About the Report
Base Year 2024UAE Semi Solid Dosage CDMO Market Overview
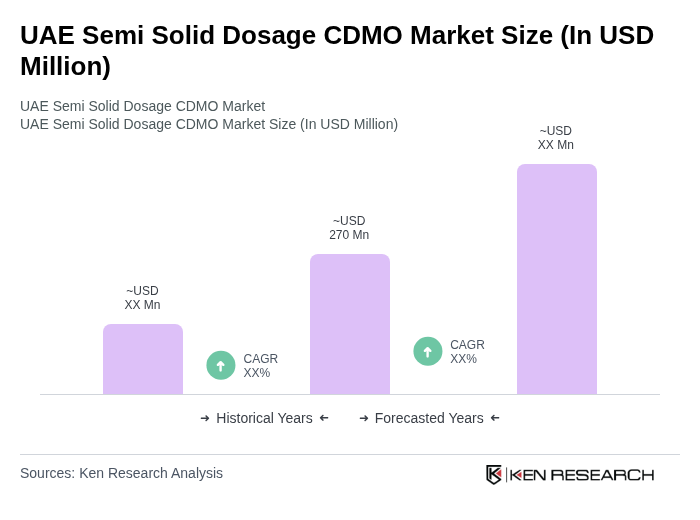
- The UAE Semi Solid Dosage CDMO Market is valued at USD 270 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for semi-solid formulations in the pharmaceutical sector, particularly for dermatological and pain management applications. The rise in chronic diseases and the need for effective topical treatments have significantly contributed to the market's expansion.
- Key players in this market include Dubai, Abu Dhabi, and Sharjah, which dominate due to their advanced healthcare infrastructure, strategic location, and supportive government policies. These cities serve as hubs for pharmaceutical manufacturing and distribution, attracting both local and international companies to establish their operations in the region.
- The UAE Federal Decree-Law No. 38 of 2022 on Pharmaceutical Products and Measures, issued by the Ministry of Health and Prevention (MOHAP), regulates pharmaceutical manufacturing and CDMO activities. This law establishes requirements for licensing, good manufacturing practices (GMP) compliance, quality control standards, and product registration, mandating CDMOs to maintain facilities meeting international GMP thresholds and undergo regular inspections to ensure safety and efficacy in production.
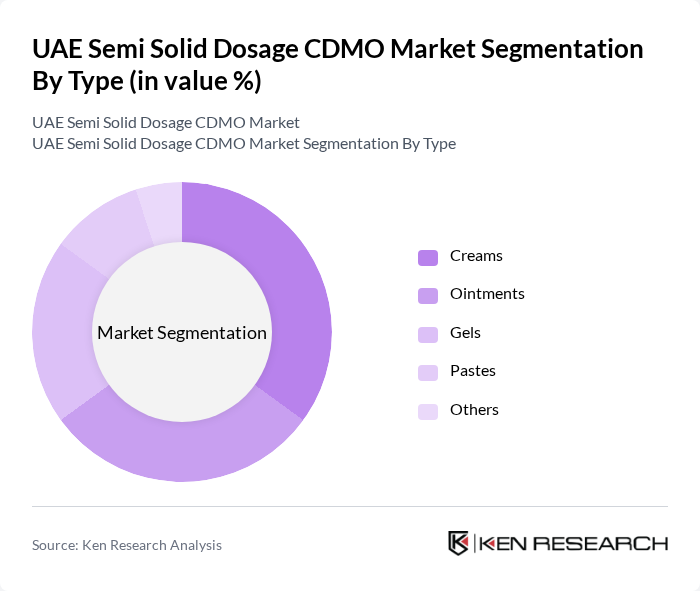
UAE Semi Solid Dosage CDMO Market Segmentation
By Type:The semi-solid dosage forms can be categorized into several types, including creams, ointments, gels, pastes, emulsions, and others. Among these, creams and ointments are the most widely used due to their versatility and effectiveness in delivering active ingredients through the skin. The demand for gels is also rising, particularly in cosmetic applications, while pastes and emulsions are gaining traction in specialized therapeutic areas.
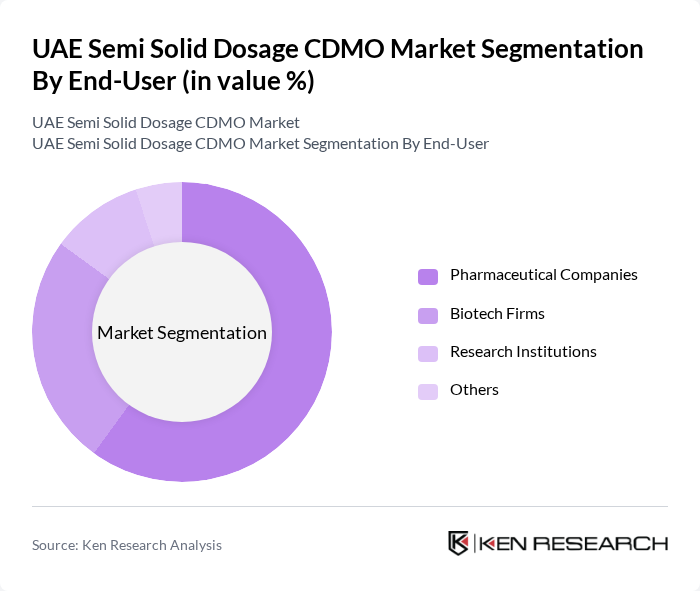
By End-User:The end-users of semi-solid dosage forms include pharmaceutical companies, contract research organizations (CROs), hospitals and clinics, retail pharmacies, and others. Pharmaceutical companies are the largest consumers, driven by the need for effective formulations in various therapeutic areas. Hospitals and clinics also play a significant role, particularly in providing specialized treatments, while retail pharmacies cater to consumer demand for over-the-counter products.
UAE Semi Solid Dosage CDMO Market Competitive Landscape
The UAE Semi Solid Dosage CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Julphar, Neopharma, Gulf Pharmaceutical Industries, Pharma International, Al Haramain Pharmaceutical, Aster DM Healthcare, Emirates Pharmaceuticals, United Pharmacies, Tabuk Pharmaceuticals, Hikma Pharmaceuticals, Bayer Middle East, Sanofi UAE, GSK UAE, Merck Sharp & Dohme, Pfizer UAE contribute to innovation, geographic expansion, and service delivery in this space.
UAE Semi Solid Dosage CDMO Market Industry Analysis
Growth Drivers
- Increasing Demand for Personalized Medicine:The UAE's healthcare expenditure is projected to reach AED 80 billion in future, driven by a growing emphasis on personalized medicine. This trend is fueled by advancements in genomics and biotechnology, leading to a surge in demand for semi-solid formulations tailored to individual patient needs. The increasing prevalence of conditions requiring customized treatments, such as cancer and autoimmune diseases, further supports this growth, creating a robust market for CDMO services in the semi-solid dosage sector.
- Rise in Chronic Diseases Requiring Semi-Solid Formulations:Chronic diseases, including diabetes and cardiovascular disorders, are on the rise in the UAE, with over 30% of the population affected. This increase necessitates effective treatment options, including semi-solid formulations that enhance drug delivery and patient compliance. The UAE's healthcare system is adapting to these needs, with a projected increase in the production of semi-solid dosage forms, thereby driving demand for CDMO services in the region.
- Expansion of Pharmaceutical Manufacturing Capabilities:The UAE government has invested AED 1.5 billion in enhancing local pharmaceutical manufacturing capabilities, aiming to reduce dependency on imports. This initiative is expected to increase the production of semi-solid dosage forms, aligning with the country's vision to become a global hub for pharmaceutical manufacturing. The establishment of new manufacturing facilities and partnerships with international firms will further bolster the CDMO market, providing a competitive edge in the region.
Market Challenges
- Stringent Regulatory Requirements:The UAE's pharmaceutical sector faces rigorous regulatory scrutiny, with the Ministry of Health and Prevention enforcing strict compliance with Good Manufacturing Practices (GMP). This regulatory landscape can pose challenges for CDMOs, as obtaining necessary approvals for semi-solid formulations can be time-consuming and costly. Companies must invest significantly in quality assurance and regulatory affairs to navigate these complexities, impacting operational efficiency and profitability.
- High Competition from Established Players:The UAE semi-solid dosage CDMO market is characterized by intense competition, with established players holding significant market share. Companies like Julphar and Neopharma dominate the landscape, leveraging their extensive experience and resources. New entrants may struggle to gain traction due to brand loyalty and established distribution networks, making it challenging to penetrate the market and achieve sustainable growth in this competitive environment.
UAE Semi Solid Dosage CDMO Market Future Outlook
The future of the UAE semi-solid dosage CDMO market appears promising, driven by ongoing advancements in pharmaceutical technologies and a growing focus on patient-centric solutions. As the demand for personalized medicine continues to rise, CDMOs are likely to invest in innovative formulation techniques and digital manufacturing technologies. Additionally, collaborations with biotech firms will enhance research and development capabilities, positioning the UAE as a key player in the regional pharmaceutical landscape, fostering growth and innovation in the sector.
Market Opportunities
- Growth in Export Potential to Neighboring Regions:The UAE's strategic location offers significant export opportunities for semi-solid dosage forms to neighboring markets, including Saudi Arabia and Oman. With a combined pharmaceutical market value exceeding AED 30 billion, the potential for cross-border trade is substantial. CDMOs can leverage this opportunity by enhancing production capabilities and establishing partnerships to meet the growing demand in these regions.
- Increasing Investment in R&D for Innovative Formulations:The UAE government is prioritizing research and development, allocating AED 2 billion towards innovation in pharmaceuticals. This investment is expected to drive the development of novel semi-solid formulations, enhancing therapeutic efficacy and patient adherence. CDMOs that focus on R&D will be well-positioned to capitalize on emerging trends and meet the evolving needs of the healthcare market, fostering long-term growth.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Creams Ointments Gels Pastes Emulsions Others |
| By End-User | Pharmaceutical Companies Contract Research Organizations Hospitals and Clinics Retail Pharmacies Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Packaging Type | Tubes Jars Sachets Others |
| By Therapeutic Area | Dermatology Pain Management Hormonal Treatments Others |
| By Formulation Complexity | Simple Formulations Complex Formulations Others |
| By Regulatory Compliance Level | FDA Approved EMA Approved Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health and Prevention, Emirates Authority for Standardization and Metrology)
Pharmaceutical Manufacturers
Contract Development and Manufacturing Organizations (CDMOs)
Logistics and Supply Chain Companies
Pharmaceutical Distributors
Healthcare Providers and Hospitals
Industry Associations (e.g., UAE Pharmaceutical Association)
Players Mentioned in the Report:
Julphar
Neopharma
Gulf Pharmaceutical Industries
Pharma International
Al Haramain Pharmaceutical
Aster DM Healthcare
Emirates Pharmaceuticals
United Pharmacies
Tabuk Pharmaceuticals
Hikma Pharmaceuticals
Bayer Middle East
Sanofi UAE
GSK UAE
Merck Sharp & Dohme
Pfizer UAE
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. UAE Semi Solid Dosage CDMO Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 UAE Semi Solid Dosage CDMO Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. UAE Semi Solid Dosage CDMO Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for personalized medicine
3.1.2 Rise in chronic diseases requiring semi-solid formulations
3.1.3 Expansion of pharmaceutical manufacturing capabilities
3.1.4 Government initiatives to boost local production
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High competition from established players
3.2.3 Limited access to advanced technologies
3.2.4 Fluctuating raw material prices
3.3 Market Opportunities
3.3.1 Growth in export potential to neighboring regions
3.3.2 Increasing investment in R&D for innovative formulations
3.3.3 Collaborations with biotech firms
3.3.4 Adoption of digital technologies in manufacturing
3.4 Market Trends
3.4.1 Shift towards sustainable and eco-friendly production
3.4.2 Increasing focus on patient-centric drug delivery systems
3.4.3 Rise of contract manufacturing partnerships
3.4.4 Integration of AI and automation in production processes
3.5 Government Regulation
3.5.1 Implementation of GMP standards
3.5.2 Regulatory approvals for new drug formulations
3.5.3 Policies promoting local manufacturing
3.5.4 Compliance with international quality standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. UAE Semi Solid Dosage CDMO Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. UAE Semi Solid Dosage CDMO Market Segmentation
8.1 By Type
8.1.1 Creams
8.1.2 Ointments
8.1.3 Gels
8.1.4 Pastes
8.1.5 Emulsions
8.1.6 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Contract Research Organizations
8.2.3 Hospitals and Clinics
8.2.4 Retail Pharmacies
8.2.5 Others
8.3 By Distribution Channel
8.3.1 Direct Sales
8.3.2 Distributors
8.3.3 Online Sales
8.3.4 Others
8.4 By Packaging Type
8.4.1 Tubes
8.4.2 Jars
8.4.3 Sachets
8.4.4 Others
8.5 By Therapeutic Area
8.5.1 Dermatology
8.5.2 Pain Management
8.5.3 Hormonal Treatments
8.5.4 Others
8.6 By Formulation Complexity
8.6.1 Simple Formulations
8.6.2 Complex Formulations
8.6.3 Others
8.7 By Regulatory Compliance Level
8.7.1 FDA Approved
8.7.2 EMA Approved
8.7.3 Others
9. UAE Semi Solid Dosage CDMO Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Production Efficiency
9.2.7 Quality Compliance Rate
9.2.8 Pricing Strategy
9.2.9 Innovation Rate
9.2.10 Supply Chain Efficiency
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Julphar
9.5.2 Neopharma
9.5.3 Gulf Pharmaceutical Industries
9.5.4 Pharma International
9.5.5 Al Haramain Pharmaceutical
9.5.6 Aster DM Healthcare
9.5.7 Emirates Pharmaceuticals
9.5.8 United Pharmacies
9.5.9 Tabuk Pharmaceuticals
9.5.10 Hikma Pharmaceuticals
9.5.11 Bayer Middle East
9.5.12 Sanofi UAE
9.5.13 GSK UAE
9.5.14 Merck Sharp & Dohme
9.5.15 Pfizer UAE
10. UAE Semi Solid Dosage CDMO Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health and Prevention
10.1.2 Ministry of Industry and Advanced Technology
10.1.3 Ministry of Economy
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.2.3 R&D Expenditure
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Healthcare Providers
10.3.3 Regulatory Bodies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Semi Solid Dosage Forms
10.4.2 Training and Support Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Case Opportunities
10.5.4 Others
11. UAE Semi Solid Dosage CDMO Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from regulatory bodies such as the UAE Ministry of Health and Prevention
- Review of market studies published by pharmaceutical associations and trade organizations in the UAE
- Examination of academic journals and publications focusing on semi-solid dosage forms and CDMO trends
Primary Research
- Interviews with key opinion leaders in the pharmaceutical manufacturing sector
- Surveys conducted with R&D heads at major pharmaceutical companies operating in the UAE
- Field interviews with quality assurance managers at contract development and manufacturing organizations (CDMOs)
Validation & Triangulation
- Cross-validation of findings through multiple data sources including trade publications and expert insights
- Triangulation of market data with sales figures and production capacity reports from industry players
- Sanity checks performed through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall pharmaceutical market size in the UAE and its growth trajectory
- Segmentation of the market by product type, focusing on semi-solid dosage forms
- Incorporation of government healthcare initiatives and their impact on market growth
Bottom-up Modeling
- Collection of production volume data from leading CDMOs in the UAE
- Cost analysis based on pricing models of semi-solid dosage forms
- Estimation of market share based on the number of contracts and partnerships within the sector
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future market trends based on historical data
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing Insights | 120 | Production Managers, Quality Control Analysts |
| Regulatory Compliance in CDMO | 100 | Regulatory Affairs Specialists, Compliance Officers |
| Market Trends in Semi-Solid Dosage Forms | 90 | Market Analysts, Product Development Managers |
| Supply Chain Dynamics in Pharmaceuticals | 110 | Supply Chain Managers, Procurement Directors |
| Consumer Preferences for Semi-Solid Products | 80 | Consumer Insights Analysts, Marketing Managers |
Frequently Asked Questions
What is the current value of the UAE Semi Solid Dosage CDMO Market?
The UAE Semi Solid Dosage CDMO Market is valued at approximately USD 270 million, reflecting a significant growth driven by the increasing demand for semi-solid formulations, particularly in dermatological and pain management applications.