Region:North America
Author(s):Dev
Product Code:KRAD4534
Pages:89
Published On:December 2025
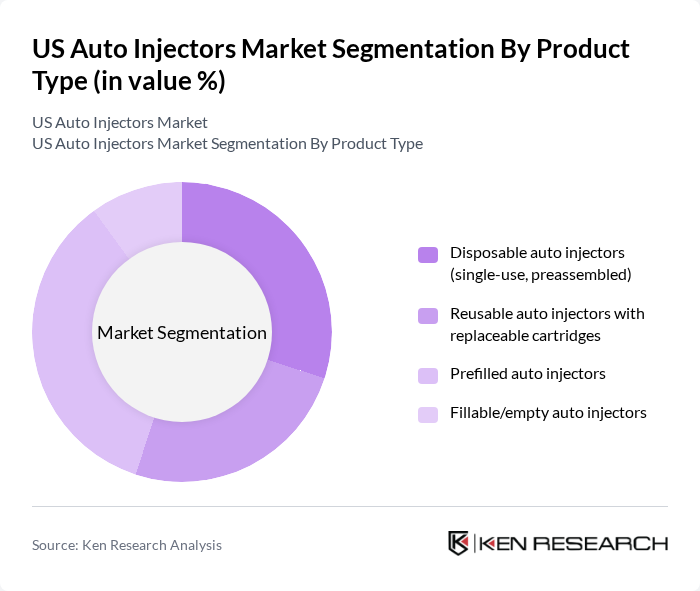
By Product Type:The product type segmentation includes various categories of auto injectors that cater to different consumer needs. The dominant subsegment is the prefilled auto injectors, which are favored for their ease of use and convenience. Disposable auto injectors are also gaining traction due to their single-use nature, which minimizes the risk of contamination. Reusable auto injectors with replaceable cartridges are popular among patients who require long-term treatment, while fillable/empty auto injectors are less common but serve niche markets.
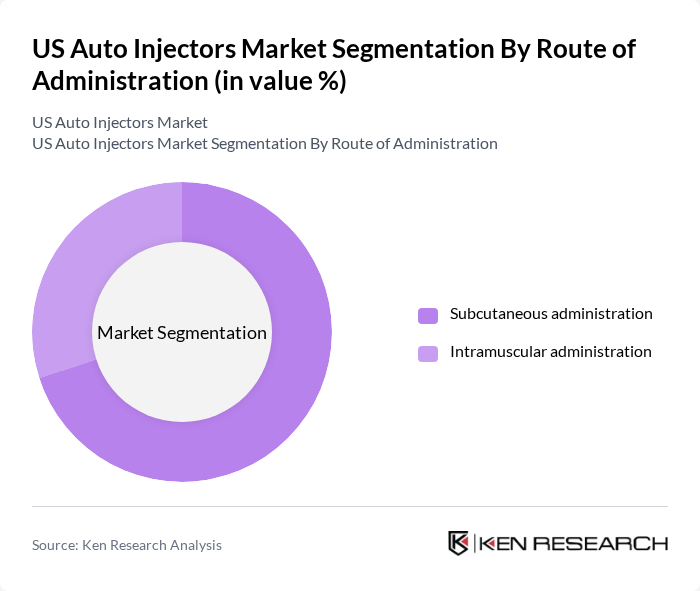
By Route of Administration:The route of administration segmentation highlights the methods through which auto injectors deliver medication. Subcutaneous administration is the leading method, preferred for its ease and effectiveness in delivering various medications, including insulin and epinephrine. Intramuscular administration is also significant, particularly for vaccines and certain hormone therapies. The preference for subcutaneous routes is driven by patient comfort and the ability to self-administer.
The US Auto Injectors Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Viatris Inc. (Mylan), Sanofi S.A., Eli Lilly and Company, Teva Pharmaceutical Industries Ltd., AbbVie Inc., Novartis AG, Bayer AG, Johnson & Johnson (Janssen / J&J Innovative Medicine), GlaxoSmithKline plc (GSK), Regeneron Pharmaceuticals, Inc., Hikma Pharmaceuticals PLC, Sandoz Group AG, Pfizer Inc., Becton, Dickinson and Company (BD) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the U.S. auto injectors market appears promising, driven by ongoing technological advancements and increasing consumer awareness. As digital health solutions gain traction, the integration of smart technology into auto injectors is expected to enhance user experience and adherence. Furthermore, the rise of telehealth services will facilitate remote consultations, allowing patients to receive timely prescriptions and education on auto injector usage, ultimately improving health outcomes and market penetration.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Disposable auto injectors (single-use, preassembled) Reusable auto injectors with replaceable cartridges Prefilled auto injectors Fillable/empty auto injectors |
| By Route of Administration | Subcutaneous administration Intramuscular administration |
| By Therapy Area | Anaphylaxis and severe allergies (e.g., epinephrine) Autoimmune diseases (e.g., rheumatoid arthritis, multiple sclerosis) Hormonal and fertility therapies Other chronic conditions (e.g., migraine, osteoporosis, oncology) |
| By End User | Homecare settings / self-administration Hospitals Clinics and physician offices Emergency medical services |
| By Distribution Channel | Hospital pharmacies Retail pharmacies and drug stores Online pharmacies Others |
| By Region | Northeast Midwest South West |
| By Patient Demographics | Pediatric patients Adult patients Geriatric patients Others |
| By Technology | Mechanical (spring-driven) auto injectors Electronic auto injectors Connected / smart auto injectors (with digital features) Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Diabetes Auto Injector Users | 140 | Patients, Endocrinologists, Diabetes Educators |
| Allergy Treatment Auto Injector Users | 100 | Patients, Allergists, Pharmacists |
| Rheumatoid Arthritis Auto Injector Users | 80 | Patients, Rheumatologists, Nurse Practitioners |
| Healthcare Professionals in Clinics | 120 | General Practitioners, Nurse Practitioners, Physician Assistants |
| Pharmacy Managers and Staff | 90 | Pharmacy Managers, Pharmacists, Pharmacy Technicians |
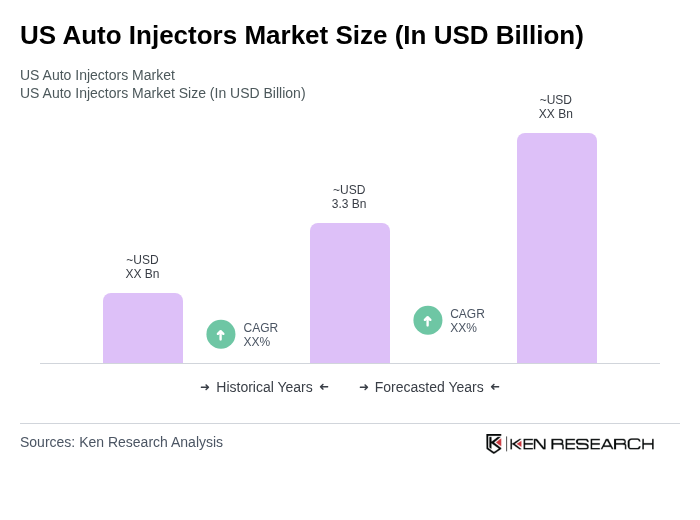
The US Auto Injectors Market is valued at approximately USD 3.3 billion, reflecting significant growth driven by the increasing prevalence of chronic diseases and the rising demand for self-administration devices.