Region:Middle East
Author(s):Dev
Product Code:KRAD1582
Pages:84
Published On:November 2025
By Service:The market is segmented into two primary services: Biorepository Services and Archiving Solution Services. Biorepository Services involve the collection, processing, and storage of biological samples, while Archiving Solution Services focus on the long-term preservation and management of these samples.
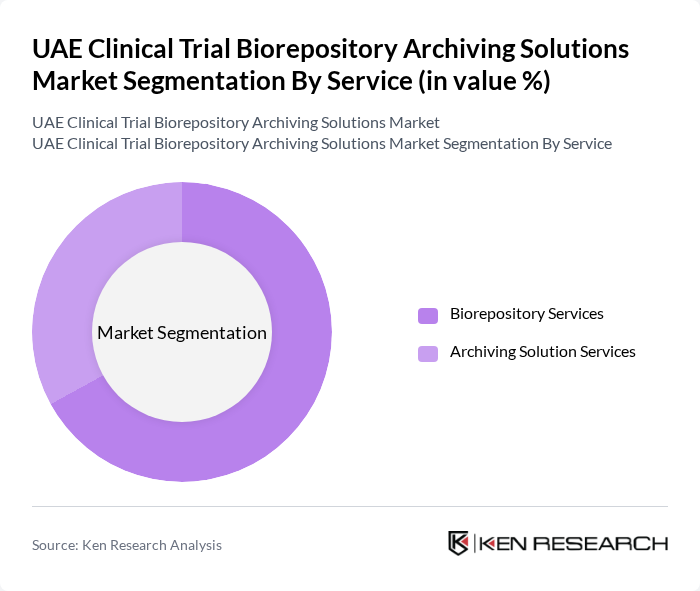
Biorepository Services dominate the market due to the increasing number of clinical trials and the growing need for sample collection, processing, and high-throughput storage. The demand for high-quality biological samples for research and development in pharmaceuticals, genomics, and biotechnology drives this segment. Additionally, advancements in automated biobanking technologies and the emphasis on precision medicine further enhance the relevance of biorepository services in the UAE.
By Phase:The market is segmented by clinical trial phases, including Preclinical, Phase I, Phase II, Phase III, and Phase IV. Each phase represents a critical stage in the clinical trial process, requiring specific biorepository and archiving solutions.
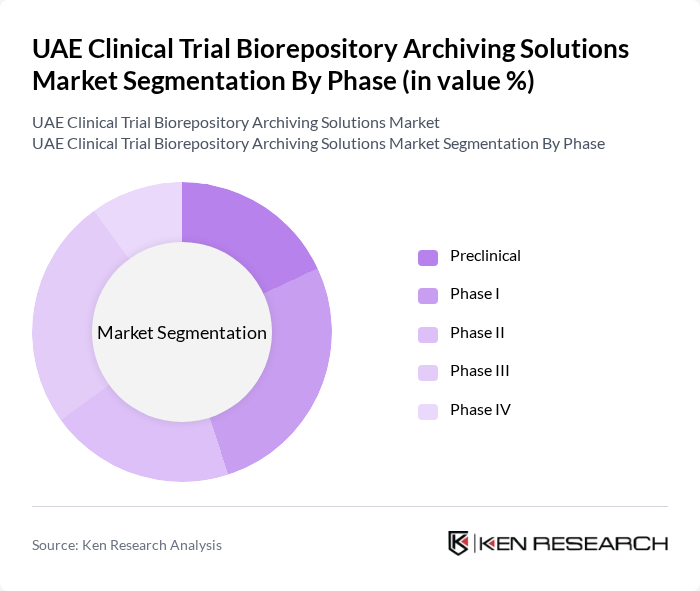
Phase I and Phase III dominate the market due to the high volume of clinical trials conducted in these stages. Phase I trials focus on safety and dosage, requiring extensive sample collection and management, while Phase III trials are pivotal for confirming efficacy and monitoring adverse reactions, necessitating robust biorepository solutions. The increasing complexity of clinical trial protocols and the need for comprehensive data archiving further drive demand in these phases.
The UAE Clinical Trial Biorepository Archiving Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abu Dhabi Stem Cells Center, Dubai Health Authority, Emirates Biobank (UAE University), Al Jalila Genomics Center, Sharjah Institute for Medical Research, Dubai Biotechnology & Research Park (DuBiotech), Emirates Health Services, Mohammed Bin Rashid University Biobank, Cleveland Clinic Abu Dhabi Biorepository, Health Authority Abu Dhabi (Department of Health – Abu Dhabi), Dubai Science Park, Al Ain Biobank, Gulf Biorepository, UAE University, Al Jalila Foundation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE Clinical Trial Biorepository Archiving Solutions Market appears promising, driven by technological advancements and a growing emphasis on patient-centric research. As the healthcare sector continues to evolve, the integration of digital solutions and AI in biorepositories will enhance data management and operational efficiency. Furthermore, increased collaboration with international research organizations is expected to foster innovation and expand the market, positioning the UAE as a leader in biobanking and clinical research.
| Segment | Sub-Segments |
|---|---|
| By Service | Biorepository Services Archiving Solution Services |
| By Phase | Preclinical Phase I Phase II Phase III Phase IV |
| By Product | Clinical Products Preclinical Products Human Tissue Organs Stem Cells Others |
| By End-User | Pharmaceutical Companies Biotechnology Companies Academic & Research Institutions Hospitals & Clinics Contract Research Organizations (CROs) Others |
| By Geographic Distribution | Abu Dhabi Dubai Sharjah Ajman Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 45 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Biotechnology Firms | 38 | Research Scientists, Biorepository Coordinators |
| Healthcare Institutions | 42 | Clinical Researchers, Ethics Committee Members |
| Regulatory Bodies | 15 | Policy Makers, Compliance Officers |
| Academic Institutions | 28 | Professors, Graduate Researchers in Clinical Trials |
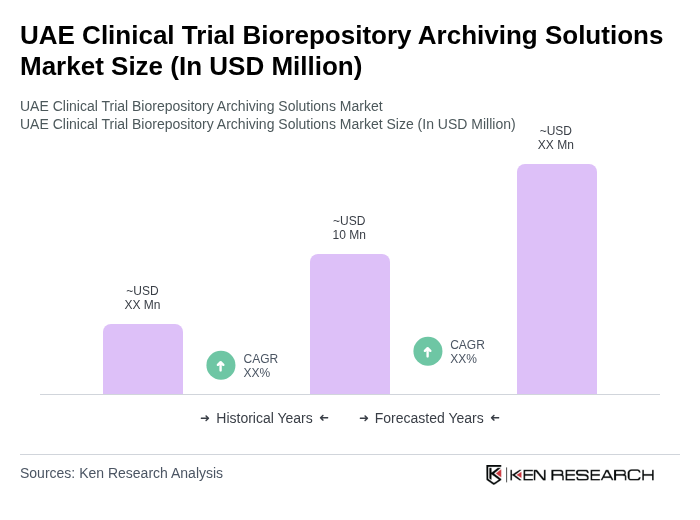
The UAE Clinical Trial Biorepository Archiving Solutions Market is valued at approximately USD 10 million, reflecting a five-year historical analysis. This growth is driven by the increasing number of clinical trials and advancements in biobanking automation.