Region:Middle East
Author(s):Dev
Product Code:KRAC3384
Pages:88
Published On:October 2025
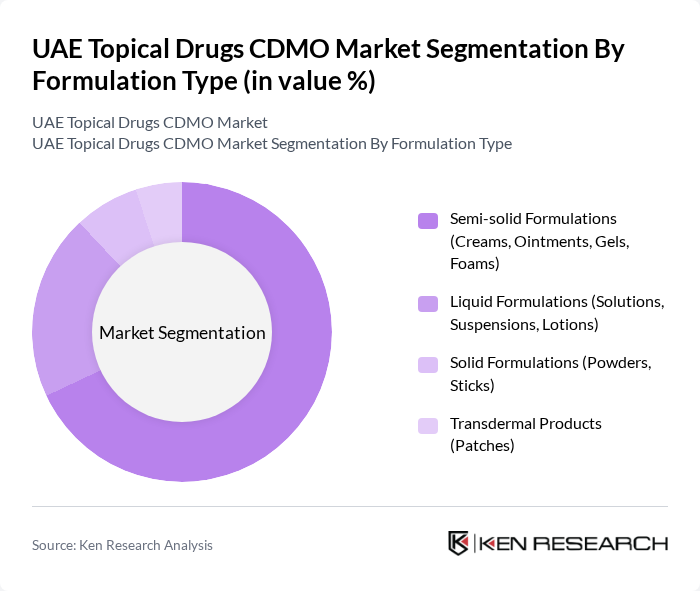
By Formulation Type:The formulation type segment includes various sub-segments such as semi-solid formulations, liquid formulations, solid formulations, and transdermal products. Among these, semi-solid formulations—which include creams, ointments, gels, and foams—dominate the market, accounting for the largest revenue share. Their versatility and effectiveness in delivering active ingredients through the skin, combined with the increasing prevalence of skin disorders and demand for effective topical treatments, drive the growth of this sub-segment. Recent industry data indicates semi-solid formulations represent over two-thirds of market revenue, reflecting their continued dominance .
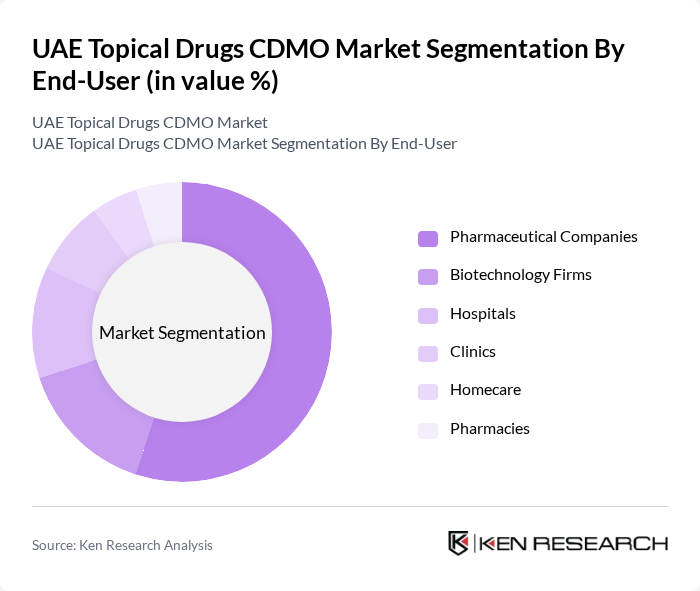
By End-User:The end-user segment encompasses pharmaceutical companies, biotechnology firms, hospitals, clinics, homecare, and pharmacies. Pharmaceutical companies are the leading end-users, driven by their need for contract development and manufacturing services to expedite the production of topical drugs. The increasing focus on research and development, outsourcing, and rapid commercialization in the pharmaceutical sector further propels demand for CDMO services. Hospitals and clinics also represent significant end-users due to the rising adoption of topical therapies for chronic and acute conditions .
The UAE Topical Drugs CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Julphar (Gulf Pharmaceutical Industries), Neopharma, Pharmax Pharmaceuticals, Medpharma, LifePharma FZE, United Pharmaceuticals, Tabuk Pharmaceuticals, Hikma Pharmaceuticals, Bayer Middle East, Pfizer UAE, Sanofi UAE, GSK UAE, Merck Sharp & Dohme, Amgen, AbbVie, Catalent Pharma Solutions, Lubrizol Life Science, Piramal Pharma Solutions, MedPharm, Contract Pharmaceuticals Limited contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE topical drugs CDMO market appears promising, driven by technological advancements and a growing focus on personalized medicine. As the demand for customized dermatological solutions increases, CDMOs are likely to invest in advanced manufacturing technologies to enhance efficiency and product quality. Additionally, the expansion of e-commerce platforms for pharmaceutical products is expected to facilitate broader market access, enabling CDMOs to reach a wider consumer base and adapt to changing market dynamics effectively.
| Segment | Sub-Segments |
|---|---|
| By Formulation Type | Semi-solid Formulations (Creams, Ointments, Gels, Foams) Liquid Formulations (Solutions, Suspensions, Lotions) Solid Formulations (Powders, Sticks) Transdermal Products (Patches) |
| By End-User | Pharmaceutical Companies Biotechnology Firms Hospitals Clinics Homecare Pharmacies |
| By Workflow | Clinical Development Commercial Manufacturing |
| By Application | Dermatology Pain Management Hormonal Treatments Antiseptics Wound Healing |
| By Packaging Type | Tubes Bottles Sachets Jars Pouches |
| By Regulatory Compliance | UAE Ministry of Health Approved FDA Approved EMA Approved Local Regulatory Compliance |
| By Price Range | Low Medium High |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing Insights | 100 | Production Managers, Quality Assurance Heads |
| Regulatory Compliance Challenges | 80 | Regulatory Affairs Managers, Compliance Officers |
| Market Entry Strategies for CDMOs | 60 | Business Development Managers, Strategy Directors |
| Trends in Biopharmaceuticals | 50 | R&D Directors, Product Development Managers |
| Contract Negotiation Practices | 70 | Procurement Managers, Legal Advisors |
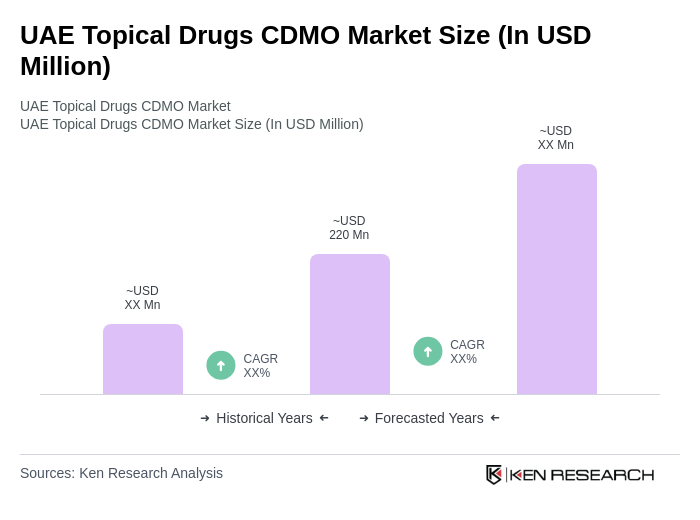
The UAE Topical Drugs CDMO Market is valued at approximately USD 220 million, reflecting a significant growth driven by increasing demand for topical formulations in dermatology, pain management, and wound care, as well as advancements in drug delivery technologies.