US Blood Based Biomarkers Market Overview
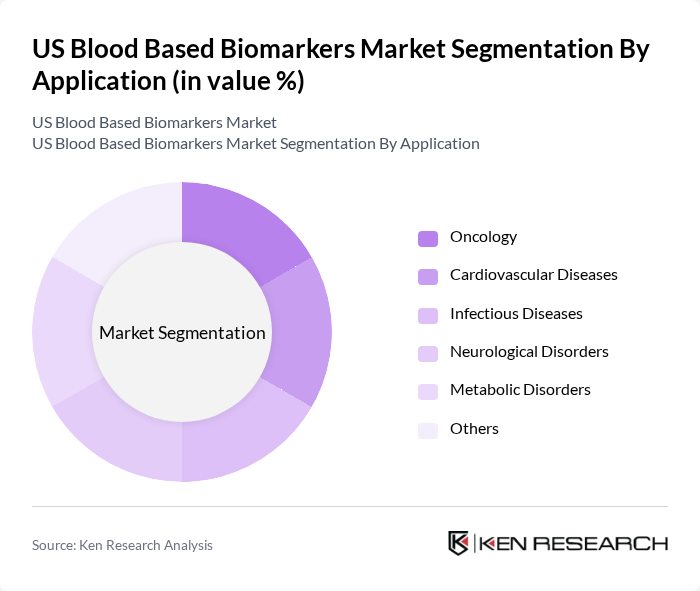
- The US Blood Based Biomarkers Market is valued at USD 3 billion, based on a five-year historical analysis. This growth is primarily driven by advancements in diagnostic technologies, increasing prevalence of chronic diseases, and a growing emphasis on personalized medicine. The rising demand for early disease detection and monitoring has further propelled the market, making blood-based biomarkers a critical component in healthcare diagnostics. Recent trends include the rapid adoption of next-generation sequencing and polymerase chain reaction technologies, as well as the integration of artificial intelligence for biomarker discovery and validation. The shift toward non-invasive diagnostics and the expansion of liquid biopsy applications in oncology are also significant growth drivers.
- Key players in this market include major cities such as New York, San Francisco, and Boston, which dominate due to their robust healthcare infrastructure, presence of leading research institutions, and a high concentration of biotechnology firms. These regions foster innovation and collaboration, driving the development and adoption of blood-based biomarker technologies.
- The 21st Century Cures Act, enacted by the US Congress in 2016 and administered by the Food and Drug Administration (FDA), aims to accelerate medical product development and bring new innovations to patients faster. This legislation specifically encourages the use of biomarkers, including those derived from blood, in clinical trials and diagnostics. It establishes frameworks for the qualification of biomarkers, streamlines regulatory pathways for biomarker-based tests, and supports the incorporation of real-world evidence in the approval process for new diagnostics and therapies. Compliance involves meeting FDA biomarker qualification program requirements, which include rigorous validation and evidentiary standards for clinical utility.
US Blood Based Biomarkers Market Segmentation
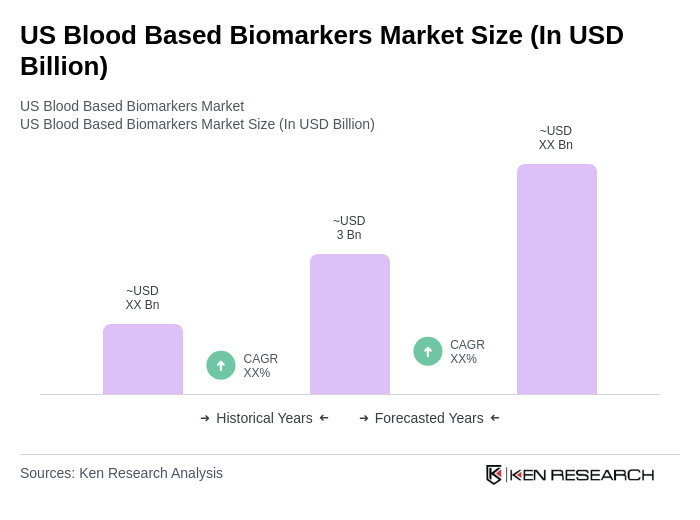
By Type:The market is segmented into various types of biomarkers, including Genetic Biomarkers, Protein Biomarkers, Metabolic Biomarkers, Cell-Based Biomarkers, and Epigenetic Biomarkers. Among these, Genetic Biomarkers currently lead the market in terms of revenue, driven by their widespread use in precision medicine and the increasing availability of advanced genomic testing platforms. Protein Biomarkers remain critical for disease diagnosis and monitoring, particularly in oncology and cardiovascular diseases, while Cell-Based Biomarkers are the fastest-growing segment due to innovations in liquid biopsy and single-cell analysis technologies. The focus on precision medicine and the development of targeted therapies continue to drive demand across all biomarker types.
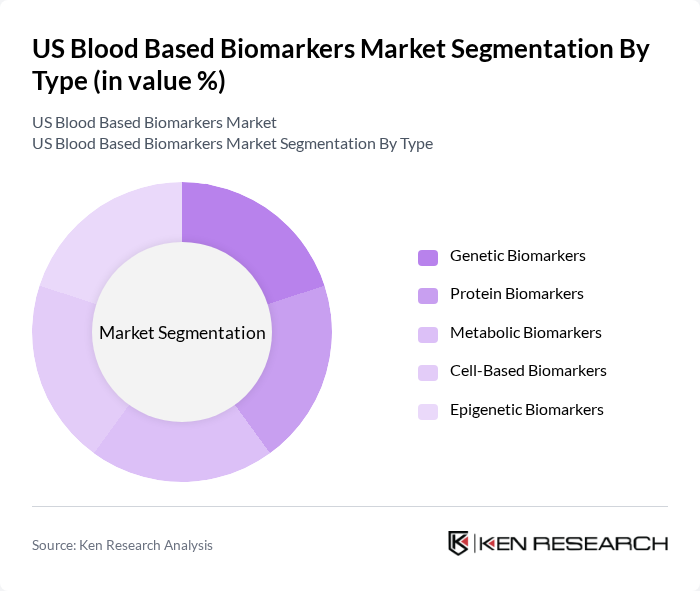
By Application:The applications of blood-based biomarkers include Oncology, Cardiovascular Diseases, Infectious Diseases, Neurological Disorders, Metabolic Disorders, and Others. Oncology is the leading application area, driven by the increasing incidence of cancer and the need for early detection and personalized treatment options. The growing adoption of liquid biopsies and advancements in biomarker discovery are also contributing to the dominance of oncology in this segment. Cardiovascular diseases represent another major application, supported by the rising burden of heart disease and the development of novel cardiac biomarkers. Neurological and metabolic disorders are emerging areas, with increasing research into biomarkers for Alzheimer’s disease, diabetes, and other chronic conditions.
US Blood Based Biomarkers Market Competitive Landscape
The US Blood Based Biomarkers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, QIAGEN N.V., Illumina, Inc., Myriad Genetics, Inc., Guardant Health, Inc., Exact Sciences Corporation, Genomic Health, Inc., Luminex Corporation, Veracyte, Inc., Foundation Medicine, Inc., Biocept, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
US Blood Based Biomarkers Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The US is witnessing a significant rise in chronic diseases, with approximately 60% of adults living with at least one chronic condition in the future. This trend is driven by an aging population and lifestyle factors, leading to a projected increase in healthcare spending, which is expected to reach $4.6 trillion. This growing patient population is driving demand for blood-based biomarkers, which are essential for early diagnosis and management of these diseases.
- Advancements in Diagnostic Technologies:The US blood-based biomarkers market is benefiting from rapid advancements in diagnostic technologies, with the market for diagnostic devices projected to reach $110 billion in the future. Innovations such as next-generation sequencing and high-throughput screening are enhancing the accuracy and efficiency of biomarker detection. These technological improvements are crucial for the development of more precise and reliable blood tests, thereby increasing their adoption in clinical settings.
- Rising Demand for Personalized Medicine:The shift towards personalized medicine is reshaping the healthcare landscape, with the personalized medicine market expected to reach $2.7 trillion in the future. Blood-based biomarkers play a pivotal role in tailoring treatments to individual patients, particularly in oncology and chronic disease management. This trend is supported by increasing investments in research and development, which are projected to exceed $210 billion in the US, further driving the demand for innovative biomarker solutions.
Market Challenges
- High Costs of Biomarker Development:The development of blood-based biomarkers is often hindered by high costs, with estimates suggesting that the average cost to bring a new biomarker to market can exceed $1.1 billion. This financial burden is exacerbated by the lengthy research and validation processes, which can take over a decade. As a result, many smaller biotech firms struggle to secure funding, limiting innovation and market entry for new biomarker tests.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape poses significant challenges for blood-based biomarker developers. The FDA's stringent guidelines for biomarker validation require extensive clinical data, which can delay product launches. In the future, it is estimated that over 32% of biomarker applications face regulatory setbacks, leading to increased costs and time-to-market. These hurdles can deter investment and slow the pace of innovation in the sector.
US Blood Based Biomarkers Market Future Outlook
The future of the US blood-based biomarkers market appears promising, driven by technological advancements and a growing emphasis on personalized healthcare. As non-invasive testing methods gain traction, the integration of artificial intelligence in biomarker discovery is expected to enhance diagnostic accuracy. Furthermore, the increasing focus on multi-omics approaches will likely lead to more comprehensive biomarker profiles, improving patient outcomes and fostering innovation in the industry. These trends indicate a dynamic and evolving market landscape.
Market Opportunities
- Expansion of Biomarker Applications in Oncology:The oncology sector presents significant opportunities for blood-based biomarkers, with an estimated 2.1 million new cancer cases expected in the US in the future. This growing patient population is driving demand for innovative biomarker tests that can aid in early detection and treatment monitoring, creating a lucrative market for developers.
- Collaborations Between Biotech Firms and Research Institutions:Strategic partnerships between biotech companies and research institutions are on the rise, with over 220 collaborations reported in the future. These alliances facilitate knowledge sharing and resource pooling, accelerating the development of novel biomarkers. Such collaborations are essential for driving innovation and expanding the application of blood-based biomarkers across various therapeutic areas.