Coordinators |
| Infectious Disease Trials | 80 | Pediatric Infectious Disease Specialists, Research Nurses |
| Rare Disease Trials | 60 | Pediatric Geneticists, Patient Advocacy Representatives |
| Vaccine Trials | 90 | Pediatric Immunologists, Public Health Officials |
| Behavioral Health Trials | 70 | Pediatric Psychiatrists, Clinical Psychologists |
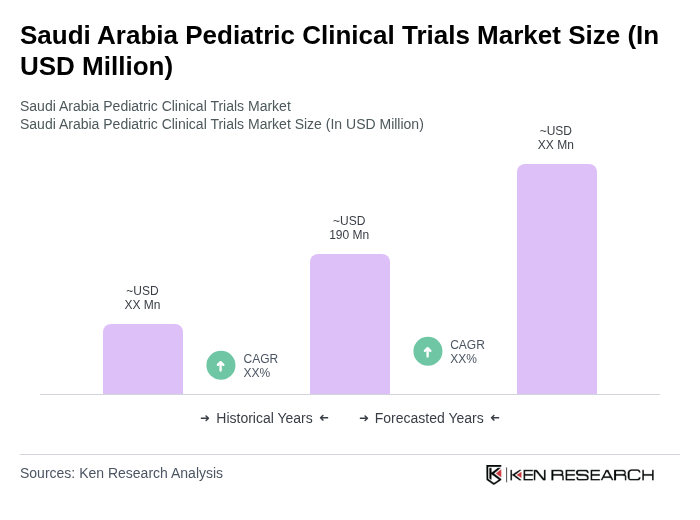
The Saudi Arabia Pediatric Clinical Trials Market is valued at approximately USD 190 million, reflecting a five-year historical analysis and recent revenue estimates for pediatric clinical research activities, driven by healthcare investments and the need for age-appropriate medications.