Region:North America
Author(s):Geetanshi
Product Code:KRAD4866
Pages:80
Published On:December 2025
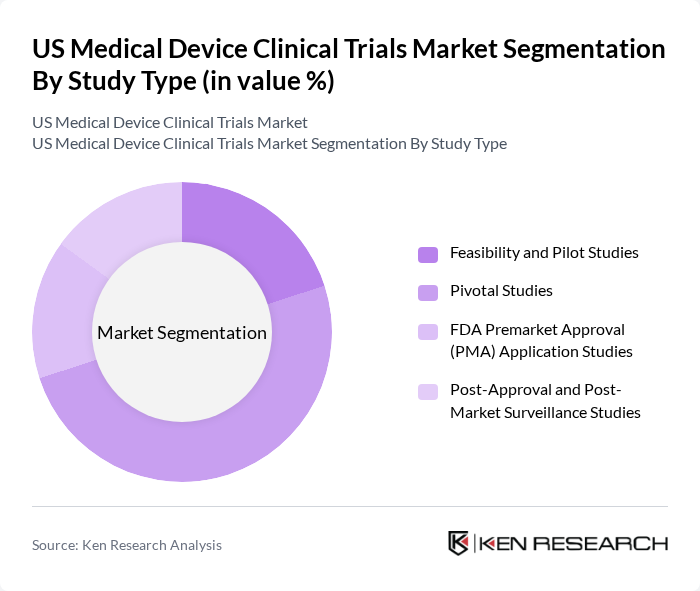
By Study Type:The study type segmentation includes various methodologies employed in clinical trials to evaluate medical devices. The subsegments are Feasibility and Pilot Studies, Pivotal Studies, FDA Premarket Approval (PMA) Application Studies, and Post-Approval and Post-Market Surveillance Studies. Feasibility and pilot studies are gaining momentum as sponsors increasingly use early?phase studies to de?risk complex technologies and refine study protocols, supported by guidance on early feasibility studies for significant risk devices. Pivotal Studies continue to account for a substantial share of spend and enrollment, given their critical role in demonstrating the safety and effectiveness of moderate? and high?risk devices for marketing authorization, especially for implantable, cardiovascular, and orthopedic products. At the same time, the importance of Post-Approval and Post-Market Surveillance Studies has increased, driven by FDA’s emphasis on real?world evidence, active surveillance, and long?term follow?up for certain device types such as implants and digital health technologies.
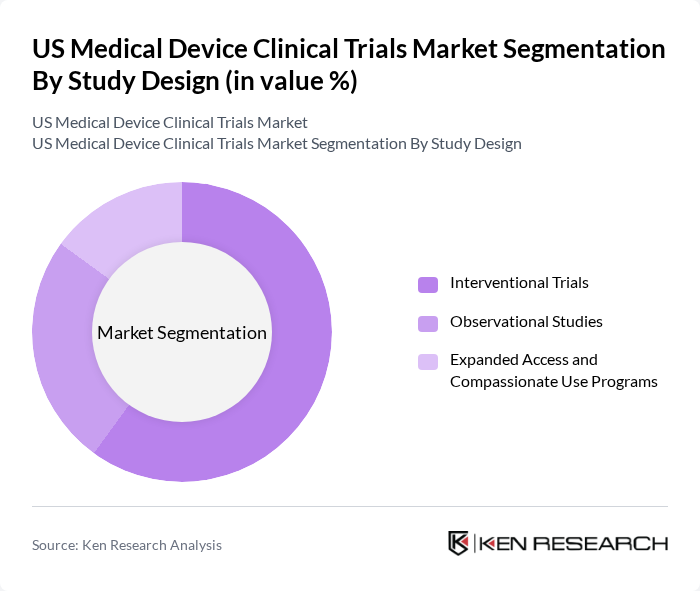
By Study Design:The study design segmentation encompasses the various frameworks used to conduct clinical trials. This includes Interventional Trials, Observational Studies, and Expanded Access and Compassionate Use Programs. Interventional Trials remain the leading subsegment, consistent with broader US clinical trial patterns where interventional designs constitute the majority of studies and revenue. They are essential for testing the efficacy and safety of new medical devices under controlled conditions and increasingly incorporate adaptive designs, Bayesian methods, and decentralized elements such as remote monitoring and eConsent. Observational Studies are expanding as sponsors and regulators place greater emphasis on real?world data, device registries, and long?term performance monitoring, while Expanded Access and Compassionate Use Programs remain a smaller but important mechanism to provide investigational devices to patients with serious or life?threatening conditions who lack alternatives.
The US Medical Device Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, Johnson & Johnson (including Ethicon & DePuy Synthes), Stryker Corporation, Zimmer Biomet Holdings, Inc., Becton, Dickinson and Company (BD), GE HealthCare Technologies Inc., Siemens Healthineers AG, Koninklijke Philips N.V. (Philips Healthcare), 3M Company (3M Health Care), Hologic, Inc., Edwards Lifesciences Corporation, Cook Group Incorporated (Cook Medical), Terumo Corporation, ICON plc (Medical Device-focused CRO), Medpace Holdings, Inc., IQVIA Inc. (Devices and Diagnostics Segment) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the US medical device clinical trials market appears promising, driven by ongoing technological advancements and a growing focus on patient-centric approaches. As the industry embraces decentralized clinical trials and integrates artificial intelligence, efficiency and participant engagement are expected to improve significantly. Furthermore, the increasing emphasis on real-world evidence will likely reshape regulatory submissions, enhancing the overall landscape for medical device development and approval in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Study Type | Feasibility and Pilot Studies Pivotal Studies FDA Premarket Approval (PMA) Application Studies Post-Approval and Post-Market Surveillance Studies |
| By Study Design | Interventional Trials Observational Studies Expanded Access and Compassionate Use Programs |
| By Device Class (FDA Classification) | Class I Devices (Low Risk) Class II Devices (Moderate Risk) Class III Devices (High Risk, Life-Sustaining/Life-Supporting) |
| By Therapeutic Area / Indication | Cardiovascular Devices Orthopedic and Musculoskeletal Devices Neurology and Neurovascular Devices Diagnostic Imaging and In Vitro Diagnostic (IVD) Devices Anesthesia, Respiratory, and Critical Care Devices Other Indications (Ophthalmology, Gastroenterology, Urology, etc.) |
| By Sponsor Type | Medical Device Manufacturers Contract Research Organizations (CROs) Academic Medical Centers and Hospitals Government and Non-profit Sponsors |
| By Clinical Development Stage | Early Feasibility / First-in-Human Traditional Feasibility Pivotal / Confirmatory Post-Market / Real-World Evidence Generation |
| By Geography (Within the US) | Northeast Midwest South West |
| By Funding Source | Industry-funded Trials Government-funded Trials (e.g., NIH, BARDA) Private Equity and Venture-backed Trials Hybrid and Other Funding Models |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiovascular Device Trials | 120 | Cardiologists, Clinical Trial Managers |
| Orthopedic Device Trials | 90 | Orthopedic Surgeons, Research Coordinators |
| Diagnostic Device Trials | 100 | Laboratory Directors, Clinical Researchers |
| Neurological Device Trials | 80 | Neurologists, Clinical Research Associates |
| Regulatory Affairs in Device Trials | 60 | Regulatory Affairs Specialists, Compliance Officers |
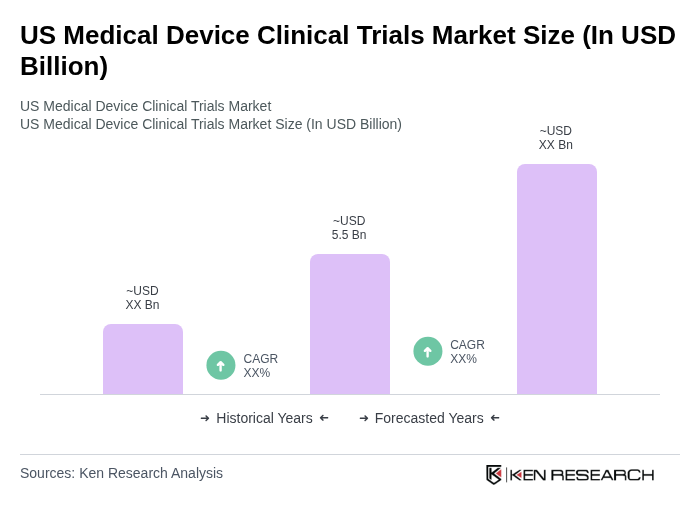
The US Medical Device Clinical Trials Market is valued at approximately USD 5.5 billion, reflecting a significant growth driven by the demand for innovative medical devices and advancements in technology, particularly in minimally invasive procedures and patient safety regulations.