Region:North America
Author(s):Dev
Product Code:KRAC2709
Pages:90
Published On:October 2025
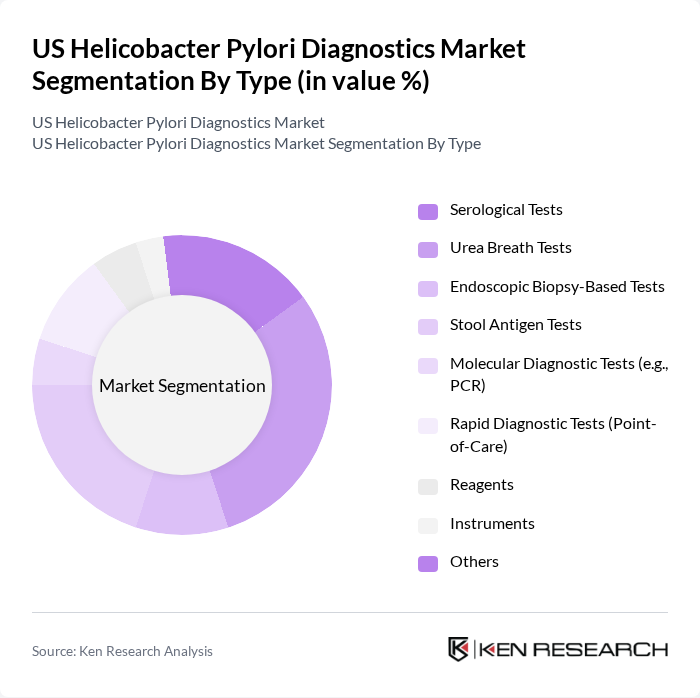
By Type:The market is segmented into various types of diagnostic tests, each catering to different diagnostic needs and preferences. The subsegments include Serological Tests, Urea Breath Tests, Endoscopic Biopsy-Based Tests, Stool Antigen Tests, Molecular Diagnostic Tests (e.g., PCR), Rapid Diagnostic Tests (Point-of-Care), Reagents, Instruments, and Others. Among these, Urea Breath Tests and Stool Antigen Tests are gaining significant traction due to their non-invasive nature and high accuracy, making them a preferred choice for both patients and healthcare providers. Molecular diagnostics, such as PCR, are increasingly adopted for their enhanced sensitivity and specificity, especially in cases of antibiotic resistance .
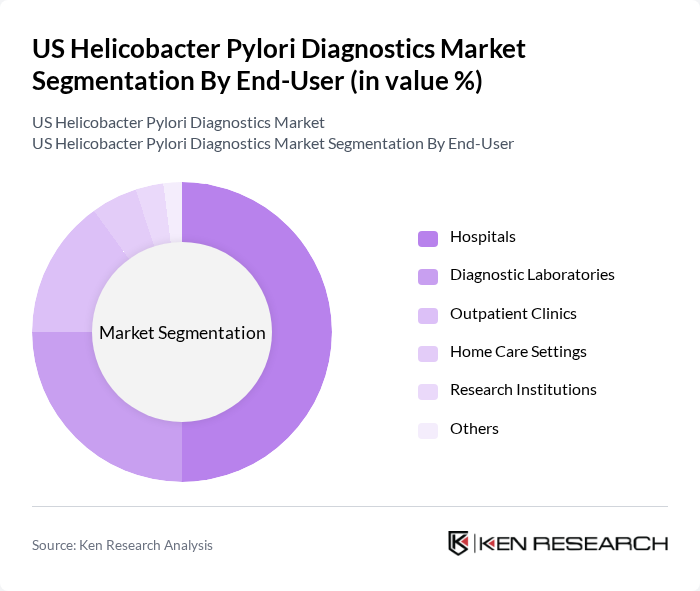
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Outpatient Clinics, Home Care Settings, Research Institutions, and Others. Hospitals are the leading end-users due to their comprehensive diagnostic capabilities and the high volume of patients requiring H. pylori testing. Diagnostic laboratories also hold a significant share, driven by the demand for specialized testing and the availability of advanced equipment. The increasing number of outpatient procedures and the shift towards home care settings are contributing to the growth of these segments. Research institutions play a key role in innovation and clinical validation of new diagnostic methods .
The US Helicobacter Pylori Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Thermo Fisher Scientific Inc., Hologic, Inc., Siemens Healthineers, Roche Diagnostics, Bio-Rad Laboratories, Inc., Cepheid, Becton, Dickinson and Company, QuidelOrtho Corporation, Meridian Bioscience, Inc., GenMark Diagnostics, Inc., Eiken Chemical Co., Ltd., DiaSorin S.p.A., Grifols S.A., Quest Diagnostics Incorporated, Biomerica, Inc., Coris BioConcept, Certest Biotec S.L. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the US Helicobacter Pylori diagnostics market appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the expansion of telemedicine services will facilitate remote consultations and diagnostics. As healthcare providers increasingly focus on personalized medicine, tailored diagnostic solutions will likely emerge, further propelling market growth and improving patient care.
| Segment | Sub-Segments |
|---|---|
| By Type | Serological Tests Urea Breath Tests Endoscopic Biopsy-Based Tests Stool Antigen Tests Molecular Diagnostic Tests (e.g., PCR) Rapid Diagnostic Tests (Point-of-Care) Reagents Instruments Others |
| By End-User | Hospitals Diagnostic Laboratories Outpatient Clinics Home Care Settings Research Institutions Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Others |
| By Region | Northeast Midwest South West Others |
| By Patient Demographics | Adults Children Elderly Others |
| By Test Complexity | Simple Tests Moderate Complexity Tests High Complexity Tests Others |
| By Price Range | Low Price Mid Price High Price Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gastroenterology Clinics | 60 | Gastroenterologists, Clinic Managers |
| Diagnostic Laboratories | 50 | Laboratory Technologists, Quality Control Managers |
| Healthcare Providers | 70 | Primary Care Physicians, Nurse Practitioners |
| Patient Advocacy Groups | 40 | Patient Representatives, Health Educators |
| Pharmaceutical Companies | 40 | Product Managers, Market Access Specialists |
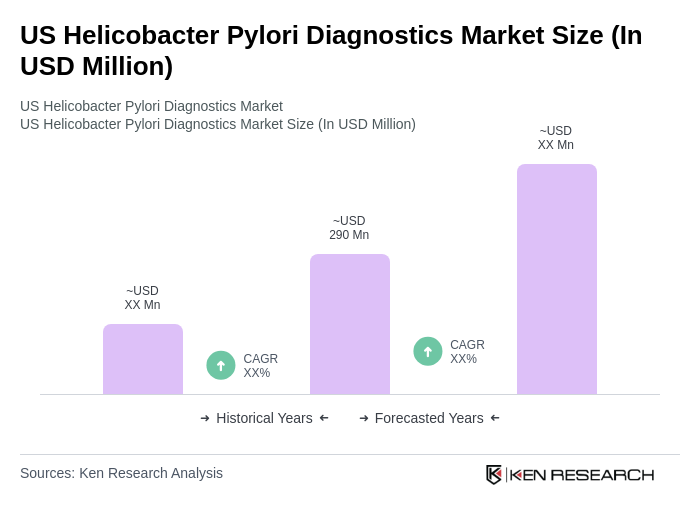
The US Helicobacter Pylori Diagnostics Market is valued at approximately USD 290 million, reflecting a significant growth driven by the increasing prevalence of H. pylori infections and advancements in diagnostic technologies.