Region:Middle East
Author(s):Rebecca
Product Code:KRAC4570
Pages:91
Published On:October 2025
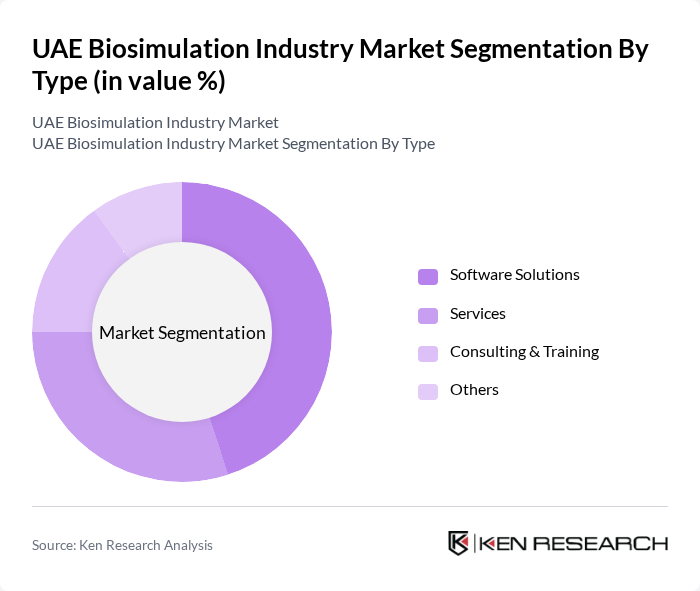
By Type:The market is segmented into various types, including Software Solutions, Services, Consulting & Training, and Others. Among these, Software Solutions are leading due to the increasing reliance on advanced software for drug development and simulation processes. The demand for user-friendly and efficient software tools is driving growth in this segment, as pharmaceutical companies seek to enhance their research capabilities. In the UAE, software solutions account for the largest share of the market, reflecting the global trend of digital transformation in pharmaceutical R&D .
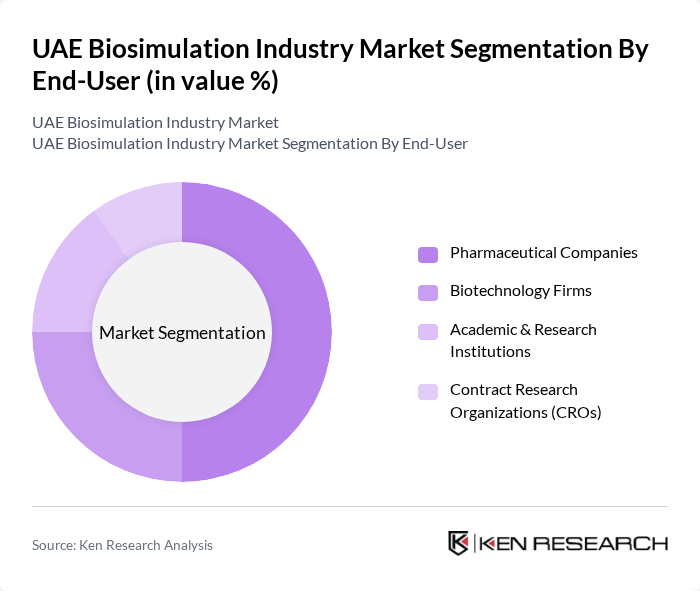
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic & Research Institutions, and Contract Research Organizations (CROs). Pharmaceutical Companies dominate this segment, driven by their need for efficient drug development processes and regulatory compliance. The increasing complexity of drug formulations and the need for faster time-to-market are pushing these companies to adopt biosimulation technologies extensively. The trend is reinforced by the UAE’s commitment to fostering pharmaceutical innovation and clinical research excellence .
The UAE Biosimulation Industry Market is characterized by a dynamic mix of regional and international players. Leading participants such as Simulations Plus, Inc., Certara, Inc., Dassault Systèmes, BIOVIA (Dassault Systèmes), InSilico Medicine, Genedata AG, Schrodinger, Inc., Chemical Computing Group ULC, Physiomics Plc, Rosa & Co. LLC, Instem plc, Immunetrics, Inc., Avantium N.V., Yokogawa Electric Corporation, Genomatix Software GmbH contribute to innovation, geographic expansion, and service delivery in this space .
The future of the UAE biosimulation industry appears promising, driven by technological advancements and increasing collaboration between public and private sectors. As machine learning and artificial intelligence continue to evolve, their integration into biosimulation processes will enhance predictive accuracy and efficiency. Furthermore, the growing emphasis on regulatory compliance will likely lead to the establishment of standardized practices, fostering trust and encouraging wider adoption of biosimulation technologies across the healthcare landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Software Solutions Services Consulting & Training Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Contract Research Organizations (CROs) |
| By Application | Drug Discovery & Development Toxicology & Safety Assessment Clinical Trial Simulation Others |
| By Region | Abu Dhabi Dubai Sharjah Northern Emirates & Others |
| By Investment Source | Private Investments Government Funding Venture Capital Others |
| By Policy Support | Research Grants Tax Incentives Regulatory Support Others |
| By Technology | In-Silico Modeling Virtual Patient Simulations Predictive Analytics & AI Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies Utilizing Biosimulation | 60 | R&D Directors, Clinical Trial Managers |
| Biotechnology Firms Engaged in Drug Development | 50 | Product Development Managers, Regulatory Affairs Specialists |
| Academic Institutions Conducting Biosimulation Research | 40 | Research Professors, Graduate Students |
| Healthcare Providers Implementing Simulation Technologies | 45 | Chief Medical Officers, IT Directors |
| Consulting Firms Advising on Biosimulation | 40 | Consultants, Market Analysts |
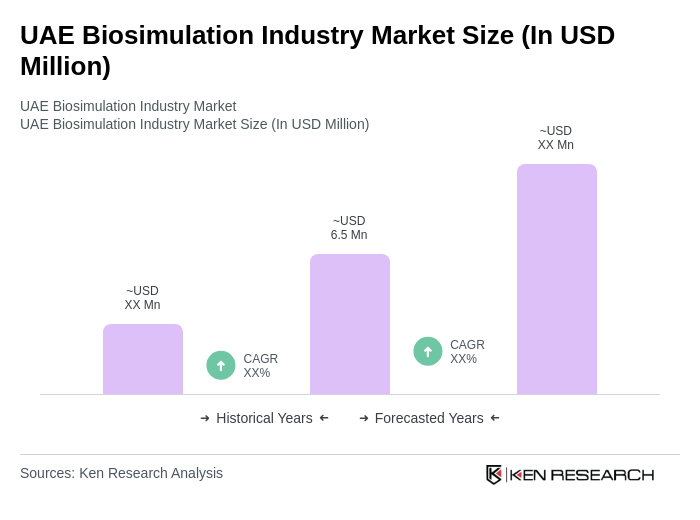
The UAE Biosimulation Industry Market is valued at approximately USD 6.5 million, reflecting significant growth driven by advancements in drug development, increased investments in biotechnology, and the rising demand for personalized medicine.