Vietnam Mononucleosis Diagnostic Market Overview
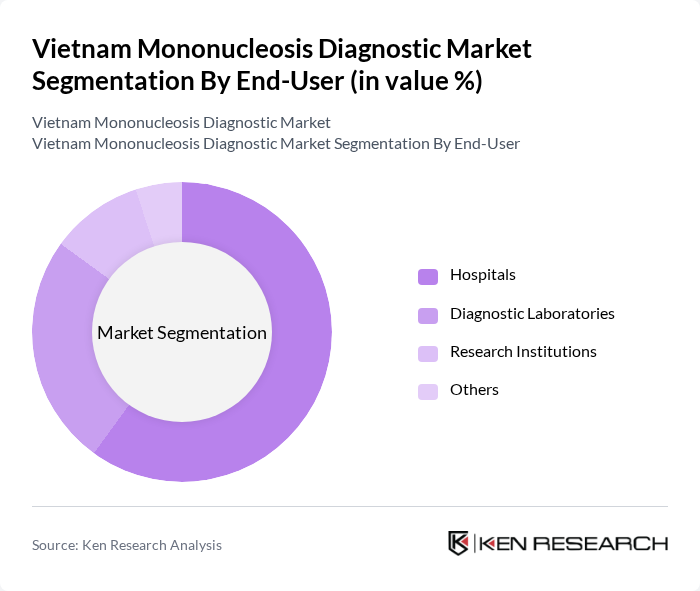
- The Vietnam Mononucleosis Diagnostic Market is valued at USD 1.6 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of infectious diseases, rising healthcare expenditure, and advancements in diagnostic technologies. The demand for accurate and rapid diagnostic tests has surged, particularly in urban areas where healthcare facilities are more accessible.
- Key cities such as Ho Chi Minh City, Hanoi, and Da Nang dominate the market due to their large populations and well-established healthcare infrastructure. These urban centers are equipped with advanced medical facilities and laboratories, which facilitate the rapid adoption of new diagnostic technologies and services, thereby driving market growth.
- The Law on Medical Examination and Treatment, 2023 issued by the National Assembly of Vietnam, mandates electronic health records and standardized diagnostic procedures for infectious diseases, including requirements for accurate testing methods and compliance by healthcare providers with minimum quality standards for laboratory diagnostics. This regulation aims to improve the accuracy of diagnoses and ensure that healthcare providers utilize reliable testing methods, thereby enhancing patient care and public health outcomes.
Vietnam Mononucleosis Diagnostic Market Segmentation
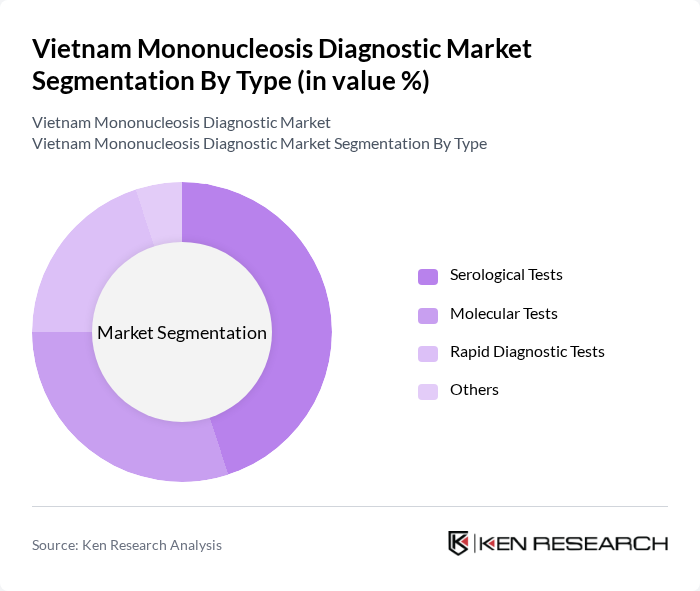
By Type:The market is segmented into various types of diagnostic tests, including serological tests, molecular tests, rapid diagnostic tests, and others. Among these, serological tests are currently the most dominant due to their widespread use in clinical settings for detecting antibodies related to mononucleosis. The ease of use and relatively low cost of serological tests make them a preferred choice for healthcare providers. Molecular tests are gaining traction due to their high sensitivity and specificity, while rapid diagnostic tests are favored for their quick results, especially in emergency settings.
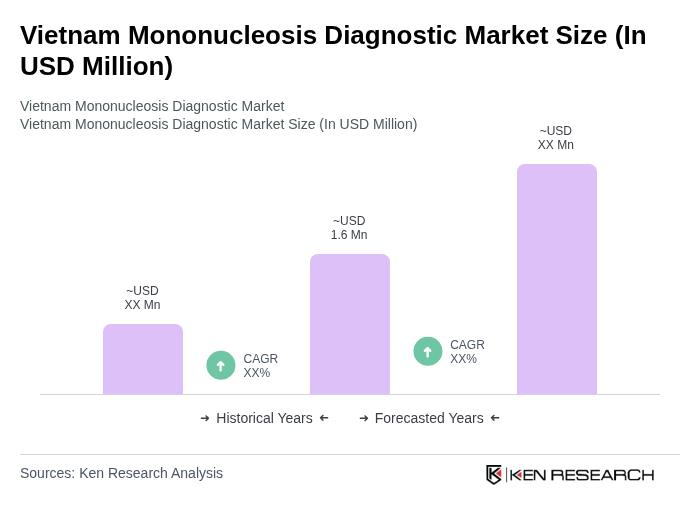
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, research institutions, and others. Hospitals are the leading end-users, as they require comprehensive diagnostic services for patient management. The increasing number of hospital admissions due to infectious diseases has led to a higher demand for diagnostic tests. Diagnostic laboratories also play a crucial role, providing specialized testing services. Research institutions contribute to the market by developing new diagnostic methods and technologies, while other end-users include clinics and private practices.
Vietnam Mononucleosis Diagnostic Market Competitive Landscape
The Vietnam Mononucleosis Diagnostic Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, Thermo Fisher Scientific, Ortho Clinical Diagnostics, Cepheid, Hologic, Quidel Corporation, Becton, Dickinson and Company, GenMark Diagnostics, PerkinElmer, Grifols, DiaSorin, Mylab Discovery Solutions contribute to innovation, geographic expansion, and service delivery in this space.
Vietnam Mononucleosis Diagnostic Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Infectious Diseases:The rise in infectious diseases in Vietnam, with over 1.6 million reported cases annually, significantly drives the demand for mononucleosis diagnostics. The World Health Organization reported that infectious diseases account for approximately 29% of total deaths in the country. This alarming statistic highlights the urgent need for effective diagnostic tools to manage and control these diseases, thereby boosting the mononucleosis diagnostic market.
- Rising Awareness about Mononucleosis:Public health campaigns have increased awareness of mononucleosis, leading to a surge in testing. In future, the Ministry of Health reported a 42% increase in diagnostic tests for mononucleosis compared to previous years. This heightened awareness is crucial as it encourages early detection and treatment, ultimately driving market growth. The focus on education about symptoms and transmission has empowered individuals to seek medical advice promptly.
- Advancements in Diagnostic Technologies:The Vietnam mononucleosis diagnostic market is benefiting from technological advancements, with the introduction of rapid diagnostic tests that can deliver results in under 25 minutes. The Ministry of Science and Technology has allocated approximately $12 million for research and development in diagnostic technologies in future. These innovations not only improve accuracy but also enhance accessibility, making it easier for healthcare providers to diagnose and treat patients effectively.
Market Challenges
- Limited Access to Healthcare Facilities:Access to healthcare facilities remains a significant challenge in Vietnam, particularly in rural areas where only 32% of the population has access to basic healthcare services. This limitation hampers the timely diagnosis of mononucleosis, as many patients are unable to reach healthcare providers. The lack of infrastructure and resources in these regions poses a barrier to effective disease management and market growth.
- High Cost of Advanced Diagnostic Tools:The cost of advanced diagnostic tools can be prohibitive, with prices ranging from $220 to $1,050 per unit. This high cost limits the availability of these tools in lower-income healthcare settings, where the need is greatest. As a result, many healthcare providers opt for less effective, traditional methods, which can delay diagnosis and treatment, ultimately affecting patient outcomes and market expansion.
Vietnam Mononucleosis Diagnostic Market Future Outlook
The Vietnam mononucleosis diagnostic market is poised for significant growth, driven by increasing healthcare investments and technological advancements. The government’s commitment to improving healthcare infrastructure, with an expected budget increase of 16% in future, will enhance access to diagnostic services. Additionally, the integration of artificial intelligence in diagnostics is anticipated to streamline processes, improve accuracy, and reduce costs, making diagnostics more accessible to the population. These trends indicate a promising future for the market.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Vietnamese government plans to invest $1.6 billion in healthcare infrastructure by future, creating opportunities for diagnostic companies to establish facilities in underserved areas. This expansion will enhance access to mononucleosis diagnostics, allowing for earlier detection and treatment, ultimately improving public health outcomes.
- Growing Demand for Rapid Diagnostic Tests:The demand for rapid diagnostic tests is increasing, with an estimated market value of $220 million in future. This trend presents opportunities for manufacturers to innovate and develop cost-effective, user-friendly tests that can be deployed in various healthcare settings, including remote areas, thus improving patient access to timely diagnosis.