Region:North America
Author(s):Rebecca
Product Code:KRAC3871
Pages:87
Published On:October 2025
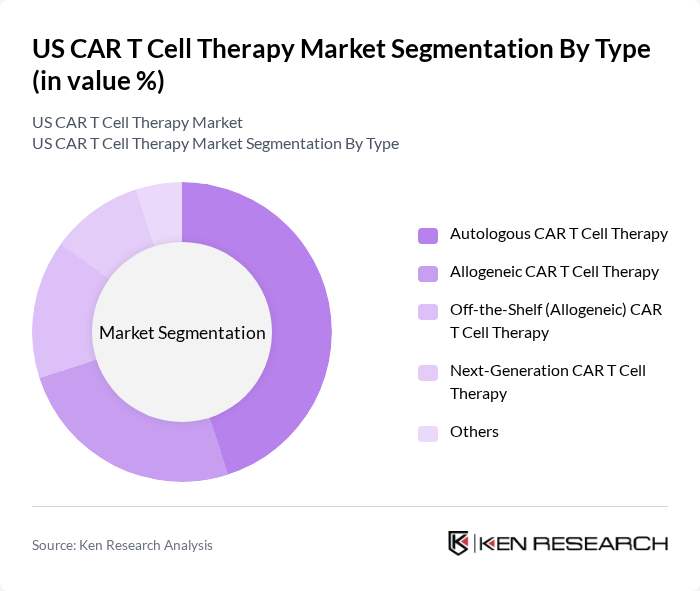
By Type:The market is segmented into various types of CAR T cell therapies, including Autologous CAR T Cell Therapy, Allogeneic CAR T Cell Therapy, Off-the-Shelf (Allogeneic) CAR T Cell Therapy, Next-Generation CAR T Cell Therapy, and Others. Autologous CAR T Cell Therapy is currently the leading segment due to its personalized approach, where a patient's own T cells are modified to target cancer cells, resulting in higher efficacy and lower risk of rejection. Recent advances in allogeneic and off-the-shelf CAR T therapies, including CRISPR-based gene editing, are expanding access and reducing manufacturing time, but autologous therapies remain dominant in clinical practice .
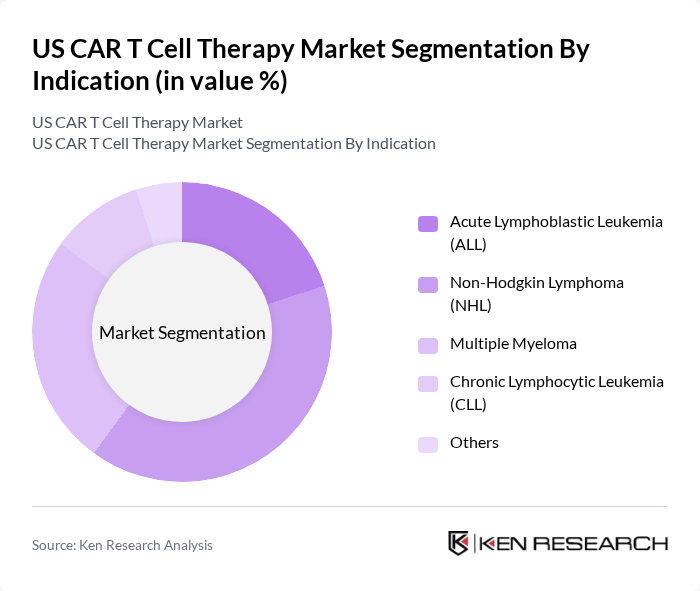
By Indication:The market is categorized based on indications such as Acute Lymphoblastic Leukemia (ALL), Non-Hodgkin Lymphoma (NHL), Multiple Myeloma, Chronic Lymphocytic Leukemia (CLL), and Others. Non-Hodgkin Lymphoma is the dominant indication due to the high incidence rate and the effectiveness of CAR T therapies in treating this type of cancer, leading to significant improvements in patient outcomes. CAR T therapies have demonstrated strong clinical results in relapsed/refractory NHL and are increasingly used for multiple myeloma and ALL, with ongoing expansion into new indications .
The US CAR T Cell Therapy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Gilead Sciences, Inc. (Kite Pharma, Inc.), Bristol-Myers Squibb Company, Johnson & Johnson (Janssen Biotech, Inc.), Legend Biotech Corporation, Bluebird Bio, Inc., Celyad Oncology SA, Adaptimmune Therapeutics PLC, Precision BioSciences, Inc., Sorrento Therapeutics, Inc., Caribou Biosciences, Inc., Mustang Bio, Inc., Autolus Therapeutics plc, TCR² Therapeutics Inc., Allogene Therapeutics, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the CAR T cell therapy market appears promising, driven by ongoing advancements in technology and increasing research investments. As the industry evolves, there is a notable shift towards combination therapies that enhance treatment efficacy. Additionally, the emergence of off-the-shelf CAR T products is expected to improve accessibility and reduce costs. These trends indicate a dynamic landscape where innovation and collaboration will play pivotal roles in shaping the future of CAR T therapies.
| Segment | Sub-Segments |
|---|---|
| By Type | Autologous CAR T Cell Therapy Allogeneic CAR T Cell Therapy Off-the-Shelf (Allogeneic) CAR T Cell Therapy Next-Generation CAR T Cell Therapy Others |
| By Indication | Acute Lymphoblastic Leukemia (ALL) Non-Hodgkin Lymphoma (NHL) Multiple Myeloma Chronic Lymphocytic Leukemia (CLL) Others |
| By Administration Route | Intravenous Intrathecal Others |
| By Patient Age Group | Pediatric Adult Geriatric |
| By Treatment Setting | Inpatient Outpatient Home Care |
| By End-Use | Hospitals Cancer Treatment Centers Specialty Clinics |
| By Region | Northeast Midwest South West |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists Specializing in CAR T Cell Therapy | 60 | Hematologists, Medical Oncologists |
| Hospital Administrators in Oncology Departments | 40 | Chief Medical Officers, Procurement Managers |
| Patients Receiving CAR T Cell Therapy | 50 | Current Patients, Caregivers |
| Clinical Trial Coordinators | 45 | Research Nurses, Clinical Research Associates |
| Payers and Insurance Representatives | 40 | Health Insurance Analysts, Reimbursement Specialists |
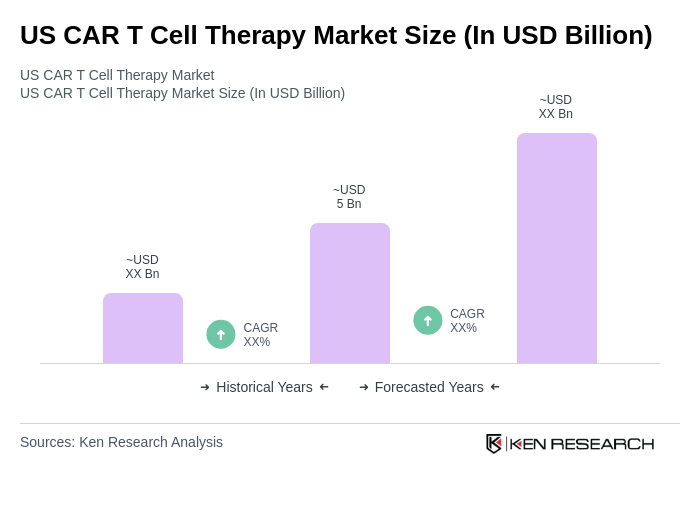
The US CAR T Cell Therapy market is valued at approximately USD 5 billion, driven by the rising prevalence of hematological malignancies and advancements in CAR T cell technology, including gene editing and automation.