Region:Asia
Author(s):Rebecca
Product Code:KRAC8467
Pages:88
Published On:November 2025
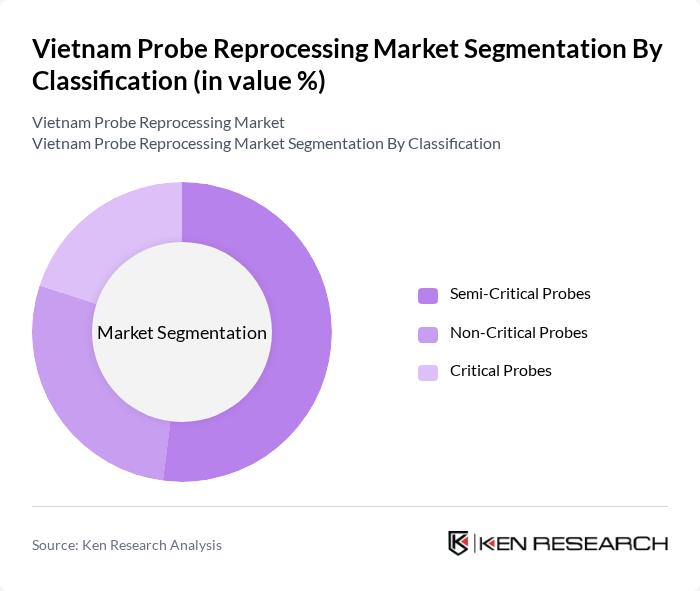
By Classification:The classification of probes into semi-critical, non-critical, and critical categories is essential for determining the appropriate reprocessing methods. Semi-critical probes, which come into contact with mucous membranes, require high-level disinfection, while non-critical probes only need low-level disinfection. Critical probes, which penetrate sterile tissues, must undergo sterilization. The demand for semi-critical probes remains high due to their extensive use in diagnostic and interventional procedures, particularly in cardiology, obstetrics, and emergency medicine .
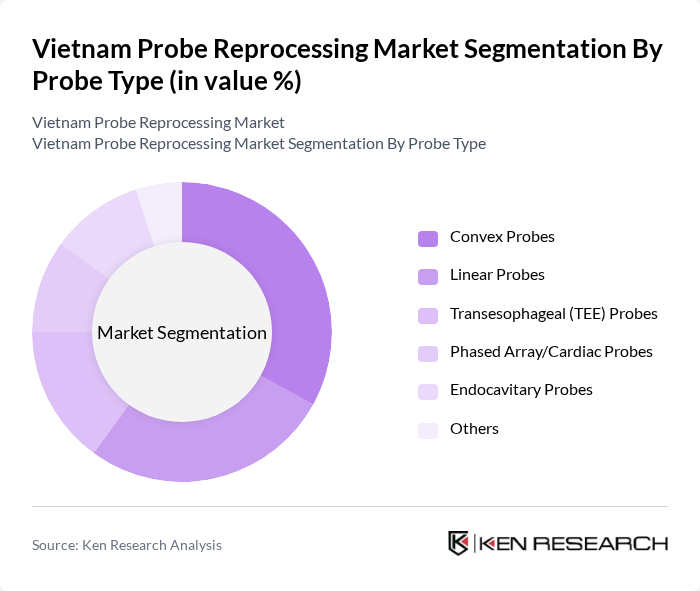
By Probe Type:The market is segmented by probe type, including convex probes, linear probes, transesophageal (TEE) probes, phased array/cardiac probes, endocavitary probes, and others. Convex probes are widely used in obstetrics and abdominal imaging, while linear probes are preferred for vascular and musculoskeletal applications. The adoption of advanced imaging techniques and the need for rapid, high-level disinfection have increased demand for specialized probes, with transesophageal probes gaining traction in cardiac and critical care settings .
The Vietnam Probe Reprocessing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Johnson & Johnson (including Ethicon and ASP), Stryker Corporation, Boston Scientific, B. Braun Melsungen AG, Smith & Nephew, CONMED Corporation, Terumo Corporation, Cook Medical, Olympus Corporation, Halyard Health (now part of Owens & Minor), 3M Company, GE HealthCare, Philips Healthcare, Siemens Healthineers, Nanosonics Limited, Germitec, Steris plc, Ecolab Inc., Medisafe International contribute to innovation, geographic expansion, and service delivery in this space.
The Vietnam probe reprocessing market is poised for significant growth as healthcare providers increasingly recognize the importance of sustainable practices. With the government's commitment to enhancing healthcare infrastructure and promoting eco-friendly solutions, the market is likely to see a surge in investments. Additionally, the integration of advanced technologies will streamline reprocessing operations, making them more efficient and cost-effective. As awareness grows, the adoption of reprocessed medical devices is expected to rise, aligning with global trends towards sustainability in healthcare.
| Segment | Sub-Segments |
|---|---|
| By Classification | Semi-Critical Probes Non-Critical Probes Critical Probes |
| By Probe Type | Convex Probes Linear Probes Transesophageal (TEE) Probes Phased Array/Cardiac Probes Endocavitary Probes Others |
| By Product | Instruments Consumables Services |
| By Reprocessing Method | Sterilization High-Level Disinfection (HLD) Intermediate-Level Disinfection Low-Level Disinfection |
| By End-Use | Hospitals and Clinics Diagnostic Imaging Centers Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam Others |
| By Regulatory Compliance | ISO Certification Local Regulatory Standards International Compliance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 65 | Procurement Managers, Supply Chain Coordinators |
| Reprocessing Facilities | 48 | Facility Managers, Quality Assurance Officers |
| Healthcare Consultants | 35 | Healthcare Strategy Advisors, Market Analysts |
| Medical Device Manufacturers | 42 | Product Managers, Regulatory Affairs Specialists |
| Government Health Officials | 28 | Policy Makers, Health Program Directors |
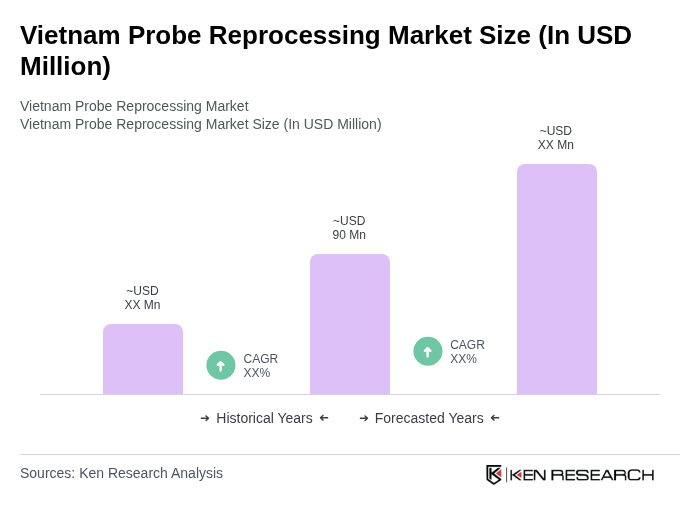
The Vietnam Probe Reprocessing Market is valued at approximately USD 90 million, driven by the increasing demand for minimally invasive procedures and stricter reprocessing protocols. This market is expected to grow as healthcare facilities invest in advanced disinfection technologies.