Region:Asia
Author(s):Dev
Product Code:KRAA3939
Pages:98
Published On:January 2026
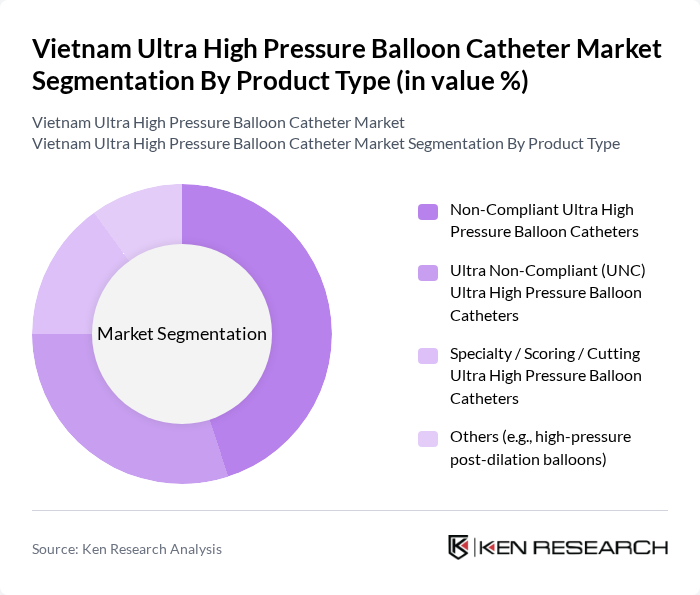
By Product Type:The product type segmentation includes Non-Compliant Ultra High Pressure Balloon Catheters, Ultra Non-Compliant (UNC) Ultra High Pressure Balloon Catheters, Specialty / Scoring / Cutting Ultra High Pressure Balloon Catheters, and Others (e.g., high-pressure post-dilation balloons), which is consistent with global ultra-high pressure balloon catheter classifications. Among these, Non-Compliant Ultra High Pressure Balloon Catheters dominate the market due to their widespread use in various interventional procedures, particularly in treating coronary artery diseases and resistant or calcified lesions. Their ability to withstand high pressures with minimal diameter change makes them a preferred choice for precise lesion preparation and post-dilation, leading to increased adoption in hospitals and high-volume cath labs.
By Application:The application segmentation includes Coronary Artery Disease (CAD) Interventions, Peripheral Artery Disease (PAD) Interventions, Renal and Urological Interventions, and Others (e.g., neurovascular, oncology), which aligns with the major clinical uses of high and ultra-high pressure balloon catheters globally. Coronary Artery Disease (CAD) Interventions lead the market due to the high and rising burden of CAD in Vietnam and across Asia Pacific, driven by lifestyle changes, urbanization, and an aging population. The increasing number of percutaneous coronary intervention and angioplasty procedures performed in tertiary hospitals, along with growing adoption of complex lesion techniques such as post-dilation and treatment of in?stent restenosis, has significantly boosted the demand for ultra-high pressure balloon catheters in this application.

The Vietnam Ultra High Pressure Balloon Catheter Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boston Scientific Corporation, Medtronic plc, Abbott Laboratories, B. Braun Melsungen AG, Terumo Corporation, Cook Medical LLC, Cordis, Johnson & Johnson MedTech, Asahi Intecc Co., Ltd., Merit Medical Systems, Inc., Cardinal Health, Inc., Koninklijke Philips N.V. (Philips Healthcare), Stryker Corporation, Edwards Lifesciences Corporation, Siemens Healthineers AG contribute to innovation, geographic expansion, and service delivery in this space.
As Vietnam's healthcare landscape evolves, the demand for ultra high pressure balloon catheters is expected to grow significantly. The integration of digital health technologies, such as telemedicine and remote monitoring, will enhance patient care and streamline procedures. Additionally, the shift towards outpatient care will likely increase the adoption of minimally invasive techniques, making these catheters more desirable. With ongoing investments in healthcare infrastructure, the market is poised for substantial growth, driven by both technological advancements and changing patient needs.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Non-Compliant Ultra High Pressure Balloon Catheters Ultra Non-Compliant (UNC) Ultra High Pressure Balloon Catheters Specialty / Scoring / Cutting Ultra High Pressure Balloon Catheters Others (e.g., high-pressure post-dilation balloons) |
| By Application | Coronary Artery Disease (CAD) Interventions Peripheral Artery Disease (PAD) Interventions Renal and Urological Interventions Others (e.g., neurovascular, oncology) |
| By Material | Nylon (Polyamide) Polyester (PET) Polyethylene and Copolymer Blends Others |
| By End-User | Tertiary Care Hospitals & Cardiac Centers Ambulatory Surgical Centers Specialized Cardiology & Vascular Clinics Others |
| By Distribution Channel | Direct Sales to Hospitals & Government Tenders Local Distributors / Importers Group Purchasing Organizations (GPOs) Others |
| By Pricing Tier | Premium High-Performance Catheters Mid-Range Catheters Value / Economy Catheters Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam Others |
| By Patient Demographics | Adult Patients Pediatric Patients Geriatric Patients Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Hospitals | 120 | Cardiologists, Interventional Radiologists |
| Medical Device Distributors | 80 | Sales Managers, Product Specialists |
| Healthcare Procurement Managers | 70 | Procurement Officers, Supply Chain Managers |
| Clinical Research Organizations | 60 | Clinical Researchers, Regulatory Affairs Specialists |
| Health Policy Makers | 50 | Health Economists, Policy Analysts |
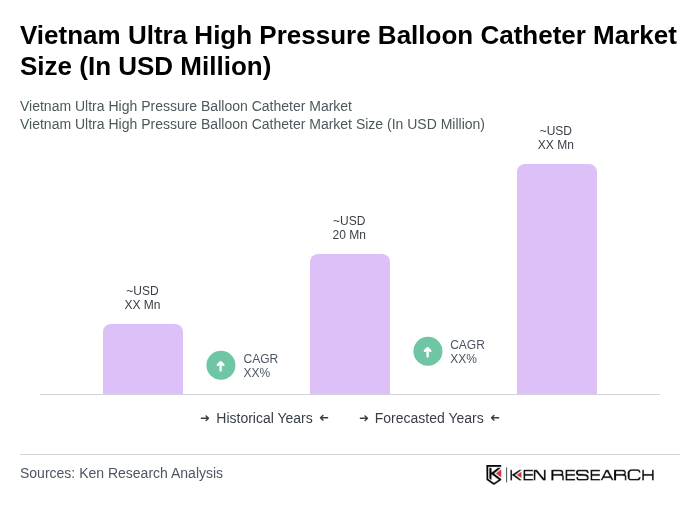
The Vietnam Ultra High Pressure Balloon Catheter Market is valued at approximately USD 20 million, reflecting a significant growth driven by the increasing prevalence of cardiovascular diseases and advancements in catheter technology.