Region:Middle East
Author(s):Geetanshi
Product Code:KRAC4405
Pages:87
Published On:October 2025
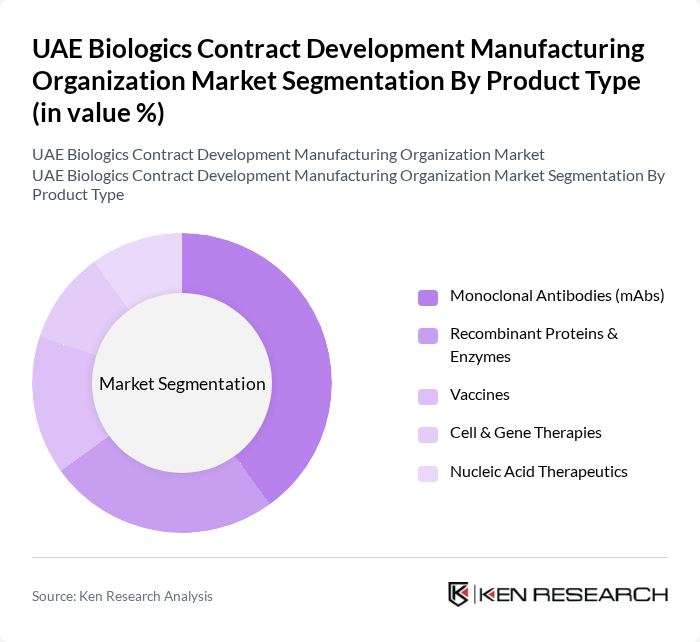
By Product Type:The product type segmentation includes Monoclonal Antibodies (mAbs), Recombinant Proteins & Enzymes, Vaccines, Cell & Gene Therapies, and Nucleic Acid Therapeutics. Among these,Monoclonal Antibodies (mAbs)dominate the market due to their widespread application in oncology and autoimmune diseases, supported by increasing investments in research and development. The growing prevalence of chronic diseases, the demand for targeted therapies, and the expansion of biosimilar production further bolster the mAbs segment .
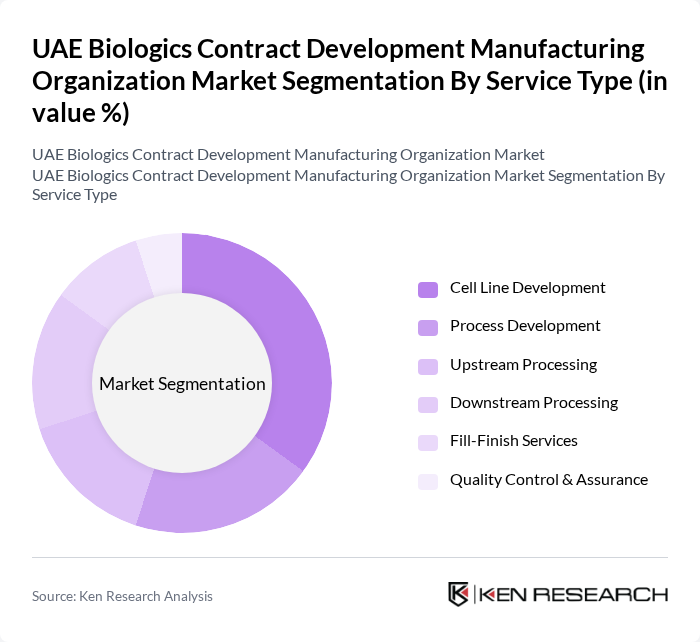
By Service Type:The service type segmentation encompasses Cell Line Development, Process Development, Upstream Processing, Downstream Processing, Fill-Finish Services, and Quality Control & Assurance. TheCell Line Developmentsegment is currently leading the market, driven by the increasing need for efficient and scalable production processes in biologics manufacturing. Growth in this segment is supported by advancements in cell line engineering, the adoption of single-use technologies, and the rising demand for personalized and precision medicine .
The UAE Biologics Contract Development Manufacturing Organization Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, Samsung Biologics, WuXi Biologics, Catalent, Inc., Boehringer Ingelheim BioXcellence, Fujifilm Diosynth Biotechnologies, Thermo Fisher Scientific (Patheon), Resilience (Joint Venture with Mubadala), AGC Biologics, KELIX Bio (Acquired by Mubadala), Recipharm AB, CordenPharma International, Emergent BioSolutions, KBI Biopharma, and Avid Bioservices contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE biologics contract development manufacturing organization market appears promising, driven by increasing investments in biotechnology and a growing focus on personalized medicine. As the region continues to enhance its infrastructure and regulatory frameworks, the market is expected to attract more global players. Additionally, the integration of digital technologies and sustainable practices will likely reshape manufacturing processes, leading to improved efficiency and reduced environmental impact, positioning the UAE as a key player in the global biologics landscape.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Monoclonal Antibodies (mAbs) Recombinant Proteins & Enzymes Vaccines Cell & Gene Therapies Nucleic Acid Therapeutics |
| By Service Type | Cell Line Development Process Development Upstream Processing Downstream Processing Fill-Finish Services Quality Control & Assurance |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Contract Research Organizations |
| By Application | Oncology Immunology Infectious Diseases Rare Diseases Others |
| By Manufacturing Scale | Small-Scale (Clinical Phase) Large-Scale (Commercial Production) |
| By Region | Abu Dhabi Dubai Sharjah Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biologics Manufacturing Processes | 100 | Production Managers, Quality Control Analysts |
| Regulatory Compliance in Biologics | 60 | Regulatory Affairs Managers, Compliance Officers |
| Market Trends in Biologics | 75 | Market Analysts, Business Development Executives |
| Investment in Biologics R&D | 55 | R&D Directors, Financial Analysts |
| Customer Insights on Biologics | 65 | Healthcare Professionals, Pharmacists |
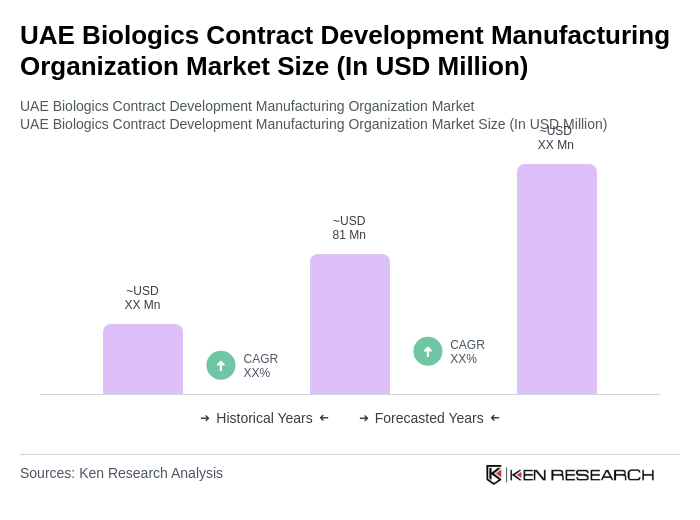
The UAE Biologics Contract Development Manufacturing Organization market is valued at approximately USD 81 million, reflecting a robust growth trajectory driven by increasing demand for biologics in therapeutic applications and advancements in biomanufacturing technologies.