Region:Middle East
Author(s):Dev
Product Code:KRAB1822
Pages:97
Published On:January 2026
By Testing Service Type:The testing service type segment includes various methodologies employed to evaluate the safety and efficacy of medical devices. The subsegments include In Vivo Testing Services, In Vitro Testing Services, Biocompatibility Testing, Microbiology & Sterility Testing, Chemistry and Analytical Testing, Toxicology and Safety Testing, and Others. Among these, In Vivo Testing Services is the most dominant due to its critical role in assessing the biological effects of medical devices in living organisms, particularly for implantable and high?risk devices, which is essential for regulatory approvals in line with international biocompatibility and safety standards.

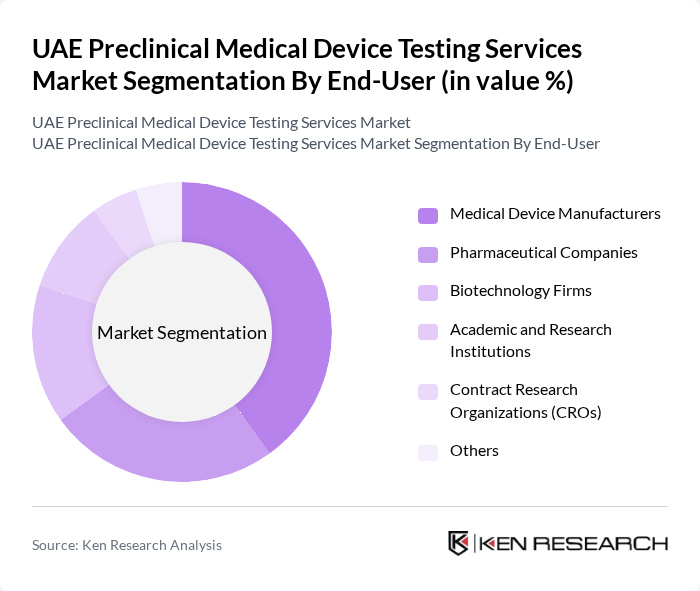
By End-User:This segment categorizes the market based on the end-users of preclinical testing services, which include Medical Device Manufacturers, Pharmaceutical Companies, Biotechnology Firms, Academic and Research Institutions, Contract Research Organizations (CROs), and Others. Medical Device Manufacturers are the leading end-users, driven by the need for comprehensive testing to meet regulatory standards, reduce time-to-market, and ensure product safety and efficacy before market launch, often by outsourcing testing to specialized global and regional service providers.
The UAE Preclinical Medical Device Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Charles River Laboratories, Labcorp Drug Development (including Covance), Eurofins Scientific, Medpace, WuXi AppTec, Syneos Health, PPD (Pharmaceutical Product Development), ICON plc, Q2 Solutions, BioReliance (Merck KGaA), SGS SA, TÜV SÜD, Intertek Group plc, OMC Medical, Qmaizen contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE preclinical medical device testing services market appears promising, driven by advancements in technology and increasing collaboration between industry and academia. The integration of artificial intelligence and machine learning in testing processes is expected to enhance efficiency and accuracy. Additionally, the growing focus on personalized medicine will likely create new avenues for innovation, necessitating robust preclinical testing frameworks to support the development of tailored medical solutions for patients.
| Segment | Sub-Segments |
|---|---|
| By Testing Service Type | In Vivo Testing Services In Vitro Testing Services Biocompatibility Testing Microbiology & Sterility Testing Chemistry and Analytical Testing Toxicology and Safety Testing Others |
| By End-User | Medical Device Manufacturers Pharmaceutical Companies Biotechnology Firms Academic and Research Institutions Contract Research Organizations (CROs) Others |
| By Device Class | Class I Devices Class II Devices Class III Devices Others |
| By Therapeutic Area | Cardiovascular Orthopedic Neurology Oncology Others |
| By Testing Phase | Pre-Clinical Phase Clinical Phase Support Services Post-Market Surveillance Support Others |
| By Region | Abu Dhabi Dubai Sharjah Ajman Others |
| By Service Provider Type | Contract Research Organizations (CROs) Academic and Research Institutions Independent Testing Laboratories In-house Testing Units of Manufacturers Others |
| By Regulatory Compliance Framework | ISO 10993 Compliant ISO 17025 Accredited UAE MOHAP / DoH Abu Dhabi / DHA Requirements CE Marking Requirements FDA and Other International Standards Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Preclinical Testing Facilities | 100 | Laboratory Managers, Quality Assurance Officers |
| Medical Device Manufacturers | 80 | R&D Directors, Regulatory Affairs Managers |
| Clinical Research Organizations | 70 | Project Managers, Clinical Trial Coordinators |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Healthcare Providers | 60 | Medical Directors, Procurement Specialists |
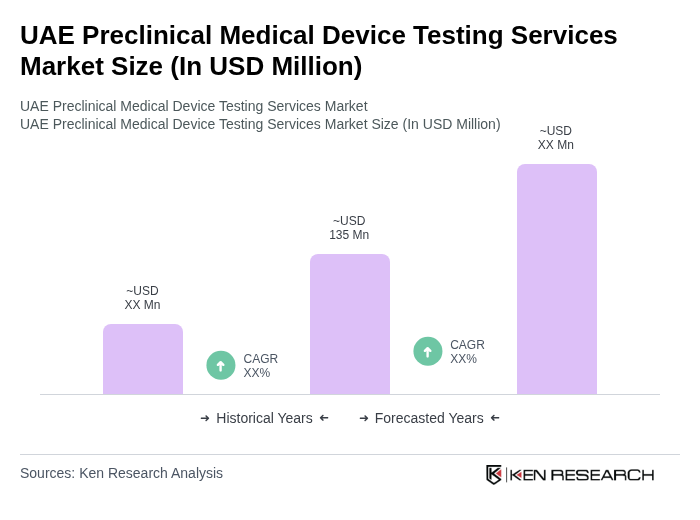
The UAE Preclinical Medical Device Testing Services Market is valued at approximately USD 135 million, reflecting its significant role within the broader medical device testing services market in the Middle East region.